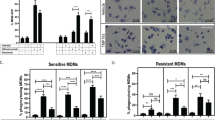
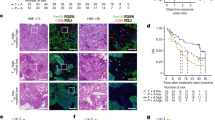
Abstract
Resistance to therapeutic antibodies in chronic lymphocytic leukaemia (CLL) is common. In this study, we show that therapeutic antibodies against CD62L (CD62L-Ab) or CD20 (obinutuzumab) were able to induce antibody-dependent cell-mediated cytotoxicity (ADCC) and phagocytosis (ADP) in primary cultures of CLL cells. CLL cells derived from patients with active disease requiring treatment displayed resistance to these antibodies, whereas patients with stable disease were sensitive. Using enrichment strategies and transcriptomic analyses, we show that antibody-dependent tumour cell killing was FcγR-dependent and mediated by macrophages. Moreover, we show that resistance cannot be attributed to total numbers or established subtypes of monocytes/macrophages, or the efficiency with which they bind an immune complex. Rather, ADCC/ADP resistance was due to reduced signalling activity through the activating FcγRs resulting in the transfer of dominance to the inhibitory FcγRIIb within macrophages. Most significantly, we show that resistance is an actionable event that could be reversed using inhibitors of FcγRIIb signalling in primary cultures of CLL cells that were previously insensitive to obinutuzumab or CD62L-Ab.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Burger JA, Okkenhaug K . Haematological cancer: idelalisib-targeting PI3Kdelta in patients with B-cell malignancies. Nat Rev Clin Oncol 2014; 11: 184–186.
Awan FT, Byrd JC . New strategies in chronic lymphocytic leukemia: shifting treatment paradigms. Clin Cancer Res 2014; 20: 5869–5874.
Robak T . Current and emerging monoclonal antibody treatments for chronic lymphocytic leukemia: state of the art. Expert Rev Hematol 2014; 7: 841–857.
Hallek M . Signaling the end of chronic lymphocytic leukemia: new frontline treatment strategies. Blood 2013; 122: 3723–3734.
Furman RR, Sharman JP, Coutre SE, Cheson BD, Pagel JM, Hillmen P et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med 2014; 370: 997–1007.
Arbones ML, Ord DC, Ley K, Ratech H, Maynard-Curry C, Otten G et al. Lymphocyte homing and leukocyte rolling and migration are impaired in L-selectin-deficient mice. Immunity 1994; 1: 247–260.
Burgess M, Gill D, Singhania R, Cheung C, Chambers L, Renyolds BA et al. CD62L as a Therapeutic Target in Chronic Lymphocytic Leukemia. Clin Cancer Res 2013; 19: 5675–5685.
Gu B, Dao LP, Wiley J . Impaired transendothelial migration of B-CLL lymphocytes: a defect linked to low L-selectin expression. Leuk Lymphoma 2001; 42: 5–12.
Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell'Aquila M, Kipps TJ . Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood 2000; 96: 2655–2663.
Tsukada N, Burger JA, Zvaifler NJ, Kipps TJ . Distinctive features of ‘nurselike’ cells that differentiate in the context of chronic lymphocytic leukemia. Blood 2002; 99: 1030–1037.
Bhattacharya N, Diener S, Idler IS, Rauen J, Habe S, Busch H et al. Nurse-like cells show deregulated expression of genes involved in immunocompetence. Br J Haematol 2011; 154: 349–356.
Boissard F, Fournie JJ, Laurent C, Poupot M, Ysebaert L . Nurse like cells: chronic lymphocytic leukemia associated macrophages. Leuk Lymphoma 2015; 56: 1570–1572.
Filip AA, Cisel B, Koczkodaj D, Wasik-Szczepanek E, Piersiak T, Dmoszynska A . Circulating microenvironment of CLL: are nurse-like cells related to tumor-associated macrophages? Blood Cells Mol Dis 2013; 50: 263–270.
Giannoni P, Pietra G, Travaini G, Quarto R, Shyti G, Benelli R et al. Chronic lymphocytic leukemia nurse-like cells express hepatocyte growth factor receptor (c-MET) and indoleamine 2,3-dioxygenase and display features of immunosuppressive type 2 skewed macrophages. Haematologica 2014; 99: 1078–1087.
Ysebaert L, Fournie JJ . Genomic and phenotypic characterization of nurse-like cells that promote drug resistance in chronic lymphocytic leukemia. Leuk Lymphoma 2011; 52: 1404–1406.
Burgess M, Ellis JJ, Mapp S, Mollee P, Mazzieri R, Mattarollo SR et al. Transcriptomic analysis of monocytes and macrophages derived from CLL patients which display differing abilities to respond to therapeutic antibody immune complexes. Genomics Data 2016; 7: 4–6.
Burgess M, Cheung C, Chambers L, Ravindranath K, Minhas G, Knop L et al. CCL2 and CXCL2 enhance survival of primary chronic lymphocytic leukemia cells in vitro. Leuk Lymphoma 2012; 53: 1988–1998.
Martinez FO, Gordon S, Locati M, Mantovani A . Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol 2006; 177: 7303–7311.
Cartron G, Watier H, Golay J, Solal-Celigny P . From the bench to the bedside: ways to improve rituximab efficacy. Blood 2004; 104: 2635–2642.
Herter S, Birk MC, Klein C, Gerdes C, Umana P, Bacac M . Glycoengineering of therapeutic antibodies enhances monocyte/macrophage-mediated phagocytosis and cytotoxicity. J Immunol 2014; 192: 2252–2260.
Nimmerjahn F, Ravetch JV . Antibodies, Fc receptors and cancer. Curr Opin Immunol 2007; 19: 239–245.
Nimmerjahn F, Ravetch JV . Fcgamma receptors as regulators of immune responses. Nat Rev Immunol 2008; 8: 34–47.
Pallasch CP, Leskov I, Braun CJ, Vorholt D, Drake A, Soto-Feliciano YM et al. Sensitizing protective tumor microenvironments to antibody-mediated therapy. Cell 2014; 156: 590–602.
Roghanian A, Teige I, Martensson L, Cox KL, Kovacek M, Ljungars A et al. Antagonistic human FcgammaRIIB (CD32B) antibodies have anti-tumor activity and overcome resistance to antibody therapy in vivo. Cancer Cell 2015; 27: 473–488.
DiLillo DJ, Ravetch JV . Differential Fc-receptor engagement drives an anti-tumor vaccinal effect. Cell 2015; 161: 1035–1045.
Bournazos S, Woof JM, Hart SP, Dransfield I . Functional and clinical consequences of Fc receptor polymorphic and copy number variants. Clin Exp Immunol 2009; 157: 244–254.
Mellor JD, Brown MP, Irving HR, Zalcberg JR, Dobrovic A . A critical review of the role of Fc gamma receptor polymorphisms in the response to monoclonal antibodies in cancer. J Hematol Oncol 2013; 6: 1.
Morabito F, Gentile M, Seymour JF, Polliack A . Ibrutinib, idelalisib and obinutuzumab for the treatment of patients with chronic lymphocytic leukemia: three new arrows aiming at the target. Leuk Lymphoma 2015; 56: 3250–3256.
Patz M, Isaeva P, Forcob N, Muller B, Frenzel LP, Wendtner CM et al. Comparison of the in vitro effects of the anti-CD20 antibodies rituximab and GA101 on chronic lymphocytic leukaemia cells. Br J Haematol 2011; 152: 295–306.
Burger JA, Keating MJ, Wierda WG, Hartmann E, Hoellenriegel J, Rosin NY et al. Safety and activity of ibrutinib plus rituximab for patients with high-risk chronic lymphocytic leukaemia: a single-arm, phase 2 study. Lancet Oncol 2014; 15: 1090–1099.
Jaglowski SM, Jones JA, Nagar V, Flynn JM, Andritsos LA, Maddocks KJ et al. Safety and activity of BTK inhibitor ibrutinib combined with ofatumumab in chronic lymphocytic leukemia: a phase 1b/2 study. Blood 2015; 126: 842–850.
Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Dohner H et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 2008; 111: 5446–5456.
Acknowledgements
We gratefully acknowledge the patients who donated samples for this study. We also gratefully acknowledge the constructive comments of Professor Maher Gandhi before submission. NS is supported by grants from the Australian NHMRC (#APP1049182) and the Cancer Council Queensland (#APP1025479). NS is supported by a Senior Research Fellowship awarded by the Cancer Council Queensland.
Author contributions
MB designed and performed the research, analysed the data and wrote the paper; SM designed the research, analysed the data, provided the patient samples and wrote the paper. RM and SRM designed the research and edited the manuscript; CC and LC designed and performed the research; PM provided the patient samples, analysed the clinical data and edited the manuscript; DG supervised the study, designed the research, provided the patient samples and edited the manuscript; NAS supervised the study, designed the research, analysed the data and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Burgess, M., Mapp, S., Mazzieri, R. et al. Increased FcγRIIB dominance contributes to the emergence of resistance to therapeutic antibodies in chronic lymphocytic leukaemia patients. Oncogene 36, 2366–2376 (2017). https://doi.org/10.1038/onc.2016.387
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.387