Abstract
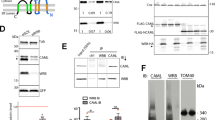
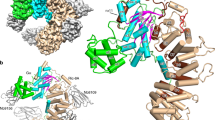
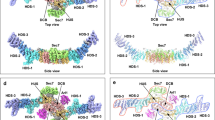
Lowe syndrome and type 2 Dent disease are caused by defects in the inositol 5-phosphatase OCRL. Most missense mutations in the OCRL ASH-RhoGAP domain that are found in affected individuals abolish interactions with the endocytic adaptors APPL1 and Ses (both Ses1 and Ses2), which bind OCRL through a short phenylalanine and histidine (F&H) motif. Using X-ray crystallography, we have identified the F&H motif binding site on the RhoGAP domain of OCRL. Missense mutations associated with disease affected F&H binding indirectly by destabilizing the RhoGAP fold. By contrast, a disease-associated mutation that does not perturb F&H binding and ASH-RhoGAP stability disrupted the interaction of OCRL with Rab5. The F&H binding site of OCRL is conserved even in species that do not have an identified homolog for APPL or Ses. Our study predicts the existence of other OCRL binding partners and shows that the perturbation of OCRL interactions has a crucial role in disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Attree, O. et al. The Lowe's oculocerebrorenal syndrome gene encodes a protein highly homologous to inositol polyphosphate-5-phosphatase. Nature 358, 239–242 (1992).
Hoopes, R.R.J. et al. Dent disease with mutations in OCRL1. Am. J. Hum. Genet. 76, 260–267 (2005).
Bockenhauer, D. et al. Renal phenotype in Lowe syndrome: a selective proximal tubular dysfunction. Clin. J. Am. Soc. Nephrol. 3, 1430–1436 (2008).
Delleman, J.W., Bleeker-Wagemakers, E.M. & van Veelen, A.W. Opacities of the lens indicating carrier status in the oculo-cerebro-renal (Lowe) syndrome. J. Pediatr. Ophthalmol. 14, 205–212 (1977).
Kenworthy, L. & Charnas, L. Evidence for a discrete behavioral phenotype in the oculocerebrorenal syndrome of Lowe. Am. J. Med. Genet. 59, 283–290 (1995).
Schurman, S.J. & Scheinman, S.J. Inherited cerebrorenal syndromes. Nat. Rev. Nephrol. 5, 529–538 (2009).
Bökenkamp, A. et al. Dent-2 disease: a mild variant of Lowe syndrome. J. Pediatr. 155, 94–99 (2009).
Shrimpton, A.E. et al. OCRL1 mutations in Dent 2 patients suggest a mechanism for phenotypic variability. Nephron Physiol. 112, 27–36 (2009).
Bothwell, S.P., Farber, L.W., Hoagland, A. & Nussbaum, R.L. Species-specific difference in expression and splice-site choice in Inpp5b, an inositol polyphosphate 5-phosphatase paralogous to the enzyme deficient in Lowe syndrome. Mamm. Genome 21, 458–466 (2010).
Jänne, P.A. et al. Functional overlap between murine Inpp5b and Ocrl1 may explain why deficiency of the murine ortholog for OCRL1 does not cause Lowe syndrome in mice. J. Clin. Invest. 101, 2042–2053 (1998).
Dressman, M.A., Olivos-Glander, I.M., Nussbaum, R.L. & Suchy, S.F. Ocrl1, a PtdIns(4,5)P(2) 5-phosphatase, is localized to the trans-Golgi network of fibroblasts and epithelial cells. J. Histochem. Cytochem. 48, 179–190 (2000).
Olivos-Glander, I.M., Janne, P.A. & Nussbaum, R.L. The oculocerebrorenal syndrome gene product is a 105-kD protein localized to the Golgi complex. Am. J. Hum. Genet. 57, 817–823 (1995).
Suchy, S.F., Olivos-Glander, I.M. & Nussabaum, R.L. Lowe syndrome, a deficiency of phosphatidylinositol 4,5-bisphosphate 5-phosphatase in the Golgi apparatus. Hum. Mol. Genet. 4, 2245–2250 (1995).
Erdmann, K.S. et al. A role of the Lowe syndrome protein OCRL in early steps of the endocytic pathway. Dev. Cell 13, 377–390 (2007).
Mao, Y. et al. A PH domain within OCRL bridges clathrin-mediated membrane trafficking to phosphoinositide metabolism. EMBO J. 28, 1831–1842 (2009).
Ungewickell, A., Ward, M.E., Ungewickell, E. & Majerus, P.W. The inositol polyphosphate 5-phosphatase Ocrl associates with endosomes that are partially coated with clathrin. Proc. Natl. Acad. Sci. USA 101, 13501–13506 (2004).
Faucherre, A. et al. Lowe syndrome protein OCRL1 interacts with Rac GTPase in the trans-Golgi network. Hum. Mol. Genet. 12, 2449–2456 (2003).
Fukuda, M., Kanno, E., Ishibashi, K. & Itoh, T. Large scale screening for novel rab effectors reveals unexpected broad Rab binding specificity. Mol. Cell. Proteomics 7, 1031–1042 (2008).
Hyvola, N. et al. Membrane targeting and activation of the Lowe syndrome protein OCRL1 by rab GTPases. EMBO J. 25, 3750–3761 (2006).
Swan, L.E., Tomasini, L., Pirruccello, M., Lunardi, J. & de Camilli, P. Two closely related endocytic proteins that share a common OCRL-binding motif with APPL1. Proc. Natl. Acad. Sci. USA 107, 3511–3516 (2010).
Noakes, C.J., Lee, G. & Lowe, M. The PH domain proteins IPIP27A and B link OCRL1 to receptor recycling in the endocytic pathway. Mol. Cell. Biol. 22, 606–623 (2011).
Zoncu, R. et al. A phosphoinositide switch controls the maturation and signaling properties of APPL endosomes. Cell 136, 1110–1121 (2009).
Miaczynska, M. et al. APPL proteins link Rab5 to nuclear signal transduction via an endosomal compartment. Cell 116, 445–456 (2004).
National Human Genome Research Institute. Database of the OCRL1 Mutations Causing Lowe Syndrome (National Human Genome Research Institute, National Institutes of Health, Bethesda, Maryland, USA, 2010). <http://research.nhgri.nih.gov/lowe/>.
Monnier, N., Satre, V., Lerouge, E., Berthoin, F. & Lunardi, J. OCRL1 mutation analysis in French Lowe syndrome patients: implications for molecular diagnosis strategy and genetic counseling. Hum. Mutat. 16, 157–165 (2000).
Addis, M., Loi, M., Lepiani, C., Cau, M. & Melis, M.A. OCRL mutation analysis in Italian patients with Lowe syndrome. Hum. Mutat. 23, 524–525 (2004).
McCrea, H.J. et al. All known patient mutations in the ASH-RhoGAP domains of OCRL affect targeting and APPL1 binding. Biochem. Biophys. Res. Commun. 369, 493–499 (2008).
Hichri, H. et al. From Lowe syndrome to Dent disease: correlations between mutations of the OCRL1 gene and clinical and biochemical phenotypes. Hum. Mutat. 32, 379–388 (2010).
Yuksel, A., Karaca, E. & Albayram, M.S. Magnetic resonance imaging, magnetic resonance spectroscopy, and facial dysmorphism in a case of Lowe syndrome with novel OCRL1 gene mutation. J. Child Neurol. 24, 93–96 (2009).
Tosetto, E. et al. Locus heterogeneity of Dent's disease: OCRL1 and TMEM27 genes in patients with no CLCN5 mutations. Pediatr. Nephrol. 24, 1967–1973 (2009).
Kawano, T., Indo, Y., Nakazato, H., Shimadzu, M. & Matsuda, I. Oculocerebrorenal syndrome of Lowe: three mutations in the OCRL1 gene derived from three patients with different phenotypes. Am. J. Med. Genet. 77, 348–355 (1998).
Lichter-Konecki, U., Farber, L.W., Cronin, J.S., Suchy, S.F. & Nussbaum, R.L. The effect of missense mutations in the RhoGAP-homology domain on ocrl1 function. Mol. Genet. Metab. 89, 121–128 (2006).
Lin, T. et al. Spectrum of mutations in the OCRL1 gene in the Lowe oculocerebrorenal syndrome. Am. J. Hum. Genet. 60, 1384–1388 (1997).
Holm, L. & Rosenstrom, P. Dali server: conservation mapping in 3D. Nucleic Acids Res. 38 (Suppl.), W545–W549 (2010).
Kaiser, S.E. et al. Structural basis of FFAT motif-mediated ER targeting. Structure 13, 1035–1045 (2005).
Gant-Branum, R.L., Broussard, J.A., Mahsut, A., Webb, D.J. & McLean, J.A. Identification of phosphorylation sites within the signaling adaptor APPL1 by mass spectrometry. J. Proteome Res. 9, 1541–1548 (2010).
Wenk, M.R. et al. Phosphoinositide profiling in complex lipid mixtures using electrospray ionization mass spectrometry. Nat. Biotechnol. 21, 813–817 (2003).
Zhu, G. et al. Structure of the APPL1 BAR-PH domain and characterization of its interaction with Rab5. EMBO J. 26, 3484–3493 (2007).
Ponting, C.P. A novel domain suggests a ciliary function for ASPM, a brain size determining gene. Bioinformatics 22, 1031–1035 (2006).
Nassar, N., Hoffman, G.R., Manor, D., Clardy, J.C. & Cerione, R.A. Structures of Cdc42 bound to the active and catalytically compromised forms of Cdc42GAP. Nat. Struct. Biol. 5, 1047–1052 (1998).
Hou, X. et al. A structural basis for Lowe syndrome caused by mutations in the Rab-binding domain of OCRL1. EMBO J. 30, 1659–1670 (2011).
Deprez, C. et al. Solution structure of the E. coli TolA C-terminal domain reveals conformational changes upon binding to the phage g3p N-terminal domain. J. Mol. Biol. 346, 1047–1057 (2005).
Painter, J. & Merritt, E.A. TLSMDweb server for the generation of multi-group TLS models. J. Appl. Cryst. 39, 109–111 (2006).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Davis, I.W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007).
Christoforidis, S. & Zerial, M. Purification and identification of novel Rab effectors using affinity chromatography. Methods 20, 403–410 (2000).
Acknowledgements
We thank J. Murphy (Yale School of Medicine Macromolecular Crystallography Facility) and K. Reinisch for advice and assistance; the Yale School of Medicine W.M. Keck small-scale peptide synthesis facility for peptide synthesis; M. Stagi for computational support; and D. Balkin and H. Sondermann for discussion. This work has been supported by the following funding sources: Browne-Cox postdoctoral fellowship (M.P.), US National Institutes of Health DK45735 and DK082700 (P.D.C.), DA018343 (E.F.-S. and P.D.C.), and grants from the Lowe Syndrome Association (P.D.C.) and the Lowe Syndrome Trust (P.D.C. and L.S.). The Biacore T100 instrumentation, and the size exclusion chromatography and light scattering instrumentation (SEC/LS system), were supported by US National Institutes of Health grants 1S10RR026992 and 1S10RR023748, respectively.
Author information
Authors and Affiliations
Contributions
M.P., L.E.S. and P.D.C. designed research; M.P., L.E.S. and E.F.-S. conducted experiments and contributed new reagents or analytical tools; M.P., L.E.S. and P.D.C. analyzed data; M.P. and P.D.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 5679 kb)
Rights and permissions
About this article
Cite this article
Pirruccello, M., Swan, L., Folta-Stogniew, E. et al. Recognition of the F&H motif by the Lowe syndrome protein OCRL. Nat Struct Mol Biol 18, 789–795 (2011). https://doi.org/10.1038/nsmb.2071
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2071
This article is cited by
-
Identification and functional characterization of a hemizygous novel intronic variant in OCRL gene causes Lowe syndrome
Clinical and Experimental Nephrology (2020)
-
Ocular Pathology of Oculocerebrorenal Syndrome of Lowe: Novel Mutations and Genotype-Phenotype Analysis
Scientific Reports (2017)
-
OCRL controls trafficking through early endosomes via PtdIns4,5P2-dependent regulation of endosomal actin
The EMBO Journal (2011)