Abstract
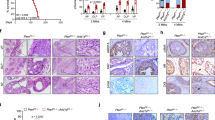
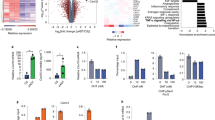
Ovarian cancer G-protein-coupled-receptor-1 (OGR1) is a tumor metastasis suppressor in prostate cancer (PCa). OGR1 knockout mice (ogr1−/−) are grossly normal under physiological conditions, however, reduced melanoma tumorigenesis has been observed, with the mechanisms of this reduction completely unknown. In this work, we demonstrated that OGR1 deficiency in host cells significantly reduced tumorigenesis of PCa in mice. Adoptive transfer of WT CD11b+ Gr1+ double positive (DP) cells, but not T cells, was sufficient to allow tumor development in ogr1−/− mice. The expression of an M1 macrophage marker, inducible nitric oxide synthase (iNOS) was higher and expression of an M2 macrophage marker, arginase-1 (Arg 1) was lower in tumors from ogr1−/− mice compared with WT mice. Furthermore, coinjection of transgenic adenocarcinoma mouse prostate (TRAMP)-C2 cells with WT, but not ogr1−/− macrophages, increased tumor incidence in ogr1−/− mice. T-cell depletion experiments suggested that T cells were required for tumor rejection in ogr1−/−mice, although OGR1 expression in T cells may not be necessary. In summary, the expression of OGR1 in myeloid-derived cells, especially in DP cells, was required for PCa tumor cell-induced immunosuppression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ludwig MG, Vanek M, Guerini D, Gasser JA, Jones CE, Junker U et al. Proton-sensing G-protein-coupled receptors. Nature 2003; 425: 93–98.
Im DS . Two ligands for a GPCR, proton vs lysolipid. Acta Pharmacol Sin 2005; 26: 1435–1441.
Singh LS, Berk M, Oates R, Zhao Z, Tan H, Jiang Y et al. Ovarian cancer G protein-coupled receptor 1, a new metastasis suppressor gene in prostate cancer. J Natl Cancer Inst 2007; 99: 1313–1327.
Li H, Wang D, Singh LS, Berk M, Tan H, Zhao Z et al. Abnormalities in osteoclastogenesis and decreased tumorigenesis in mice deficient for ovarian cancer G protein-coupled receptor 1. PLoS One 2009; 4: e5705.
Thompson TC, Southgate J, Kitchener G, Land H . Multistage carcinogenesis induced by ras and myc oncogenes in a reconstituted organ. Cell 1989; 56: 917–930.
Foster BA, Gingrich JR, Kwon ED, Madias C, Greenberg NM . Characterization of prostatic epithelial cell lines derived from transgenic adenocarcinoma of the mouse prostate (TRAMP) model. Cancer Res 1997; 57: 3325–3330.
Ostrand-Rosenberg S, Sinha P, Beury DW, Clements VK . Cross-talk between myeloid-derived suppressor cells (MDSC), macrophages, and dendritic cells enhances tumor-induced immune suppression. Semin Cancer Biol 2012; 22: 275–281.
Talmadge JE . Pathways mediating the expansion and immunosuppressive activity of myeloid-derived suppressor cells and their relevance to cancer therapy. Clin Cancer Res 2007; 13 (18 Pt 1): 5243–5248.
Nagaraj S, Schrum AG, Cho HI, Celis E, Gabrilovich DI . Mechanism of T cell tolerance induced by myeloid-derived suppressor cells. J Immunol 2010; 184: 3106–3116.
Ostrand-Rosenberg S . Myeloid-derived suppressor cells: more mechanisms for inhibiting antitumor immunity. Cancer Immunol Immunother 2010; 59: 1593–1600.
Luczynski W, Krawczuk-Rybak M, Stasiak-Barmuta A . [Myeloid-derived suppressor cells - the new mechanism of immunosuppression in cancer]. Postepy Hig Med Dosw (Online) 2008; 62: 18–22.
Nagaraj S, Youn JI, Weber H, Iclozan C, Lu L, Cotter MJ et al. Anti-inflammatory triterpenoid blocks immune suppressive function of MDSCs and improves immune response in cancer. Clin Cancer Res 2010; 16: 1812–1823.
Nefedova Y, Fishman M, Sherman S, Wang X, Beg AA, Gabrilovich DI . Mechanism of all-trans retinoic acid effect on tumor-associated myeloid-derived suppressor cells. Cancer Res 2007; 67: 11021–11028.
Fricke I, Mirza N, Dupont J, Lockhart C, Jackson A, Lee JH et al. Vascular endothelial growth factor-trap overcomes defects in dendritic cell differentiation but does not improve antigen-specific immune responses. Clin Cancer Res 2007; 13: 4840–4848.
Koide N, Nishio A, Sato T, Sugiyama A, Miyagawa S . Significance of macrophage chemoattractant protein-1 expression and macrophage infiltration in squamous cell carcinoma of the esophagus. Am J Gastroenterol 2004; 99: 1667–1674.
Silliman CC, Dickey WO, Paterson AJ, Thurman GW, Clay KL, Johnson CA et al. Analysis of the priming activity of lipids generated during routine storage of platelet concentrates. Transfusion 1996; 36: 133–139.
Biswas SK, Sica A, Lewis CE . Plasticity of macrophage function during tumor progression: regulation by distinct molecular mechanisms. J Immunol 2008; 180: 2011–2017.
Umemura N, Saio M, Suwa T, Kitoh Y, Bai J, Nonaka K et al. Tumor-infiltrating myeloid-derived suppressor cells are pleiotropic-inflamed monocytes/macrophages that bear M1- and M2-type characteristics. J Leukoc Biol 2008; 83: 1136–1144.
Hasita H, Komohara Y, Okabe H, Masuda T, Ohnishi K, Lei XF et al. Significance of alternatively activated macrophages in patients with intrahepatic cholangiocarcinoma. Cancer Sci 2010; 101: 1913–1919.
Baley PA, Yoshida K, Qian W, Sehgal I, Thompson TC . Progression to androgen insensitivity in a novel in vitro mouse model for prostate cancer. J Steroid Biochem Mol Biol 1995; 52: 403–413.
Xu Y, Casey G . Identification of human OGR1, a novel G protein-coupled receptor that maps to chromosome 14. Genomics 1996; 35: 397–402.
Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P et al. HIF-1alpha regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J Exp Med 2010; 207: 2439–2453.
Eruslanov E, Daurkin I, Ortiz J, Vieweg J, Kusmartsev S . Pivotal Advance: tumor-mediated induction of myeloid-derived suppressor cells and M2-polarized macrophages by altering intracellular PGE2 catabolism in myeloid cells. J Leukoc Biol 2010; 88: 839–848.
Elkabets M, Ribeiro VS, Dinarello CA, Ostrand-Rosenberg S, Di Santo JP, Apte RN et al. IL-1beta regulates a novel myeloid-derived suppressor cell subset that impairs NK cell development and function. Eur J Immunol 2010; 40: 3347–3357.
Hong DS, Angelo LS, Kurzrock R . Interleukin-6 and its receptor in cancer: implications for translational therapeutics. Cancer 2007; 110: 1911–1928.
LaTulippe E, Satagopan J, Smith A, Scher H, Scardino P, Reuter V et al. Comprehensive gene expression analysis of prostate cancer reveals distinct transcriptional programs associated with metastatic disease. Cancer Res 2002; 62: 4499–4506.
Gingrich JR, Barrios RJ, Morton RA, Boyce BF, DeMayo FJ, Finegold MJ et al. Metastatic prostate cancer in a transgenic mouse. Cancer Res 1996; 56: 4096–4102.
Rauhala HE, Porkka KP, Saramaki OR, Tammela TL, Visakorpi T . Clusterin is epigenetically regulated in prostate cancer. Int J Cancer 2008; 123: 1601–1609.
Fasso M, Waitz R, Hou Y, Rim T, Greenberg NM, Shastri N et al. SPAS-1 (stimulator of prostatic adenocarcinoma-specific T cells)/SH3GLB2: a prostate tumor antigen identified by CTLA-4 blockade. Proc Nat Acad Sci Usa 2008; 105: 3509–3514.
Grossmann ME, Wood M, Celis E . Expression, specificity and immunotherapy potential of prostate-associated genes in murine cell lines. World J Urol 2001; 19: 365–370.
Zhang Q, Yang X, Pins M, Javonovic B, Kuzel T, Kim SJ et al. Adoptive transfer of tumor-reactive transforming growth factor-beta-insensitive CD8+ T cells: eradication of autologous mouse prostate cancer. Cancer Res 2005; 65: 1761–1769.
Babiarova K, Kutinova L, Zurkova K, Krystofova J, Brabcova E, Hainz P et al. Immunization with WT1-derived peptides by tattooing inhibits the growth of TRAMP-C2 prostate tumor in mice. J Immunother 2012; 35: 478–487.
Pearson TC, Darby CR, Bushell AR, West LJ, Morris PJ, Wood KJ . The assessment of transplantation tolerance induced by anti-CD4 monoclonal antibody in the murine model. Transplantation 1993; 55: 361–367.
Yin DP, Ma LL, Sankary HN, Shen J, Zeng H, Varghese A et al. Role of CD4+ and CD8+ T cells in the rejection of concordant pancreas xenografts. Transplantation 2002; 74: 1236–1241.
Acknowledgements
We thank Mrs Helen Jan Chin-Sinex from Dr Marc S. Mendonca's lab at Indiana University School of Medicine for technical assistance in the preparation of irradiated TRAMP-C2 tumor cells. We thank Mrs Huiying Gu from Dr Yansheng Du's lab for technical assistance in the preparation of tumor frozen sections. We also thank Dr Caryl Antalis and Dr Cheryl Dsouza for critical reading and editing of the manuscript. This work was supported in part by the National Institutes of Health (RO1 155145 to YX and LZ); and the Mary Fendrich-Hulman Charitable Trust Fund to YX.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Yan, L., Singh, L., Zhang, L. et al. Role of OGR1 in myeloid-derived cells in prostate cancer. Oncogene 33, 157–164 (2014). https://doi.org/10.1038/onc.2012.566
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.566
Keywords
This article is cited by
-
Role of pH-sensing receptors in colitis
Pflügers Archiv - European Journal of Physiology (2024)
-
GPR68 limits the severity of chemical-induced oral epithelial dysplasia
Scientific Reports (2023)
-
Metastasis suppressor genes in clinical practice: are they druggable?
Cancer and Metastasis Reviews (2023)
-
Ovarian cancer G protein-coupled receptor 1 inhibits A549 cells migration through casein kinase 2α intronless gene and neutral endopeptidase
BMC Cancer (2022)
-
Physiological relevance of proton-activated GPCRs
Pflügers Archiv - European Journal of Physiology (2022)