Key Points
-
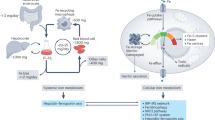
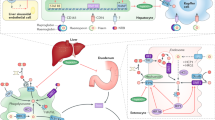
Iron is essential but toxic. Mammals regulate systemic iron through acquisition and storage. Iron is absorbed in the gut and transported into plasma by an apical divalent metal transporter, DMT1, and a basolateral transporter, ferroportin. Only 1–2 mg of iron is absorbed per day in the gut.
-
Most of the iron in the body is found as haem in red blood cells. Old red blood cells are ingested by macrophages and degraded; iron is then recycled back into plasma by ferroportin. Iron in plasma is carried by the protein transferrin, which provides a chelating environment in plasma and a delivery system to cells that express transferrin receptors.
-
Iron in cells can be used for cellular processes or stored in the cytosolic protein ferritin. Levels of iron transporters, carriers and storage proteins are regulated transcriptionally and post-transcriptionally according to iron status.
-
Hepcidin, a peptide hormone secreted by the liver, is the key molecule that regulates systemic iron metabolism by regulating iron entry into plasma. The transcription of hepcidin is tightly regulated by signalling molecules, which sense iron levels, oxygen levels and inflammation. Hepcidin binds to ferroportin, leading to ferroportin degradation and a consequent decrease in cellular iron export.
-
Iron-overload diseases result from inappropriate iron acquisition in response to iron need. Excess iron can damage tissue, cause fibrosis and give rise to organ failure. Iron-deficiency disorders result in anaemia, which in turn give rise to poor oxygenation of tissue. Insight into the regulation of iron metabolism and iron-related diseases has occurred through genetics and the use of model organisms.
Abstract
Mammalian iron homeostasis must be meticulously regulated so that this essential element is available for use, but at the same time prevented from promoting the formation of toxic radicals. Controlling the entry of iron into blood plasma is the main mechanism by which iron stores in the body are physiologically manipulated and regulated. Defects in iron acquisition at the cellular and systemic levels lead to human disorders, which involve either iron overload or iron deficiency. Discoveries of iron transporters and insights into their regulation have provided important information about iron metabolism and genetic iron disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
May, P. M. & Williams, D. R. in Iron in Biochemistry and Medicine (eds Jacobs, A. & Worwood, M.) 1–28 (Academic Press, New York, 1980).
Shayeghi, M. et al. Identification of an intestinal heme transporter. Cell 122, 789–801 (2005).
Qiu, A. et al. Identification of an intestinal folate transporter and the molecular basis for hereditary folate malabsorption. Cell 127, 917–928 (2006).
Gunshin, H. et al. Cybrd1 (duodenal cytochrome b) is not necessary for dietary iron absorption in mice. Blood 106, 2879–2883 (2005).
Gunshin, H. et al. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 388, 482–488 (1997). Identified the apical iron transporter DMT1 by expression cloning.
Fleming, M. D. et al. Microcytic anaemia mice have a mutation in Nramp2, a candidate iron transporter gene. Nature Genet. 16, 383–386 (1997). Identified the apical iron transporter DMT1 by positional cloning.
Fleming, M. D. et al. Nramp2 is mutated in the anemic Belgrade (b) rat: evidence of a role for Nramp2 in endosomal iron transport. Proc. Natl Acad. Sci. USA 95, 1148–1153 (1998).
Gunshin, H. et al. Slc11a2 is required for intestinal iron absorption and erythropoiesis but dispensable in placenta and liver. J. Clin. Invest. 115, 1258–1266 (2005).
Abboud, S. & Haile, D. J. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J. Biol. Chem. 275, 19906–19912 (2000).
Donovan, A. et al. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 403, 776–781 (2000).
McKie, A. T. et al. A novel duodenal iron-regulated transporter, IREG1, implicated in the basolateral transfer of iron to the circulation. Mol. Cell 5, 299–309 (2000). References 9 – 11 reported the discovery of the only known iron exporter.
Donovan, A. et al. The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metab. 1, 191–200 (2005).
Liu, X. B., Yang, F. & Haile, D. J. Functional consequences of ferroportin 1 mutations. Blood Cells Mol. Dis. 35, 33–46 (2005).
De Domenico, I., Ward, D. M., Musci, G. & Kaplan, J. Evidence for the multimeric structure of ferroportin. Blood 109, 2205–2209 (2007).
Osaki, S., Johnson, D. A. & Frieden, E. The possible significance of the ferrous oxidase activity of ceruloplasmin in normal human serum. J. Biol. Chem. 241, 2746–2751 (1966). A seminal study that connected iron and copper metabolism.
Roeser, H. P., Lee, G. R., Nacht, S. & Cartwright, G. E. The role of ceruloplasmin in iron metabolism. J. Clin. Invest. 49, 2408–2417 (1970).
Bush, J. A. et al. Studies on copper metabolism. XIX. The kinetics of iron metabolism and erythrocyte life-span in copper-deficient swine. J. Exp. Med. 103, 701–712 (1956).
Ragan, H. A., Nacht, S., Lee, G. R., Bishop, C. R. & Cartwright, G. E. Effect of ceruloplasmin on plasma iron in copper-deficient swine. Am. J. Physiol. 217, 1320–1323 (1969).
Harris, Z. L., Durley, A. P., Man, T. K. & Gitlin, J. D. Targeted gene disruption reveals an essential role for ceruloplasmin in cellular iron efflux. Proc. Natl Acad. Sci. USA 96, 10812–10817 (1999).
Jeong, S. Y. & David, S. Glycosylphosphatidylinositol-anchored ceruloplasmin is required for iron efflux from cells in the central nervous system. J. Biol. Chem. 278, 27144–27148 (2003).
Harris, Z. L., Klomp, L. W. & Gitlin, J. D. Aceruloplasminemia: an inherited neurodegenerative disease with impairment of iron homeostasis. Am. J. Clin. Nutr. 67, 972S–977S (1998).
De Domenico, I. et al. Ferroxidase activity is required for the stability of cell surface ferroportin in cells expressing GPI-ceruloplasmin. EMBO J. 26, 2823–2831 (2007).
Vulpe, C. D. et al. Hephaestin, a ceruloplasmin homologue implicated in intestinal iron transport, is defective in the sla mouse. Nature Genet. 21, 195–199 (1999).
Hamill, R. L., Woods, J. C. & Cook, B. A. Congenital atransferrinemia. A case report and review of the literature. Am. J. Clin. Pathol. 96, 215–218 (1991).
Bernstein, S. E. Hereditary hypotransferrinemia with hemosiderosis, a murine disorder resembling human atransferrinemia. J. Lab. Clin. Med. 110, 690–705 (1987).
Aisen, P. Transferrin receptor 1. Int. J. Biochem. Cell Biol. 36, 2137–2143 (2004).
Cheng, Y., Zak, O., Aisen, P., Harrison, S. C. & Walz, T. Structure of the human transferrin receptor–transferrin complex. Cell 116, 565–576 (2004).
Levy, J. E., Jin, O., Fujiwara, Y., Kuo, F. & Andrews, N. C. Transferrin receptor is necessary for development of erythrocytes and the nervous system. Nature Genet. 21, 396–399 (1999).
Sipe, D. M. & Murphy, R. F. Binding to cellular receptors results in increased iron release from transferrin at mildly acidic pH. J. Biol. Chem. 266, 8002–8007 (1991).
Bali, P. K., Zak, O. & Aisen, P. A new role for the transferrin receptor in the release of iron from transferrin. Biochemistry 30, 324–328 (1991).
Ohgami, R. S. et al. Identification of a ferrireductase required for efficient transferrin-dependent iron uptake in erythroid cells. Nature Genet. 37, 1264–1269 (2005).
van Renswoude, J., Bridges, K. R., Harford, J. B. & Klausner, R. D. Receptor-mediated endocytosis of transferrin and the uptake of Fe in K562 cells: identification of a nonlysosomal acidic compartment. Proc. Natl Acad. Sci. USA 79, 6186–6190 (1982).
Dautry-Varsat, A., Ciechanover, A. & Lodish, H. F. pH and the recycling of transferrin during receptor-mediated endocytosis. Proc. Natl Acad. Sci. USA 80, 2258–2262 (1983).
Oudit, G. Y. et al. L-type Ca2+ channels provide a major pathway for iron entry into cardiomyocytes in iron-overload cardiomyopathy. Nature Med. 9, 1187–1194 (2003).
Oudit, G. Y., Trivieri, M. G., Khaper, N., Liu, P. P. & Backx, P. H. Role of L-type Ca2+ channels in iron transport and iron-overload cardiomyopathy. J. Mol. Med. 84, 349–364 (2006).
Liuzzi, J. P., Aydemir, F., Nam, H., Knutson, M. D. & Cousins, R. J. Zip14 (Slc39a14) mediates non-transferrin-bound iron uptake into cells. Proc. Natl Acad. Sci. USA 103, 13612–13617 (2006).
Theil, E. C. Iron, ferritin, and nutrition. Annu. Rev. Nutr. 24, 327–343 (2004).
De Domenico, I. et al. Ferroportin-mediated mobilization of ferritin iron precedes ferritin degradation by the proteasome. EMBO J. 25, 5396–5404 (2006).
Kwak, E. L., Larochelle, D. A., Beaumont, C., Torti, S. V. & Torti, F. M. Role for NF-κB in the regulation of ferritin H by tumor necrosis factor-α. J. Biol. Chem. 270, 15285–15293 (1995).
Ponka, P. Tissue-specific regulation of iron metabolism and heme synthesis: distinct control mechanisms in erythroid cells. Blood 89, 1–25 (1997).
Rouault, T. A. The role of iron regulatory proteins in mammalian iron homeostasis and disease. Nature Chem. Biol. 2, 406–414 (2006).
Galy, B., Ferring, D., Benesova, M., Benes, V. & Hentze, M. W. Targeted mutagenesis of the murine IRP1 and IRP2 genes reveals context-dependent RNA processing differences in vivo. RNA 10, 1019–1025 (2004).
Delaby, C., Pilard, N., Goncalves, A. S., Beaumont, C. & Canonne-Hergaux, F. Presence of the iron exporter ferroportin at the plasma membrane of macrophages is enhanced by iron loading and down-regulated by hepcidin. Blood 106, 3979–3984 (2005).
Mok, H. et al. Disruption of ferroportin 1 regulation causes dynamic alterations in iron homeostasis and erythropoiesis in polycythaemia mice. Development 131, 1859–1868 (2004).
Crosby, W. H. Mucosal block. An evaluation of concepts relating to control of iron absorption. Semin. Hematol. 3, 299–313 (1966).
Frazer, D. M. et al. A rapid decrease in the expression of DMT1 and Dcytb but not Ireg1 or hephaestin explains the mucosal block phenomenon of iron absorption. Gut 52, 340–346 (2003).
Pak, M., Lopez, M. A., Gabayan, V., Ganz, T. & Rivera, S. Suppression of hepcidin during anemia requires erythropoietic activity. Blood 108, 3730–3735 (2006).
Loreal, O. et al. Hepcidin in iron metabolism. Curr. Protein Pept. Sci. 6, 279–291 (2005).
Ganz, T. Hepcidin — a regulator of intestinal iron absorption and iron recycling by macrophages. Best Pract. Res. Clin. Haematol. 18, 171–182 (2005).
Ganz, T. Defensins and host defense. Science 286, 420–421 (1999).
Nemeth, E. et al. The N-terminus of hepcidin is essential for its interaction with ferroportin: structure–function study. Blood 107, 328–333 (2006).
Nicolas, G. et al. Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice. Proc. Natl Acad. Sci. USA 98, 8780–8785 (2001). The first report of hepcidin as an iron metabolism regulatory protein.
Roetto, A. et al. Mutant antimicrobial peptide hepcidin is associated with severe juvenile hemochromatosis. Nature Genet. 33, 21–22 (2003).
Nicolas, G. et al. Severe iron deficiency anemia in transgenic mice expressing liver hepcidin. Proc. Natl Acad. Sci. USA 99, 4596–4601. (2002).
Weinstein, D. A. et al. Inappropriate expression of hepcidin is associated with iron refractory anemia: implications for the anemia of chronic disease. Blood 100, 3776–3781 (2002).
De Domenico, I. et al. The molecular mechanism of hepcidin-mediated ferroportin down-regulation. Mol. Biol. Cell 18, 2569–2578 (2007).
Nemeth, E. et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306, 2090–2093 (2004). Identified the connection between hepcidin and ferroportin in regulating iron export.
De Domenico, I. et al. The molecular basis of ferroportin-linked hemochromatosis. Proc. Natl Acad. Sci. USA 102, 8955–8960 (2005). Described how mutations in ferroportin give rise to dominantly inherited haemochromatosis.
Nicolas, G. et al. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J. Clin. Invest. 110, 1037–1044 (2002).
Babitt, J., Huang, F. W., Xia, Y., Sidis, Y., Andrews, N. C. & Lin, H. Modulation of bone morphogenetic protein signaling in vivo regulates systemic iron balance. J. Clin. Invest. 117, 1933–1939 (2007).
Babitt, J. L. et al. Bone morphogenetic protein signaling by hemojuvelin regulates hepcidin expression. Nature Genet. 38, 531–539 (2006).
Wang, R. H. et al. A role of SMAD4 in iron metabolism through the positive regulation of hepcidin expression. Cell Metab. 2, 399–409 (2005). Described a role for the SMAD system in hepcidin expression.
Nemeth, E. et al. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J. Clin. Invest. 113, 1271–1276 (2004).
Kaelin, W. G. Jr. The von Hippel–Lindau protein, HIF hydroxylation, and oxygen sensing. Biochem. Biophys. Res. Commun. 338, 627–638 (2005).
Peyssonnaux, C. et al. Regulation of iron homeostasis by the hypoxia-inducible transcription factors (HIFs). J. Clin. Invest. 117, 1926–1932 (2007).
Yoon, D. et al. Hypoxia-inducible factor-1 deficiency results in dysregulated erythropoiesis signaling and iron homeostasis in mouse development. J. Biol. Chem. 281, 25703–25711 (2006).
Tanno, T. et al. High levels of GDF15 in thalassemia suppress expression of the iron regulatory protein hepcidin. Nature Med. 13, 1096–1101 (2007). Describes an erythroid signal that regulates hepcidin expression.
Crosby, W. H. Hemochromatosis: the unsolved problems. Semin. Hematol. 14, 135–143 (1977).
Craven, C. M. et al. Tissue distribution and clearance kinetics of non-transferrin-bound iron in the hypotransferrinemic mouse: a rodent model for hemochromatosis. Proc. Natl Acad. Sci. USA 84, 3457–3461 (1987).
Feder, J. N. et al. A novel MHC class I-like gene is mutated in patients with hereditary haemochromatosis. Nature Genet. 13, 399–408 (1996). Identified the mutated gene ( HFE ) that is responsible for the most common form of hereditary haemochromatosis.
Feder, J. N. et al. The hemochromatosis gene product complexes with the transferrin receptor and lowers its affinity for ligand binding. Proc. Natl Acad. Sci. USA 95, 1472–1477 (1998).
Feder, J. N. et al. The hemochromatosis founder mutation in HLA-H disrupts β2-microglobulin interaction and cell surface expression. J. Biol. Chem. 272, 14025–14028 (1997).
Camaschella, C. Juvenile haemochromatosis. Baillieres Clin. Gastroenterol. 12, 227–235 (1998).
Papanikolaou, G. et al. Mutations in HFE2 cause iron overload in chromosome 1q-linked juvenile hemochromatosis. Nature Genet. 36, 77–82 (2004).
Lin, L., Goldberg, Y. P. & Ganz, T. Competitive regulation of hepcidin mRNA by soluble and cell-associated hemojuvelin. Blood 106, 2884–2889 (2005).
Zhang, A. S. et al. Evidence that inhibition of hemojuvelin shedding in response to iron is mediated through neogenin. J. Biol. Chem. 282, 12547–12556 (2007).
Camaschella, C. et al. The gene TFR2 is mutated in a new type of haemochromatosis mapping to 7q22. Nature Genet. 25, 14–15 (2000).
Roetto, A. et al. Hemochromatosis due to mutations in transferrin receptor 2. Blood Cells Mol. Dis. 29, 465–470 (2002).
Kawabata, H. et al. Molecular cloning of transferrin receptor 2. A new member of the transferrin receptor-like family. J. Biol. Chem. 274, 20826–20832 (1999).
Johnson, M. B. & Enns, C. A. Diferric transferrin regulates transferrin receptor 2 protein stability. Blood 104, 4287–4293 (2004).
Montosi, G. et al. Autosomal-dominant hemochromatosis is associated with a mutation in the ferroportin (SLC11A3) gene. J. Clin. Invest. 108, 619–623 (2001).
Pietrangelo, A. The ferroportin disease. Blood Cells Mol. Dis. 32, 131–138 (2004).
Drakesmith, H. et al. Resistance to hepcidin is conferred by hemochromatosis-associated mutations of ferroportin. Blood 106, 1092–1097 (2005).
Schimanski, L. M. et al. In vitro functional analysis of human ferroportin (FPN) and hemochromatosis-associated FPN mutations. Blood 105, 4096–4102 (2005).
McGregor, J. A. et al. Impaired iron transport activity of ferroportin 1 in hereditary iron overload. J. Membr. Biol. 206, 3–7 (2005).
Zohn, I.E. et al. The flatiron mutation in mouse ferroportin acts as a dominant negative to cause ferroportin disease. Blood 109, 4174–4180 (2007).
Goncalves, A. S. et al. Wild-type and mutant ferroportins do not form oligomers in transfected cells. Biochem. J. 396, 265–275 (2006).
De Domenico, I. et al. Zebrafish as a model for defining the functional impact of mammalian ferroportin mutations. Blood 28 Aug 2007 (doi:10.1182/blood-2007-07-100248).
Cartwright, G. E., Lauritsen, M. A., Jones, P. J., Merrill, I. M. & Wintrobe, M. M. The anemia of infection. I. Hypoferremia, hypercupremia, and alterations in porphyrin metabolism in patients. J. Clin. Invest. 25, 65–80 (1946).
Schaible, U. E. & Kaufmann, S. H. Iron and microbial infection. Nature Rev. Microbiol. 2, 946–953 (2004).
Baraldi-Junkins, C. A., Beck, A. C. & Rothstein, G. Hematopoiesis and cytokines. Relevance to cancer and aging. Hematol. Oncol. Clin. North Am. 14, 45–61 (2000).
Bruunsgaard, H., Pedersen, M. & Pedersen, B. K. Aging and proinflammatory cytokines. Curr. Opin. Hematol. 8, 131–136 (2001).
Pietrangelo, A. et al. STAT3 is required for IL-6–gp130-dependent activation of hepcidin in vivo. Gastroenterology 132, 294–300 (2007).
Verga Falzacappa, M. V. et al. STAT3 mediates hepatic hepcidin expression and its inflammatory stimulation. Blood 109, 353–358 (2007).
Wrighting, D. M. & Andrews, N. C. Interleukin-6 induces hepcidin expression through STAT3. Blood 108, 3204–3209 (2006).
Truksa, J., Peng, H., Lee, P. & Beutler, E. Bone morphogenetic proteins 2, 4, and 9 stimulate murine hepcidin 1 expression independently of Hfe, transferrin receptor 2 (Tfr2), and IL-6. Proc. Natl Acad. Sci. USA 103, 10289–10293 (2006).
Bulaj, Z. J. et al. Disease-related conditions in relatives of patients with hemochromatosis. N. Engl. J. Med. 343, 1529–1535 (2000).
Beutler, E. The HFE Cys282Tyr mutation as a necessary but not sufficient cause of clinical hereditary hemochromatosis. Blood 101, 3347–3350 (2003).
Wang, F. et al. Genetic variation in Mon1a affects protein trafficking and modifies macrophage iron loading in mice. Nature Genet. 39, 1025–1032 (2007).
Acknowledgements
The authors wish to express their appreciation to members of the Kaplan laboratory for reading the manuscript. The authors thank J. Miller for sharing unpublished data. This work is supported by a National Institutes of Health grant to J.K.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
OMIM
ZFIN
FURTHER INFORMATION
Glossary
- Haem
-
A prosthetic group (a non-protein component of a conjugated protein) that consists of an iron atom within a large heterocyclic organic ring called a porphyrin. Not all porphyrins contain iron, but a substantial fraction of porphyrin-containing metalloproteins have haem as their prosthetic subunit; these are known as haemoproteins.
- Enterocyte
-
An absorptive cell that lines the intestine.
- Apical membrane
-
The membrane of an epithelial or endothelial cell that faces the lumen of a cavity or tube, or the outside of the organism.
- Basolateral membrane
-
The membrane of an epithelial cell that adjoins underlying tissue.
- Plasma
-
The liquid component of blood that accounts for about half of the total blood volume. Plasma is a solution of nutrients and various proteins.
- Anaemia
-
A pathological condition characterized by abnormally low levels of healthy red blood cells or haemoglobin (the component of red blood cells that delivers oxygen to tissues throughout the body).
- Iron overload
-
A pathological condition in which the body accumulates abnormally high levels of iron. Iron deposits can form in organs and cause life-threatening damage.
- Parenchymal tissue
-
A tissue that is part of the essential structure and/or function of an organ, in contrast to stroma or blood vessels.
- NADP
-
A coenzyme similar to NAD that is present in most living cells. It functions as a reducing agent in different metabolic processes and is thought to supply the electrons for the transmembrane reduction of iron from the ferric state (Fe(III)) to the ferrous state (Fe(II)).
- Endosome
-
A vesicle that is formed by invagination of the plasma membrane.
- Erythropoiesis
-
The formation of erythrocytes (red blood cells).
- Glycophosphatidylinositol
-
A phosphatidylinositol to which a carbohydrate chain is linked through the C-6 hydroxyl of the inositol. It functions as an anchor that localizes proteins to the exoplasmic leaflet of membranes by linking to proteins through an ethanolamine phosphate moiety.
- Sex-linked iron-limited anaemia
-
Anaemia that results from a sex-chromosome-inherited defect of intestinal iron absorption.
- Erythron
-
A term describing all circulating red blood cells, reticulocytes and the tissues where they are produced.
- Lysosome
-
A type of organelle that is characterized by a low internal pH, contains hydrolytic enzymes and is involved in the post-translational maturation of proteins, the degradation of receptors and the extracellular release of active enzymes.
- Clathrin-coated pit
-
The initial site of invagination of a clathrin-coated vesicle, which is a transport vesicle that buds with the aid of a coat protein known as clathrin.
- Reticulocyte
-
The youngest of the red blood cells normally found in the circulation, freshly released from the bone marrow (or other site of erythropoiesis).
- Iron–sulphur cluster
-
Iron ions that are complexed by inorganic sulphur and by the amino acid Cys. Such clusters function as redox elements in electron-transfer reactions.
- Hypoxia
-
A pathological condition in which the body is deprived of adequate oxygen.
- Defensin
-
One of a class of small, 15–25-residue, Cys-rich proteins. They have a broad antimicrobial activity against bacteria, fungi and enveloped viruses.
- Bone morphogenetic protein
-
(BMP). One of a family of secreted proteins that interact with surface receptors. BMPs are cytokines and signalling molecules and are members of the transforming growth factor-β superfamily. They have important effects on development.
- Major histocompatibility complex
-
(MHC). A gene family that is important in the immune system. The encoded proteins are cell-surface proteins that bind to and display fragments of proteins that are endogenous (self) or foreign (non-self) for recognition by the immune system.
- Founder effect
-
A gene mutation that is observed in high frequency in a specific population owing to its presence in a single ancestor or a small number of ancestors.
- Dominant-negative mutant
-
A protein encoded by a mutated gene that adversely affects the function of the wild-type protein within the same cell.
- Haploinsufficiency
-
A state in which the loss of only one of two alleles of a gene detectably disables its function.
Rights and permissions
About this article
Cite this article
De Domenico, I., McVey Ward, D. & Kaplan, J. Regulation of iron acquisition and storage: consequences for iron-linked disorders. Nat Rev Mol Cell Biol 9, 72–81 (2008). https://doi.org/10.1038/nrm2295
Issue Date:
DOI: https://doi.org/10.1038/nrm2295
This article is cited by
-
Ferroptosis regulation through Nrf2 and implications for neurodegenerative diseases
Archives of Toxicology (2024)
-
Genome-wide screening in pluripotent cells identifies Mtf1 as a suppressor of mutant huntingtin toxicity
Nature Communications (2023)
-
Increased IL-6 Levels and the Upregulation of Iron Regulatory Biomarkers Contribute to the Progression of Japanese Encephalitis Virus Infection’s Pathogenesis
NeuroMolecular Medicine (2023)
-
Prevalence and risk factors of human Balantidium coli infection and its association with haematological and biochemical parameters in Ga West Municipality, Ghana
BMC Infectious Diseases (2021)
-
A comparison of transferrin-receptor and epithelial cellular adhesion molecule targeting for microfluidic separation of cancer cells
Biomedical Microdevices (2021)