Abstract
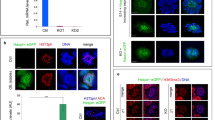
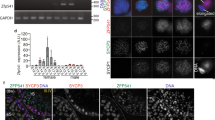
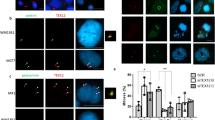
Testis-specific protein Y-encoded (TSPY) is the putative gene for the gonadoblastoma locus on the Y chromosome (GBY). TSPY and an X-homologue, TSPX, harbor a conserved domain, designated as SET/NAP domain, but differ at their C termini. Ectopic expression of TSPY accelerates cell proliferation by abbreviating the G2/M stage, whereas overexpression of TSPX retards cells at the same stage of the cell cycle. Previous studies demonstrated that the SET oncoprotein is capable of binding to cyclin B. Using various protein interaction techniques, we demonstrated that TSPY and TSPX indeed bind competitively to cyclin B at their SET/NAP domains in vitro and in vivo. TSPY colocalizes with cyclin B1 during the cell cycle, particularly on the mitotic spindles at metaphase. TSPY enhances while TSPX represses the cyclin B1–CDK1 phosphorylation activity. The inhibitory effect of TSPX on the cyclin B1–CDK1 complex has been mapped to its carboxyl acidic domain that is absent in TSPY, suggesting that TSPX could serve a normal function in modulating cell-cycle progression at the G2/M stage, whereas TSPY has acquired a specialized function in germ cell renewal and differentiation. Epigenetic dysregulation of TSPY in incompatible germ or somatic cells could promote cell proliferation and predispose susceptible cells to tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Adachi Y, Pavlakis GN, Copeland TD . (1994). Identification and characterization of SET, a nuclear phosphoprotein encoded by the translocation break point in acute undifferentiated leukemia. J Biol Chem 269: 2258–2262.
Bowles J, Koopman P . (2007). Retinoic acid, meiosis and germ cell fate in mammals. Development 134: 3401–3411.
Canela N, Rodriguez-Vilarrupla A, Estanyol JM, Diaz C, Pujol MJ, Agell N et al. (2003). The SET protein regulates G2/M transition by modulating cyclin B-cyclin-dependent kinase 1 activity. J Biol Chem 278: 1158–1164.
Chai Z, Sarcevic B, Mawson A, Toh BH . (2001). SET-related cell division autoantigen-1 (CDA1) arrests cell growth. J Biol Chem 276: 33665–33674.
Compagnone NA, Zhang P, Vigne JL, Mellon SH . (2000). Novel role for the nuclear phosphoprotein SET in transcriptional activation of P450c17 and initiation of neurosteroidogenesis. Mol Endocrinol 14: 875–888.
Decordier I, Cundari E, Kirsch-Volders M . (2008). Mitotic checkpoints and the maintenance of the chromosome karyotype. Mutat Res 651: 3–13.
Delbridge ML, Longepied G, Depetris D, Mattei MG, Disteche CM, Marshall Graves JA et al. (2004). TSPY, the candidate gonadoblastoma gene on the human Y chromosome, has a widely expressed homologue on the X—implications for Y chromosome evolution. Chromosome Res 12: 345–356.
Estanyol JM, Jaumot M, Casanovas O, Rodriguez-Vilarrupla A, Agell N, Bachs O . (1999). The protein SET regulates the inhibitory effect of p21(Cip1) on cyclin E-cyclin-dependent kinase 2 activity. J Biol Chem 274: 33161–33165.
Fukasawa K . (2007). Oncogenes and tumour suppressors take on centrosomes. Nat Rev Cancer 7: 911–924.
Gallagher WM, Bergin OE, Rafferty M, Kelly ZD, Nolan IM, Fox EJ et al. (2005). Multiple markers for melanoma progression regulated by DNA methylation: insights from transcriptomic studies. Carcinogenesis 26: 1856–1867.
Gamble MJ, Erdjument-Bromage H, Tempst P, Freedman LP, Fisher RP . (2005). The histone chaperone TAF-I/SET/INHAT is required for transcription in vitro of chromatin templates. Mol Cell Biol 25: 797–807.
Graves JA . (1995). The evolution of mammalian sex chromosomes and the origin of sex determining genes. Philos Trans R Soc Lond B Biol Sci 350: 305–311; discussion 311–312.
Hoei-Hansen C, Sehested A, Juhler M, Lau Y-FC, Skakkebaek NE, Laursen H et al. (2006). New evidence for the origin of intracranial germ cell tumours from primordial germ cells: expression of pluripotency and cell differentiation markers. J Path 209: 25–33.
Honecker F, Stoop H, De Krijger RR, Chris Lau YF, Bokemeyer C Looijenga LH . (2004). Pathobiological implications of the expression of markers of testicular carcinoma in situ by fetal germ cells. J Pathol 203: 849–857.
Kandilci A, Grosveld GC . (2005). SET-induced calcium signaling and MAPK/ERK pathway activation mediate dendritic cell-like differentiation of U937 cells. Leukemia 19: 1439–1445.
Kellogg DR, Kikuchi A, Fujii-Nakata T, Turck CW, Murray AW . (1995). Members of the NAP/SET family of proteins interact specifically with B-type cyclins. J Cell Biol 130: 661–673.
Kersemaekers AM, Honecker F, Stoop H, Cools M, Molier M, Wolffenbuttel K et al. (2005). Identification of germ cells at risk for neoplastic transformation in gonadoblastoma: an immunohistochemical study for OCT3/4 and TSPY. Hum Pathol 36: 512–521.
Kido T, Lau YF . (2005). A Cre gene directed by a human TSPY promoter is specific for germ cells and neurons. Genesis 42: 263–275.
Kido T, Lau YF . (2006). The rat Tspy is preferentially expressed in elongated spermatids and interacts with the core histones. Biochem Biophys Res Commun 350: 56–67.
Krick R, Jakubiczka S, Arnemann J . (2003). Expression, alternative splicing and haplotype analysis of transcribed testis specific protein (TSPY) genes. Gene 302: 11–19.
Lau Y, Chou P, Iezzoni J, Alonzo J, Komuves L . (2000). Expression of a candidate gene for the gonadoblastoma locus in gonadoblastoma and testicular seminoma. Cytogenet Cell Genet 91: 160–164.
Lau YF, Kido T, Li YM . (2007). The TSPY Gene Family in ‘The Y Chromosome and Male Germ Cell Biology’. In: Lau YF, Chan WY (eds). World Scientific Press: New Jersey, pp 73–90.
Lau YF . (1999). Gonadoblastoma, testicular and prostate cancers, and the TSPY gene. Am J Hum Genet 64: 921–927.
Lau YF, Lau HW, Komuves LG . (2003). Expression pattern of a gonadoblastoma candidate gene suggests a role of the Y chromosome in prostate cancer. Cytogenet Genome Res 101: 250–260.
Lau YF, Zhang J . (2000). Expression analysis of thirty one Y chromosome genes in human prostate cancer. Mol Carcinog 27: 308–321.
Li Y, Oh HJ, Lau YF . (2006). The poly(ADP-ribose) polymerase 1 interacts with Sry and modulates its biological functions. Mol Cell Endocrinol 256: 69–80.
Li Y, Tabatabai ZL, Lee TL, Hatakeyama S, Ohyama C, Chan WY et al. (2007a). The Y-encoded TSPY protein: a significant marker potentially plays a role in the pathogenesis of testicular germ cell tumors. Hum Pathol 38: 1470–1481.
Li Y, Vilain E, Conte F, Rajpert-De Meyts E, Lau YF . (2007b). Testis-specific protein Y-encoded gene is expressed in early and late stages of gonadoblastoma and testicular carcinoma in situ. Urol Oncol 25: 141–146.
Lobrich M, Jeggo PA . (2007). The impact of a negligent G2/M checkpoint on genomic instability and cancer induction. Nat Rev Cancer 7: 861–869.
Looijenga LH, Oosterhuis JW . (2002). Pathobiology of testicular germ cell tumors: views and news. Anal Quant Cytol Histol 24: 263–279.
Masai H, Matsui E, You Z, Ishimi Y, Tamai K, Arai K . (2000). Human Cdc7-related kinase complex. In vitro phosphorylation of MCM by concerted actions of Cdks and Cdc7 and that of a criticial threonine residue of Cdc7 bY Cdks. J Biol Chem 275: 29042–29052.
Oh HJ, Li Y, Lau YF . (2005). Sry associates with the heterochromatin protein 1 complex by interacting with a KRAB domain protein. Biol Reprod 72: 407–415.
Oosterhuis JW, Looijenga LH . (2005). Testicular germ-cell tumours in a broader perspective. Nat Rev Cancer 5: 210–222.
Oram SW, Liu XX, Lee TL, Chan WY, Lau YF . (2006). TSPY potentiates cell proliferation and tumorigenesis by promoting cell cycle progression in HeLa and NIH3T3 cells. BMC Cancer 6: 154.
Ozbun LL, Martinez A, Angdisen J, Umphress S, Kang Y, Wang M et al. (2003). Differentially expressed nucleolar TGF-beta1 target (DENTT) in mouse development. Dev Dyn 226: 491–511.
Ozbun LL, Martinez A, Jakowlew SB . (2005). Differentially expressed nucleolar TGF-beta1 target (DENTT) shows tissue-specific nuclear and cytoplasmic localization and increases TGF-beta1-responsive transcription in primates. Biochim Biophys Acta 1728: 163–180.
Ozbun LL, You L, Kiang S, Angdisen J, Martinez A, Jakowlew SB . (2001). Identification of differentially expressed nucleolar TGF-beta1 target (DENTT) in human lung cancer cells that is a new member of the TSPY/SET/NAP-1 superfamily. Genomics 73: 179–193.
Page DC . (1987). Hypothesis: a Y-chromosomal gene causes gonadoblastoma in dysgenetic gonads. Development 101 (Suppl): 151–155.
Pines J, Hunter T . (1992). Cyclins A and B1 in the human cell cycle. Ciba Found Symp 170: 187–196; discussion 196–204.
Porter LA, Donoghue DJ . (2003). Cyclin B1 and CDK1: nuclear localization and upstream regulators. Prog Cell Cycle Res 5: 335–347.
Puffenberger EG, Hu-Lince D, Parod JM, Craig DW, Dobrin SE, Conway AR et al. (2004). Mapping of sudden infant death with dysgenesis of the testes syndrome (SIDDT) by a SNP genome scan and identification of TSPYL loss of function. Proc Natl Acad Sci USA 101: 11689–11694.
Rajesh C, Pittman DL . (2006). Cell cycle regulation in mammalian germ cells. Results Probl Cell Differ 42: 343–367.
Rajpert-De Meyts E . (2006). Developmental model for the pathogenesis of testicular carcinoma in situ: genetic and environmental aspects. Hum Reprod Update 12: 303–323.
Repping S, van Daalen SK, Brown LG, Korver CM, Lange J, Marszalek JD et al. (2006). High mutation rates have driven extensive structural polymorphism among human Y chromosomes. Nat Genet 38: 463–467.
Richter JD . (2001). Think globally, translate locally: what mitotic spindles and neuronal synapses have in common. Proc Natl Acad Sci USA 98: 7069–7071.
Salo P, Kaariainen H, Petrovic V, Peltomaki P, Page DC, De la Chapelle A . (1995). Molecular mapping of the putative gonadoblastoma locus on the Y chromosome. Genes Chromosomes Cancer 14: 210–214.
Schmit TL, Ahmad N . (2007). Regulation of mitosis via mitotic kinases: new opportunities for cancer management. Mol Cancer Ther 6: 1920–1931.
Schnieders F, Dork T, Arnemann J, Vogel T, Werner M, Schmidtke J . (1996). Testis-specific protein, Y-encoded (TSPY) expression in testicular tissues. Hum Mol Genet 5: 1801–1807.
Seo SB, McNamara P, Heo S, Turner A, Lane WS, Chakravarti D . (2001). Regulation of histone acetylation and transcription by INHAT, a human cellular complex containing the set oncoprotein. Cell 104: 119–130.
Skaletsky H, Kuroda-Kawaguchi T, Minx PJ, Cordum HS, Hillier L, Brown LG et al. (2003). The male-specific region of the human Y chromosome is a mosaic of discrete sequence classes. Nature 423: 825–837.
Smits VA, Medema RH . (2001). Checking out the G(2)/M transition. Biochim Biophys Acta 1519: 1–12.
Tschop K, Muller GA, Grosche J, Engeland K . (2006). Human cyclin B3. mRNA expression during the cell cycle and identification of three novel nonclassical nuclear localization signals. Febs J 273: 1681–1695.
Tsuchiya K, Reijo R, Page DC, Disteche CM . (1995). Gonadoblastoma: molecular definition of the susceptibility region on the Y chromosome. Am J Hum Genet 57: 1400–1407.
Vera J, Jaumot M, Estanyol JM, Brun S, Agell N, Bachs O . (2005). Heterogeneous nuclear ribonucleoprotein A2 is a SET-binding protein and a PP2A inhibitor. Oncogene 25: 260–270.
Vijayakumar S, Hall DC, Reveles XT, Troyer DA, Thompson IM, Garcia D et al. (2006). Detection of recurrent copy number loss at Yp11.2 involving TSPY gene cluster in prostate cancer using array-based comparative genomic hybridization. Cancer Res 66: 4055–4064.
Vogel T, Dittrich O, Mehraein Y, Dechend F, Schnieders F, Schmidtke J . (1998). Murine and human TSPYL genes: novel members of the TSPY-SET-NAP1L1 family. Cytogenet Cell Genet 81: 265–270.
Wang GS, Hong CJ, Yen TY, Huang HY, Ou Y, Huang TN et al. (2004a). Transcriptional modification by a CASK-interacting nucleosome assembly protein. Neuron 42: 113–128.
Wang Q, Hirohashi Y, Furuuchi K, Zhao H, Liu Q, Zhang H et al. (2004b). The centrosome in normal and transformed cells. DNA Cell Biol 23: 475–489.
Whittaker SR, Te Poele RH, Chan F, Linardopoulos S, Walton MI, Garrett MD et al. (2007). The cyclin-dependent kinase inhibitor seliciclib (R-roscovitine; CYC202) decreases the expression of mitotic control genes and prevents entry into mitosis. Cell Cycle 6: 3114–3131.
Yin YH, Li YY, Qiao H, Wang HC, Yang XA, Zhang HG et al. (2005). TSPY is a cancer testis antigen expressed in human hepatocellular carcinoma. Br J Cancer 93: 458–463.
Zhang JS, Yang-Feng TL, Muller U, Mohandas TK, De Jong PJ, Lau YF . (1992). Molecular isolation and characterization of an expressed gene from the human Y chromosome. Hum Mol Genet 1: 717–726.
Acknowledgements
We thank Dr Tatsuo Kido for assistance in immunofluorescence of transfected cells presented in Figures 4f–h. This work was partially supported by a Merit Review grant and a Prostate Cancer REAP from the Department of Veterans Affairs and an Idea Grant from the Department of Defense to Y-FCL. Y-FC Lau is a research career scientist of the Department of Veterans Affairs.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Li, Y., Lau, YF. TSPY and its X-encoded homologue interact with cyclin B but exert contrasting functions on cyclin-dependent kinase 1 activities. Oncogene 27, 6141–6150 (2008). https://doi.org/10.1038/onc.2008.206
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.206
Keywords
This article is cited by
-
Targeting CDK1 in cancer: mechanisms and implications
npj Precision Oncology (2023)
-
Sex specific regulation of TSPY-Like 2 in the DNA damage response of cancer cells
Cell Death & Disease (2023)
-
Y chromosome is moving out of sex determination shadow
Cell & Bioscience (2022)
-
Y chromosome in health and diseases
Cell & Bioscience (2020)
-
The Y-linked proto-oncogene TSPY contributes to poor prognosis of the male hepatocellular carcinoma patients by promoting the pro-oncogenic and suppressing the anti-oncogenic gene expression
Cell & Bioscience (2019)