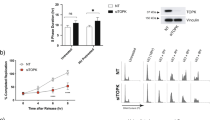
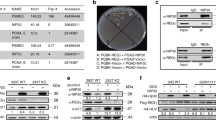
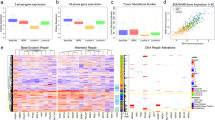
Abstract
DNA damage checkpoints cause cell cycle arrest, allowing DNA repair before resumption of the cell cycle. These checkpoints can be activated through several signaling pathways. Checkpoint activators include p53, checkpoint kinase 1 (CHK1), checkpoint kinase 2 and/or MAPKAP kinase 2 (MK2). Many cancer cells lack p53 activity and, therefore, depend on alternative checkpoint activators to arrest the cell cycle following DNA damage. Inhibition of these pathways is expected to specifically sensitize these p53-deficient cells to DNA damage caused by chemotherapy. Using isogenic p53-proficient and p53-deficient cancer cell lines, we show that inactivation of CHK1, but not MK2, abrogates cell cycle arrest following chemotherapy, specifically in p53-deficient cells. However, we show that CHK1 is required to maintain genome integrity and cell viability, and that p53-proficient cells are no less sensitive than p53-deficient cells to CHK1 inhibition in the presence of DNA damage. Thus, combining CHK1 inhibition with DNA damage does not lead to preferential killing of p53-deficient over p53-proficient cells, and inhibiting CHK1 does not appear to be a promising approach for potentiation of cancer chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Abraham RT . (2001). Cell cycle checkpoint signaling through the ATM and ATR kinases. Genes Dev 15: 2177–2196.
Ashwell S, Janetka JW, Zabludoff S . (2008). Keeping checkpoint kinases in line: new selective inhibitors in clinical trials. Expert Opin Investig Drugs 17: 1331–1340.
Bartek J, Lukas J . (2003). Chk1 and Chk2 kinases in checkpoint control and cancer. GGGGZ 3: 421–429.
Brummelkamp TR, Bernards R, Agami R . (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296: 550–553.
Bucher N, Britten CD . (2008). G2 checkpoint abrogation and checkpoint kinase-1 targeting in the treatment of cancer. Br J Cancer 98: 523–528.
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP et al. (1998). Requirement for p53 and p21 to Sustain G2 Arrest After DNA Damage. Science 282: 1497–1501.
Chen Z, Xiao Z, Gu WZ, Xue J, Bui MH, Kovar P et al. (2006). Selective Chk1 inhibitors differentially sensitize p53-deficient cancer cells to cancer therapeutics. Int J Cancer 119: 2784–2794.
Cho SH, Toouli CD, Fujii GH, Crain C, Parry D . (2005). Chk1 is essential for tumor cell viability following activation of the replication checkpoint. Cell Cycle 4: 131–139.
Dai J, Sultan S, Taylor SS, Higgins JM . (2005). The kinase haspin is required for mitotic histone H3 Thr 3 phosphorylation and normal metaphase chromosome alignment. Genes Dev 19: 472–488.
Dent MF, Hubbold L, Radford H, Wilson AP . (1995). The methylene blue colorimetric microassay for determining cell line response to growth factors. Cytotechnology 17: 27–33.
Donzelli M, Draetta GF . (2003). Regulating mammalian checkpoints through Cdc25 inactivation. EMBO Rep 4: 671–677.
Falck J, Mailand N, Syljuåsen RG, Bartek J, Lukas J . (2001). The ATM–Chk2–Cdc25A checkpoint pathway guards against radioresistant DNA synthesis. Nature 410: 842–847.
Finkel E . (1999). Does cancer therapy trigger cell suicide? Science 286: 2256–2258.
Harris SL, Levine AJ . (2005). The p53 pathway: positive and negative feedback loops. Oncogene 24: 2899–2908.
Hirose Y, Berger MS, Pieper RO . (2001). Abrogation of the Chk1-mediated G(2) checkpoint pathway potentiates temozolomide-induced toxicity in a p53-independent manner in human glioblastoma cells. Cancer Res 61: 5843–5849.
Karlsson-Rosenthal C, Millar JB . (2006). Cdc25: mechanisms of checkpoint inhibition and recovery. Trends Cell Biol 16: 285–292.
Kawabe T . (2004). G2 checkpoint abrogators as anticancer drugs. Mol Cancer Ther 3: 513–519.
Kravchenko-Balasha N, Mizrachy-Schwartz S, Klein S, Levitzki A . (2009). Shift from apoptotic to necrotic cell death during human papillomavirus-induced transformation of keratinocytes. J Biol Chem 284: 11717–11727.
Levesque AA, Fanous AA, Poh A, Eastman A . (2008). Defective p53 signaling in p53 wild-type tumors attenuates p21waf1 induction and cyclin B repression rendering them sensitive to Chk1 inhibitors that abrogate DNA damage-induced S and G2 arrest. Mol Cancer Ther 7: 252–262.
Lieberman HB . (2008). DNA damage repair and response proteins as targets for cancer therapy. Curr Med Chem 15: 360–367.
Manke IA, Nguyen A, Lim D, Stewart MQ, Elia AE, Yaffe MB . (2005). MAPKAP kinase-2 is a cell cycle checkpoint kinase that regulates the G2/M transition and S phase progression in response to UV irradiation. Mol Cell 17: 37–48.
Meek DW . (2004). The p53 response to DNA damage. DNA Repair (Amst) 3: 1049–1056.
O'Connor MJ, Martin NM, Smith GC . (2007). Targeted cancer therapies based on the inhibition of DNA strand break repair. Oncogene 26: 7816–7824.
Oren M . (2003). Decision making by p53: life, death and cancer. Cell Death Differ 10: 431–442.
Petermann E, Caldecott KW . (2006). Evidence that the ATR/Chk1 pathway maintains normal replication fork progression during unperturbed S phase. Cell Cycle 5: 2203–2209.
Raver-Shapira N, Marciano E, Meiri E, Spector Y, Rosenfeld N, Moskovits N et al. (2007). Transcriptional activation of miR-34a contributes to p53-mediated apoptosis. Mol Cell 26: 731–743.
Reinhardt HC, Aslanian AS, Lees JA, Yaffe MB . (2007). p53-deficient cells rely on ATM- and ATR-mediated checkpoint signaling through the p38MAPK/MK2 pathway for survival after DNA damage. Cancer Cell 11: 175–189.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM . (1998). DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 273: 5858–5868.
Senderowicz AM . (2003). Novel direct and indirect cyclin-dependent kinase modulators for the prevention and treatment of human neoplasms. Cancer Chemother Pharmacol 52 (Suppl 1): S61–S73.
Shao RG, Cao CX, Shimizu T, O'Connor PM, Kohn KW, Pommier Y . (1997). Abrogation of an S-phase checkpoint and potentiation of camptothecin cytotoxicity by 7-hydroxystaurosporine (UCN-01) in human cancer cell lines, possibly influenced by p53 function. Cancer Res 57: 4029–4035.
Shiloh Y . (2003). ATM and related protein kinases: safeguarding genome integrity. Nat Rev Cancer 3: 155–168.
Stark GR, Taylor WR . (2006). Control of the G2/M transition. Mol Biotechnol 32: 227–248.
Tao Y, Leteur C, Yang C, Zhang P, Castedo M, Pierré A et al. (2009). Radiosensitization by Chir-124, a selective CHK1 inhibitor: effects of p53 and cell cycle checkpoints. Cell Cycle 15: 1196–1205.
Tse AN, Schwartz GK . (2004). Potentiation of cytotoxicity of topoisomerase i poison by concurrent and sequential treatment with the checkpoint inhibitor UCN-01 involves disparate mechanisms resulting in either p53-independent clonogenic suppression or p53-dependent mitotic catastrophe. Cancer Res 64: 6635–6644.
Vakifahmetoglu H, Olsson M, Zhivotovsky B . (2008). Death through a tragedy: mitotic catastrophe. Cell Death Differ 15: 1153–1162.
Vogelstein B, Lane D, Levine AJ . (2000). Surfing the p53 network. Nature 408: 307–310.
Wang Q, Fan S, Eastman A, Worland PJ, Sausville EA, O'Connor PM . (1996). UCN-01: a potent abrogator of G2 checkpoint function in cancer cells with disrupted p53. J Natl Cancer Inst 88: 956–965.
Xiao Z, Xue J, Sowin TJ, Zhang H . (2006). Differential roles of checkpoint kinase 1, checkpoint kinase 2, and mitogen-activated protein kinase-activated protein kinase 2 in mediating DNA damage-induced cell cycle arrest: implications for cancer therapy. Mol Cancer Ther 5: 1935–1943.
Zhang WH, Poh A, Fanous AA, Eastman A . (2008). DNA damage-induced S phase arrest in human breast cancer depends on Chk1, but G2 arrest can occur independently of Chk1, Chk2 or MAPKAPK2. Cell Cycle 7: 1668–1677.
Zhou BB, Elledge SJ . (2000). The DNA damage response: putting checkpoints in perspective. Nature 408: 433–439.
Zhou BB, Anderson HJ, Roberge M . (2003). Targeting DNA checkpoint kinases in cancer therapy. Cancer Biol Ther 2: S16–S22.
Zhou BB, Bartek J . (2004). Targeting the checkpoint kinases: chemosensitization versus chemoprotection. Nat Rev Cancer 4: 216–225.
Acknowledgements
We would like to thank Dr Shoshana Klein for discussions and editing. This work was partially supported by the Lady Davis Fellowship Trust and by Algen Biopharmaceuticals, Ltd.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Zenvirt, S., Kravchenko-Balasha, N. & Levitzki, A. Status of p53 in human cancer cells does not predict efficacy of CHK1 kinase inhibitors combined with chemotherapeutic agents. Oncogene 29, 6149–6159 (2010). https://doi.org/10.1038/onc.2010.343
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.343
Keywords
This article is cited by
-
Combined inhibition of the cell cycle related proteins Wee1 and Chk1/2 induces synergistic anti-cancer effect in melanoma
BMC Cancer (2015)
-
Death of p53-defective cells triggered by forced mitotic entry in the presence of DNA damage is not uniquely dependent on Caspase-2 or the PIDDosome
Cell Death & Disease (2013)
-
Checkpoint kinase 1 in DNA damage response and cell cycle regulation
Cellular and Molecular Life Sciences (2013)
-
Efficacy of CHK inhibitors as single agents in MYC-driven lymphoma cells
Oncogene (2012)
-
ATR mediates cisplatin resistance in a p53 genotype-specific manner
Oncogene (2011)