Abstract
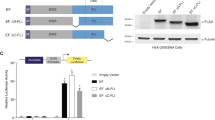
Misregulation of REL, a nuclear factor-κB family transcription factor, has been implicated in several human lymphoid malignancies. REL has a conserved N-terminal DNA-binding/dimerization domain called the Rel homology domain (RHD) and a C-terminal transactivation domain (TAD). Here, we define the sequences (amino acids (aa) 323–422) between the RHD and TAD as a REL inhibitory domain (RID) because deletion of these sequences increases both REL transactivation and DNA binding. Furthermore, we have characterized two REL mRNA splice variants that encode proteins with alterations near RID: one lacking exon 9 sequences (aa 308–330; RELΔ9) and one with an exonized Alu fragment insertion of 32 aa after aa 307 (REL+Alu). Deletion of RID or exon 9-encoded sequences increases transactivation by GAL4–REL by approximately threefold. Moreover, deletion of RID or exon 9 sequences increases transactivation by full-length REL from certain κB site-containing promoters and increases DNA binding by REL. Deletion of RID does not affect REL's ability to transform chicken spleen cells. Reverse transcriptase-polymerase chain reaction analysis of mRNA from both primary lymphoma samples and several transformed tissue culture cell lines indicates that the RELΔ9 splice variant is preferentially expressed in lymphoma, suggesting that the REL transcript lacking exon 9 could serve as a marker for certain types of lymphoid tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Abid MR, Schoots IG, Spokes KC, Wu SQ, Mawhinney C, Aird WC . (2004). Vascular endothelial growth factor-mediated induction of manganese superoxide dismutase occurs through redox-dependent regulation of forkhead and IκB/NF-κB. J Biol Chem 279: 44030–44038.
Bourdon JC, Fernandes K, Murray-Zmijewski F, Liu G, Diot A, Xirodimas DP et al. (2005). p53 isoforms can regulate p53 transcriptional activity. Genes Dev 19: 2122–2137.
Brown PJ, Ashe SL, Leich E, Burek C, Barrans S, Fenton JA et al. (2007). Potentially oncogenic B-cell activation induced smaller isoforms of FOXP1 are highly expressed in the activated B-cell-like subtype of DLBCL. Blood 111: 2816–2824.
Brownell E, Mittereder N, Rice NR . (1989). A human rel proto-oncogene cDNA containing an Alu fragment as a potential coding exon. Oncogene 4: 935–942.
Epinat J-C, Kazandjian D, Harkness DD, Petros S, Dave J, White DW et al. (2000). Mutant envelope residues confer a transactivation function onto N-terminal sequences of the v-Rel oncoprotein. Oncogene 19: 599–607.
Gilmore TD, Cormier C, Jean-Jacques J, Gapuzan M-E . (2001). Malignant transformation of primary chicken spleen cells by human transcription factor c-Rel. Oncogene 20: 7098–7103.
Gilmore TD, Kalaitzidis D, Liang M-C, Starczynowski DT . (2004). The c-Rel transcription factor and B-cell proliferation: a deal with the devil. Oncogene 23: 2275–2286.
Grumont RJ, Gerondakis S . (1994). The subunit composition of NF-κB complexes changes during B-cell development. Cell Growth Differ 5: 1321–1331.
Grumont RJ, Rourke IJ, Gerondakis S . (1999). Rel-dependent induction of A1 transcription is required to protect B cells from antigen receptor ligation-induced apoptosis. Genes Dev 13: 400–411.
Hayden MS, Ghosh S . (2008). Shared principles in NF-κB signaling. Cell 132: 344–362.
Huang DB, Chen YQ, Ruetsche M, Phelps CB, Ghosh G . (2001). X-ray crystal structure of proto-oncogene product c-Rel bound to the CD28 response element of IL-2. Structure 9: 669–678.
Kalaitzidis D, Davis RE, Rosenwald A, Staudt LM, Gilmore TD . (2002). The human B-cell lymphoma cell line RC-K8 has multiple genetic alterations that dysregulate the Rel/NF-κB signal transduction pathway. Oncogene 21: 8759–8768.
Kalaitzidis D, Gilmore TD . (2002). Genomic organization and expression of the rearranged REL proto-oncogene in the human B-cell lymphoma cell line RC-K8. Genes Chromosomes Cancer 34: 129–135.
Köntgen F, Grumont RJ, Strasser A, Metcalf D, Li R, Tarlinton D et al. (1995). Mice lacking the c-rel proto-oncogene exhibit defects in lymphocyte proliferation, humoral immunity, and interleukin-2 expression. Genes Dev 9: 1965–1977.
Leeman JR, Gilmore TD . (2008). Alternative splicing in the NF-κB signaling pathway. Gene (in press).
Liou H-C, Sha WC, Scott ML, Baltimore D . (1994). Sequential induction of NF-κB/Rel family proteins during B-cell terminal differentiation. Mol Cell Biol 14: 5349–5359.
Martin AG, San-Antonio B, Fresno M . (2001). Regulation of NF-κB transactivation. Implication of phosphatidylinositol 3-kinase and protein kinase C-ζ in c-Rel activation by tumor necrosis factor α. J Biol Chem 276: 15840–15849.
Mitchell T, Sugden B . (1995). Stimulation of NF-κB-mediated transcription by mutant derivatives of the latent membrane protein of Epstein–Barr virus. J Virol 69: 2968–2976.
O'Rourke JP, Ness SA . (2008). Alternative RNA splicing produces multiple forms of c-Myb with unique transcriptional activities. Mol Cell Biol 28: 2091–2101.
Sambrook J, Fritsch EF, Maniatis T . (1989). Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Press: Cold Spring Harbor, NY.
Sánchez-Aguilera A, García JF, Sánchez-Beato M, Piris MA . (2006). Hodgkin's lymphoma cells express alternatively spliced forms of HDM2 with multiple effects on cell cycle control. Oncogene 25: 2565–2574.
Schatzle JD, Kralova J, Bose Jr HR . (1995). Avian IκBα is transcriptionally induced by c-Rel and v-Rel with different kinetics. J Virol 69: 5383–5390.
Stamm S, Ben-Ari S, Rafalska I, Tang Y, Zhang Z, Toiber D et al. (2005). Function of alternative splicing. Gene 344: 1–20.
Starczynowski DT, Reynolds JG, Gilmore TD . (2003). Deletion of either C-terminal transactivation subdomain enhances the in vitro transforming activity of human transcription factor REL in chicken spleen cells. Oncogene 22: 6928–6936.
Starczynowski DT, Reynolds JG, Gilmore TD . (2005). Mutations of tumor necrosis factor α-responsive serine residues within the C-terminal transactivation domain of human transcription factor REL enhance its in vitro transforming ability. Oncogene 24: 7355–7368.
Starczynowski DT, Trautmann H, Pott C, Harder L, Arnold N, Africa JA et al. (2007). Mutation of an IKK phosphorylation site within the transactivation domain of REL in two patients with B-cell lymphoma enhances REL's in vitro transforming activity. Oncogene 26: 2685–2694.
Stoilov P, Meshorer E, Gencheva M, Glick D, Soreq H, Stamm S . (2002). Defects in pre-mRNA processing as causes of and predisposition to diseases. DNA Cell Biol 21: 803–818.
Yu SH, Chiang WC, Shih HM, Wu KJ . (2004). Stimulation of c-Rel transcriptional activity by PKA catalytic subunit β. J Mol Med 82: 621–628.
Zentrale Ethikkommission bei der Bundesärztekammer . (2003). Die (weiter-)verwendung von menschlichen körpermaterialien für zwecke medizinischer forschung. Dtsch Arztebl 100: A1632–A1635.
Zhong H, May MJ, Jimi E, Ghosh S . (2002). The phosphorylation status of nuclear NF-κB determines its association with CBP/p300 or HDAC-1. Mol Cell 9: 625–636.
Zhong H, Voll RE, Ghosh S . (1998). Phosphorylation of NF-κB p65 by PKA stimulates transcriptional activity by promoting a novel bivalent interaction with the coactivator CBP/p300. Mol Cell 1: 661–671.
Acknowledgements
We thank Mike Garbati, Melanie Herscovitch, Daniel Starczynowski and Francis Wolenski for helpful discussions, and Michaela Buck, Karola Dorsch, Iwona Nerbas and Ingo Melzner for technical assistance. This work was supported by NIH Grant CA47763 (TDG) and a grant from Deutsche Krebshilfe (108060; TFB).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Leeman, J., Weniger, M., Barth, T. et al. Deletion analysis and alternative splicing define a transactivation inhibitory domain in human oncoprotein REL. Oncogene 27, 6770–6781 (2008). https://doi.org/10.1038/onc.2008.284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.284
Keywords
This article is cited by
-
The implications of alternative pre-mRNA splicing in cell signal transduction
Experimental & Molecular Medicine (2023)
-
A novel role of MNT as a negative regulator of REL and the NF-κB pathway
Oncogenesis (2021)
-
Regulation of B-cell function by NF-kappaB c-Rel in health and disease
Cellular and Molecular Life Sciences (2020)
-
c-Rel and its many roles in cancer: an old story with new twists
British Journal of Cancer (2016)
-
Identification of an NF-κB p50/p65-responsive site in the human MIR155HG promoter
BMC Molecular Biology (2013)