Abstract
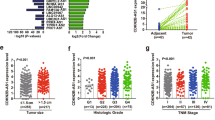
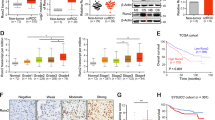
The Wnt signaling pathway is involved in normal embryonic development and controls the homeostatic self-renewal of stem cells in adult tissues. Constitutive activation of Wnt signaling contributes to cancer development and progression. We identified a CXXC4 homozygous deletion at 4q24 in an aggressive renal cell carcinoma (RCC) using single-nucleotide polymorphism (SNP) arrays. CXXC4 encodes Idax, which negatively regulates Wnt signaling by binding to the PDZ domain of Dishevelled. CXXC4 mRNA levels in tumor samples were significantly lower in patients with metastases compared with those without (P=0.0016). Patients whose tumors had lower CXXC4 expression than normal kidney showed a poorer cause-specific survival outcome than those with higher expression (P=0.0095). Decreased expression of CXXC4 also correlated with cytoplasmic staining of β-catenin. Knockdown of CXXC4 induced the nuclear translocation of β-catenin and altered expression of a set of genes involved in cell proliferation, invasion and survival. Furthermore, reduced expression of CXXC4 by small interfering RNAs promoted cell proliferation and inhibited apoptosis after 5-FU and doxorubicin treatment in RCC cells. These data suggest that CXXC4 plays a critical role in tumor progression of RCC through Wnt signaling. Wnt signaling could thus be a potential molecular target in RCC indicating decreased CXXC4 expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Barker N, Clevers H . (2006). Mining the Wnt pathway for cancer therapeutics. Nat Rev Drug Discov 5: 997–1014.
Bignell GR, Huang J, Greshock J, Watt S, Butler A, West S et al. (2004). High-resolution analysis of DNA copy number using oligonucleotide microarrays. Genome Res 14: 287–295.
Bilim V, Kawasaki T, Katagiri A, Wakatsuki S, Takahashi K, Tomita Y . (2000). Altered expression of beta-catenin in renal cell cancer and transitional cell cancer with the absence of beta-catenin gene mutations. Clin Cancer Res 6: 460–466.
Cohen HT, McGovern FJ . (2005). Renal-cell carcinoma. N Engl J Med 353: 2477–2490.
Crnkovic-Mertens I, Wagener N, Semzow J, Grone EF, Haferkamp A, Hohenfellner M et al. (2007). Targeted inhibition of Livin resensitizes renal cancer cells towards apoptosis. Cell Mol Life Sci 64: 1137–1144.
Dahl E, Wiesmann F, Woenckhaus M, Stoehr R, Wild PJ, Veeck J et al. (2007). Frequent loss of SFRP1 expression in multiple human solid tumours: association with aberrant promoter methylation in renal cell carcinoma. Oncogene 26: 5680–5691.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M et al. (2007). Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356: 125–134.
Gavert N, Conacci-Sorrell M, Gast D, Schneider A, Altevogt P, Brabletz T et al. (2005). L1, a novel target of beta-catenin signaling, transforms cells and is expressed at the invasive front of colon cancers. J Cell Biol 168: 633–642.
Guinan P, Sobin LH, Algaba F, Badellino F, Kameyama S, MacLennan G et al. (1997). TNM staging of renal cell carcinoma: Workgroup No. 3. Union International Contre le Cancer (UICC) and the American Joint Committee on Cancer (AJCC). Cancer 80: 992–993.
Gumz ML, Zou H, Kreinest PA, Childs AC, Belmonte LS, LeGrand SN et al. (2007). Secreted frizzled-related protein 1 loss contributes to tumor phenotype of clear cell renal cell carcinoma. Clin Cancer Res 13: 4740–4749.
Hino S, Kishida S, Michiue T, Fukui A, Sakamoto I, Takada S et al. (2001). Inhibition of the Wnt signaling pathway by Idax, a novel Dvl-binding protein. Mol Cell Biol 21: 330–342.
Jiang F, Desper R, Papadimitriou CH, Schaffer AA, Kallioniemi OP, Richter J et al. (2000). Construction of evolutionary tree models for renal cell carcinoma from comparative genomic hybridization data. Cancer Res 60: 6503–6509.
Jiang F, Moch H, Richter J, Egenter C, Gasser T, Bubendorf L et al. (1998). Comparative genomic hybridization reveals frequent chromosome 13q and 4q losses in renal carcinomas with sarcomatoid transformation. J Pathol 185: 382–388.
Kasof GM, Gomes BC . (2001). Livin, a novel inhibitor of apoptosis protein family member. J Biol Chem 276: 3238–3246.
Kempkensteffen C, Hinz S, Schrader M, Christoph F, Magheli A, Krause H et al. (2007). Gene expression and promoter methylation of the XIAP-associated factor 1 in renal cell carcinomas: correlations with pathology and outcome. Cancer Lett 254: 227–235.
Krig SR, Jin VX, Bieda MC, O'Geen H, Yaswen P, Green R et al. (2007). Identification of genes directly regulated by the oncogene ZNF217 using chromatin immunoprecipitation (ChIP)-chip assays. J Biol Chem 282: 9703–9712.
Kurose K, Sakaguchi M, Nasu Y, Ebara S, Kaku H, Kariyama R et al. (2004). Decreased expression of REIC/Dkk-3 in human renal clear cell carcinoma. J Urol 171: 1314–1318.
Lee MG, Huh JS, Chung SK, Lee JH, Byun DS, Ryu BK et al. (2006). Promoter CpG hypermethylation and downregulation of XAF1 expression in human urogenital malignancies: implication for attenuated p53 response to apoptotic stresses. Oncogene 25: 5807–5822.
Levy DA, Slaton JW, Swanson DA, Dinney CP . (1998). Stage specific guidelines for surveillance after radical nephrectomy for local renal cell carcinoma. J Urol 159: 1163–1167.
Liston P, Fong WG, Kelly NL, Toji S, Miyazaki T, Conte D et al. (2001). Identification of XAF1 as an antagonist of XIAP anti-caspase activity. Nat Cell Biol 3: 128–133.
Mann B, Gelos M, Siedow A, Hanski ML, Gratchev A, Ilyas M et al. (1999). Target genes of beta-catenin-T cell-factor/lymphoid-enhancer-factor signaling in human colorectal carcinomas. Proc Natl Acad Sci USA 96: 1603–1608.
Matsuzaki H, Dong S, Loi H, Di X, Liu G, Hubbell E et al. (2004). Genotyping over 100,000 SNPs on a pair of oligonucleotide arrays. Nat Methods 1: 109–111.
Michiue T, Fukui A, Yukita A, Sakurai K, Danno H, Kikuchi A et al. (2004). XIdax, an inhibitor of the canonical Wnt pathway, is required for anterior neural structure formation in Xenopus. Dev Dyn 230: 79–90.
Mitra AB, Murty VV, Li RG, Pratap M, Luthra UK, Chaganti RS . (1994). Allelotype analysis of cervical carcinoma. Cancer Res 54: 4481–4487.
Moch H, Presti Jr JC, Sauter G, Buchholz N, Jordan P, Mihatsch MJ et al. (1996). Genetic aberrations detected by comparative genomic hybridization are associated with clinical outcome in renal cell carcinoma. Cancer Res 56: 27–30.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O et al. (2007). Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356: 115–124.
Nannya Y, Sanada M, Nakazaki K, Hosoya N, Wang L, Hangaishi A et al. (2005). A robust algorithm for copy number detection using high-density oligonucleotide single nucleotide polymorphism genotyping arrays. Cancer Res 65: 6071–6079.
Ohba K, Miyata Y, Kanda S, Koga S, Hayashi T, Kanetake H . (2005). Expression of urokinase-type plasminogen activator, urokinase-type plasminogen activator receptor and plasminogen activator inhibitors in patients with renal cell carcinoma: correlation with tumor associated macrophage and prognosis. J Urol 174: 461–465.
Qiao Q, Ramadani M, Gansauge S, Gansauge F, Leder G, Beger HG . (2001). Reduced membranous and ectopic cytoplasmic expression of beta -catenin correlate with cyclin D1 overexpression and poor prognosis in pancreatic cancer. Int J Cancer 95: 194–197.
Ried T, Knutzen R, Steinbeck R, Blegen H, Schrock E, Heselmeyer K et al. (1996). Comparative genomic hybridization reveals a specific pattern of chromosomal gains and losses during the genesis of colorectal tumors. Genes Chromosomes Cancer 15: 234–245.
Shimokawa T, Furukawa Y, Sakai M, Li M, Miwa N, Lin YM et al. (2003). Involvement of the FGF18 gene in colorectal carcinogenesis, as a novel downstream target of the beta-catenin/T-cell factor complex. Cancer Res 63: 6116–6120.
Storkel S, Eble JN, Adlakha K, Amin M, Blute ML, Bostwick DG et al. (1997). Classification of renal cell carcinoma: Workgroup No. 1. Union Internationale Contre le Cancer (UICC) and the American Joint Committee on Cancer (AJCC). Cancer 80: 987–989.
Suzuki H, Ueda T, Komiya A, Okano T, Isaka S, Shimazaki J et al. (1997). Mutational state of von Hippel-Lindau and adenomatous polyposis coli genes in renal tumors. Oncology 54: 252–257.
Swiercz R, Wolfe JD, Zaher A, Jankun J . (1998). Expression of the plasminogen activation system in kidney cancer correlates with its aggressive phenotype. Clin Cancer Res 4: 869–877.
Ueta T, Ikeguchi M, Hirooka Y, Kaibara N, Terada T . (2002). Beta-catenin and cyclin D1 expression in human hepatocellular carcinoma. Oncol Rep 9: 1197–1203.
Wagener N, Crnkovic-Mertens I, Vetter C, Macher-Goppinger S, Bedke J, Grone EF et al. (2007). Expression of inhibitor of apoptosis protein Livin in renal cell carcinoma and non-tumorous adult kidney. Br J Cancer 97: 1271–1276.
Wang TL, Maierhofer C, Speicher MR, Lengauer C, Vogelstein B, Kinzler KW et al. (2002). Digital karyotyping. Proc Natl Acad Sci USA 99: 16156–16161.
Yeh SH, Chen PJ, Lai MY, Chen DS . (1996). Allelic loss on chromosomes 4q and 16q in hepatocellular carcinoma: association with elevated alpha-fetoprotein production. Gastroenterology 110: 184–192.
Yoshimoto T, Matsuura K, Karnan S, Tagawa H, Nakada C, Tanigawa M et al. (2007). High-resolution analysis of DNA copy number alterations and gene expression in renal clear cell carcinoma. J Pathol 213: 392–401.
Yuan D, Liu L, Gu D . (2007). Transcriptional regulation of livin by beta-catenin/TCF signaling in human lung cancer cell lines. Mol Cell Biochem 306: 171–178.
Yuan E, Li CM, Yamashiro DJ, Kandel J, Thaker H, Murty VV et al. (2005). Genomic profiling maps loss of heterozygosity and defines the timing and stage dependence of epigenetic and genetic events in Wilms' tumors. Mol Cancer Res 3: 493–502.
Acknowledgements
We thank Taeko Asano and Miyuki Katto for immunohistochemistry and microarray, respectively. We also thank Noriko Kunita for technical support. This study was performed in part as a collaborative research effort with the Clinical Informatics Research Initiative at the Organ Development Research Laboratory of the National Institute of Advanced Industrial Science and Technology (AIST), Japan.This study was supported by a grant from Scientific Research from the Japan Society for the Promotion of Science (JSPS), Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Kojima, T., Shimazui, T., Hinotsu, S. et al. Decreased expression of CXXC4 promotes a malignant phenotype in renal cell carcinoma by activating Wnt signaling. Oncogene 28, 297–305 (2009). https://doi.org/10.1038/onc.2008.391
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.391
Keywords
This article is cited by
-
Study of the effect of sFRP1 protein on molecules involved in the regulation of DNA methylation in CML cell line
Medical Oncology (2024)
-
CXXC4 mediates glucose-induced β-cell proliferation
Acta Diabetologica (2020)
-
Crimean-Congo hemorrhagic fever virus infection triggers the upregulation of the Wnt signaling pathway inhibitor genes
Virus Genes (2020)
-
Genome wide association study identifies novel potential candidate genes for bovine milk cholesterol content
Scientific Reports (2018)
-
Exosomal microRNA miR-1246 induces cell motility and invasion through the regulation of DENND2D in oral squamous cell carcinoma
Scientific Reports (2016)