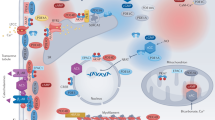
Key Points
-
Sustained exposure to pathological stress stimulates the heart to hypertrophy, increasing disease and mortality risk. New therapies to blunt this process include targeting enzymes below the cell membrane that serve as strategic nodes for hypertrophy regulation.
-
Rho kinase (ROCK) is activated in cardiac hypertrophy and stimulates fibrosis and heart dysfunction. Inhibition by HA-1077 (fasudil) or Y-27632 has antihypertrophic effects and improves function in models of hypertensive–hypertrophy and infarction. Newer agents may provide better specificity.
-
Rapamycin inhibits the growth factor mTOR (mammalian target of rapamycin), which is activated by both physiological and pathological hypertrophy. Inhibition of mTOR by rapamycin reduces hypertrophy and improves function.
-
Reactive oxygen species are another target, and can be treated by antioxidants and such as resveratrol or isorhapontigenin. More potent effects, however, have been obtained by preventing uncoupling of nitric oxide synthase (NOS) by tetrahydrobiopterin (BH4).
-
Cyclic GMP and protein kinase G (PKG) are intrinsic brake systems inhibiting several hypertrophic cascades. They can be stimulated by exogenous nitric oxide donors and natriuretic peptides, although these have clinical limitations. Inhibition of phosphodiesterase 5a (PDE5a) with sildenafil may provide an attractive alternative approach.
-
Calcineurin plays a key role in pathological hypertrophy. Direct inhibition has proved less effective, but new agents that enhance calcineurin inhibitory proteins such as modulatory calcineurin-interactin protein (MCIP1; also known as DSCR1), calsarcin, and atrogin are presenting attractive alternatives.
-
Pathological stimuli result in the activation of canonical transient receptor potential channels (TRPCs) that mediate non-voltage gated influx of calcium. This stimulates calcineurin/NFAT (nuclear factor of activated T cells) and hypertrophy. A new series of 3,5-bistrifluoromethyl pyrazole inhibitors appears to target TRPC and may suppress this signal.
-
Ca2+/calmodulin-dependent protein kinase II (CaMKII) adversely modulates myocyte calcium regulation and is pro-growth and pro-arrhythmic, though targeted drug inhibition is still unavailable. Protein kinase C, in particular PKCα, also worsens calcium cycling and contractility. New bisindolylmaleimide inhibitors of PKC (Ro-32-0432, and Ro-31-8220) improve function.
-
Last, nuclear transcription regulators such as histone deacetylases (HDACs) have been targeted, and drugs such as Scriptaid and SK-7041 block HDAC and suppress pressure-overload hypertrophy.
-
These and other new agents under development pose fertile ground for quite potent approaches to block pathologic hypertrophy, perhaps opening a new chapter in the treatment of heart disease.
Abstract
Pathological stress from cardiovascular disease stimulates hypertrophy of heart cells, which increases the risk of cardiac morbidity and mortality. Recent evidence has indicated that inhibiting such hypertrophy could be beneficial, encouraging drug discovery and development efforts for agents that could achieve this goal. Most existing therapies that have antihypertrophic effects target outside–in signalling in cardiac cells, but their effectiveness seems limited, and so attention has recently turned to the potential of targeting intracellular signalling pathways. Here, we focus on new developments with small-molecule inhibitors of cardiac hypertrophy, summarizing both agents that have been in or are poised for clinical testing, and pathways that offer further promising potential therapeutic targets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Frey, N., Katus, H. A., Olson, E. N. & Hill, J. A. Hypertrophy of the heart: a new therapeutic target? Circulation 109, 1580–1589 (2004).
Koren, M. J., Devereux, R. B., Casale, P. N., Savage, D. D. & Laragh, J. H. Relation of left ventricular mass and geometry to morbidity and mortality in uncomplicated essential hypertension. Ann. Intern. Med. 114, 345–352 (1991).
Esposito, G. et al. Genetic alterations that inhibit in vivo pressure-overload hypertrophy prevent cardiac dysfunction despite increased wall stress. Circulation 105, 85–92 (2002).
Nienaber, J. J. et al. Inhibition of receptor-localized PI3K preserves cardiac β-adrenergic receptor function and ameliorates pressure overload heart failure. J. Clin. Invest 112, 1067–1079 (2003).
Devereux, R. B. et al. Prognostic significance of left ventricular mass change during treatment of hypertension. JAMA 292, 2350–2356 (2004).
Gardin, J. M. & Lauer, M. S. Left ventricular hypertrophy: the next treatable, silent killer? JAMA 292, 2396–2398 (2004).
Devereux, R. B. et al. Regression of hypertensive left ventricular hypertrophy by losartan compared with atenolol: the losartan intervention for endpoint reduction in hypertension (LIFE) trial. Circulation 110, 1456–1462 (2004).
Brown, J. H., Del Re, D. P. & Sussman, M. A. The Rac and Rho hall of fame: a decade of hypertrophic signaling hits. Circ. Res. 98, 730–742 (2006).
Nakagawa, O. et al. ROCK-I and ROCK-II, two isoforms of Rho-associated coiled–coil forming protein serine/threonine kinase in mice. FEBS Lett. 392, 189–193 (1996).
Riento, K. & Ridley, A. J. Rocks: multifunctional kinases in cell behaviour. Nature Rev. Mol. Cell Biol. 4, 446–456 (2003).
Hiroki, J. et al. Inflammatory stimuli upregulate Rho-kinase in human coronary vascular smooth muscle cells. J. Mol. Cell. Cardiol. 37, 537–546 (2004).
Thumkeo, D. et al. Targeted disruption of the mouse rho-associated kinase 2 gene results in intrauterine growth retardation and fetal death. Mol. Cell. Biol. 23, 5043–5055 (2003).
Rikitake, Y. et al. Decreased perivascular fibrosis but not cardiac hypertrophy in Rock1+/− haploinsufficient mice. Circulation 112, 2959–2965 (2005).
Shimizu, Y. et al. ROCK-I regulates closure of the eyelids and ventral body wall by inducing assembly of actomyosin bundles. J. Cell Biol. 168, 941–953 (2005).
Kimura, K. et al. Regulation of myosin phosphatase by Rho and Rho-associated kinase (Rho-kinase). Science 273, 245–248 (1996).
Somlyo, A. P. & Somlyo, A. V. Signal transduction and regulation in smooth muscle. Nature 372, 231–236 (1994).
Somlyo, A. P. & Somlyo, A. V. Signal transduction by G-proteins, rho-kinase and protein phosphatase to smooth muscle and non-muscle myosin II. J. Physiol. 522, 177–185 (2000).
Vahebi, S., Kobayashi, T., Warren, C. M., de Tombe, P. P. & Solaro, R. J. Functional effects of rho-kinase-dependent phosphorylation of specific sites on cardiac troponin. Circ. Res. 96, 740–747 (2005).
Li, Z. et al. Regulation of PTEN by Rho small GTPases. Nature Cell Biol. 7, 399–404 (2005).
Oudit, G. Y. et al. The role of phosphoinositide-3 kinase and PTEN in cardiovascular physiology and disease. J. Mol. Cell. Cardiol. 37, 449–471 (2004).
Wolfrum, S. et al. Inhibition of Rho-kinase leads to rapid activation of phosphatidylinositol 3-kinase/protein kinase Akt and cardiovascular protection. Arterioscler. Thromb. Vasc. Biol. 24, 1842–1847 (2004).
Ennis, I. L. et al. Regression of isoproterenol-induced cardiac hypertrophy by Na+/H+ exchanger inhibition. Hypertension 41, 1324–1329 (2003).
Denker, S. P., Yan, W. & Barber, D. L. Effect of Rho GTPases on Na–H exchanger in mammalian cells. Methods Enzymol. 325, 334–348 (2000).
Tominaga, T. & Barber, D. L. Na–H exchange acts downstream of RhoA to regulate integrin-induced cell adhesion and spreading. Mol. Biol. Cell 9, 2287–2303 (1998).
Lai, A. & Frishman, W. H. Rho-kinase inhibition in the therapy of cardiovascular disease. Cardiol. Rev. 13, 285–292 (2005).
Shimokawa, H. & Takeshita, A. Rho-kinase is an important therapeutic target in cardiovascular medicine. Arterioscler. Thromb. Vasc. Biol. 25, 1767–1775 (2005).
Tamura, M. et al. Development of specific Rho-kinase inhibitors and their clinical application. Biochim. Biophys. Acta 1754, 245–252 (2005).
Higashi, M. et al. Long-term inhibition of Rho-kinase suppresses angiotensin II-induced cardiovascular hypertrophy in rats in vivo: effect on endothelial NAD(P)H oxidase system. Circ. Res. 93, 767–775 (2003).
Wang, Y. X. et al. Inhibition of Rho-kinase by fasudil attenuated angiotensin II-induced cardiac hypertrophy in apolipoprotein E deficient mice. Eur. J. Pharmacol. 512, 215–222 (2005).
Hattori, T. et al. Long-term inhibition of Rho-kinase suppresses left ventricular remodeling after myocardial infarction in mice. Circulation 109, 2234–2239 (2004).
Satoh, S. et al. Chronic inhibition of Rho kinase blunts the process of left ventricular hypertrophy leading to cardiac contractile dysfunction in hypertension-induced heart failure. J. Mol. Cell. Cardiol. 35, 59–70 (2003). This demonstrates the effectiveness of a Rho-kinase inhibitor to blunt progressive hypertrophy and heart failure in the Dahl sensitive rat.
Kobayashi, N. et al. Critical role of Rho-kinase pathway for cardiac performance and remodeling in failing rat hearts. Cardiovasc. Res. 55, 757–767 (2002).
Martin G, et al. Statin-induced inhibition of the Rho-signaling pathway activates PPARα and induces HDL apoA-I. J. Clin. Invest. 107, 1423–1432 (2001).
Liao, J. K. & Laufs, U. Pleiotropic effects of statins. Annu. Rev. Pharmacol. Toxicol. 45, 89–118 (2005).
Nakagami, H. & Liao, J. K. Statins and myocardial hypertrophy. Coron. Artery Dis. 15, 247–250 (2004).
Laufs, U. & Liao, J. K. Post-transcriptional regulation of endothelial nitric oxide synthase mRNA stability by Rho GTPase. J. Biol. Chem. 273, 24266–24271 (1998).
Liao, J. K. Statin therapy for cardiac hypertrophy and heart failure. J. Investig. Med. 52, 248–253 (2004).
Takemoto, M. et al. Statins as antioxidant therapy for preventing cardiac myocyte hypertrophy. J. Clin. Invest. 108, 1429–1437 (2001). This shows that statin therapy suppresses hypertrophy coupled to the inhibition of GTP binding to RhoA and RAC1.
Dechend, R. et al. Amelioration of angiotensin II-induced cardiac injury by a 3-hydroxy-3-methylglutaryl coenzyme a reductase inhibitor. Circulation 104, 576–581 (2001).
Hasegawa, H. et al. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors prevent the development of cardiac hypertrophy and heart failure in rats. J. Mol. Cell. Cardiol. 35, 953–960 (2003).
Patel, R. et al. Simvastatin induces regression of cardiac hypertrophy and fibrosis and improves cardiac function in a transgenic rabbit model of human hypertrophic cardiomyopathy. Circulation 104, 317–324 (2001).
Fukuta, H., Sane, D. C., Brucks, S. & Little, W. C. Statin therapy may be associated with lower mortality in patients with diastolic heart failure: a preliminary report. Circulation 112, 357–363 (2005).
Vezina, C., Kudelski, A. & Sehgal, S. N. Rapamycin (AY-22,989), a new antifungal antibiotic. I. Taxonomy of the producing streptomycete and isolation of the active principle. J. Antibiot. (Tokyo) 28, 721–726 (1975).
Brown, E. J. et al. A mammalian protein targeted by G1-arresting rapamycin-receptor complex. Nature 369, 756–758 (1994).
Jacinto, E. & Hall, M. N. Tor signalling in bugs, brain and brawn. Nature Rev. Mol. Cell Biol. 4, 117–126 (2003).
Inoki, K., Corradetti, M. N. & Guan, K. L. Dysregulation of the TSC-mTOR pathway in human disease. Nature Genet. 37, 19–24 (2005).
Proud, C. G. Ras, PI3-kinase and mTOR signaling in cardiac hypertrophy. Cardiovasc. Res. 63, 403–413 (2004).
Manning, B. D. & Cantley, L. C. Rheb fills a GAP between TSC and TOR. Trends Biochem. Sci. 28, 573–576 (2003).
Luo, J. et al. Class IA phosphoinositide 3-kinase regulates heart size and physiological cardiac hypertrophy. Mol. Cell Biol. 25, 9491–9502 (2005).
Shibata, R. et al. Adiponectin-mediated modulation of hypertrophic signals in the heart. Nature Med 10, 1384–1389 (2004).
Chan, A. Y., Soltys, C. L., Young, M. E., Proud, C. G. & Dyck, J. R. Activation of AMP-activated protein kinase inhibits protein synthesis associated with hypertrophy in the cardiac myocyte. J. Biol. Chem. 279, 32771–32779 (2004).
Sadoshima, J. & Izumo, S. Rapamycin selectively inhibits angiotensin II-induced increase in protein synthesis in cardiac myocytes in vitro. Potential role of 70-kD S6 kinase in angiotensin II-induced cardiac hypertrophy. Circ. Res. 77, 1040–1052 (1995).
Boluyt, M. O. et al. Rapamycin inhibits α1-adrenergic receptor-stimulated cardiac myocyte hypertrophy but not activation of hypertrophy-associated genes. Evidence for involvement of p70 S6 kinase. Circ. Res. 81, 176–186 (1997).
Shioi, T. et al. Rapamycin attenuates load-induced cardiac hypertrophy in mice. Circulation 107, 1664–1670 (2003). The first study to demonstrate the benefit of rapamycin in treating pre-existing load-induced cardiac hypertrophy.
Boluyt, M. O. et al. The mTOR/p70S6K signal transduction pathway plays a role in cardiac hypertrophy and influences expression of myosin heavy chain genes in vivo. Cardiovasc. Drugs Ther. 18, 257–267 (2004).
McMullen, J. R. et al. Inhibition of mTOR signaling with rapamycin regresses established cardiac hypertrophy induced by pressure overload. Circulation 109, 3050–3055 (2004).
Crackower, M. A. et al. Regulation of myocardial contractility and cell size by distinct PI3K–PTEN signaling pathways. Cell 110, 737–749 (2002).
Hardt, S. E. & Sadoshima, J. Glycogen synthase kinase-3β: a novel regulator of cardiac hypertrophy and development. Circ. Res. 90, 1055–1063 (2002).
Badorff, C., Seeger, F. H., Zeiher, A. M. & Dimmeler, S. Glycogen synthase kinase 3β inhibits myocardin-dependent transcription and hypertrophy induction through site-specific phosphorylation. Circ. Res. 97, 645–654 (2005).
Dorn, G. W. & Force, T. Protein kinase cascades in the regulation of cardiac hypertrophy. J. Clin. Invest. 115, 527–537 (2005).
Schiekofer, S. et al. Microarray analysis of Akt1 activation in transgenic mouse hearts reveal transcript expression profiles associated with compensatory hypertrophy and failure. Physiol. Genomics 27, 156–170 (2006).
Matsui, T. et al. Phenotypic spectrum caused by transgenic overexpression of activated Akt in the heart. J. Biol. Chem. 277, 22896–22901 (2002).
Shiojima, I. et al. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J. Clin. Invest. 115, 2108–2118 (2005).
Kato, T. et al. Atrial natriuretic peptide promotes cardiomyocyte survival by cGMP-dependent nuclear accumulation of zyxin and Akt. J. Clin. Invest. 115, 2716–2730 (2005).
Tsujita, Y. et al. Nuclear targeting of Akt antagonizes aspects of cardiomyocyte hypertrophy. Proc. Natl Acad. Sci. USA 103, 11946–11951 (2006).
Rota, M. et al. Nuclear targeting of Akt enhances ventricular function and myocyte contractility. Circ. Res. 97, 1332–1341 (2005).
Giordano F. J. Oxygen, oxidative stress, hypoxia, and heart failure. J. Clin. Invest. 115, 500–508 (2005)
Casagrande, S. et al. Glutathionylation of human thioredoxin: a possible crosstalk between the glutathione and thioredoxin systems. Proc. Natl Acad. Sci. USA 99, 9745–9749 (2002).
Yamawaki, H. & Berk, B. C. Thioredoxin: a multifunctional antioxidant enzyme in kidney, heart and vessels. Curr. Opin. Nephrol. Hypertens. 14, 149–153 (2005).
Xie, Z. et al. Intracellular reactive oxygen species mediate the linkage of Na+/K+-ATPase to hypertrophy and its marker genes in cardiac myocytes. J. Biol. Chem. 274, 19323–19328 (1999).
Nakamura, K. et al. Inhibitory effects of antioxidants on neonatal rat cardiac myocyte hypertrophy induced by tumor necrosis factor-α and angiotensin II. Circulation 98, 794–799 (1998).
Amin, J. K. et al. Reactive oxygen species mediate α-adrenergic receptor-stimulated hypertrophy in adult rat ventricular myocytes. J. Mol. Cell. Cardiol. 33, 131–139 (2001).
Date, M. O. et al. The antioxidant N-2-mercaptopropionyl glycine attenuates left ventricular hypertrophy in in vivo murine pressure-overload model. J. Am. Coll. Cardiol. 39, 907–912 (2002).
Siwik, D. A. et al. Inhibition of copper-zinc superoxide dismutase induces cell growth, hypertrophic phenotype, and apoptosis in neonatal rat cardiac myocytes in vitro. Circ. Res. 85, 147–153 (1999).
Ide, T. et al. Direct evidence for increased hydroxyl radicals originating from superoxide in the failing myocardium. Circ. Res. 86, 152–157 (2000).
Kinugawa, S. et al. Treatment with dimethylthiourea prevents left ventricular remodeling and failure after experimental myocardial infarction in mice: role of oxidative stress. Circ. Res. 87, 392–398 (2000).
Liu, H., Colavitti, R., Rovira, I. I. & Finkel, T. Redox-dependent transcriptional regulation. Circ. Res. 97, 967–974 (2005).
Pimentel, D. R. et al. Strain-stimulated hypertrophy in cardiac myocytes is mediated by reactive oxygen species-dependent Ras S-glutathiolation. J. Mol. Cell. Cardiol. 41, 613–622 (2006).
Yamamoto, M. et al. Inhibition of endogenous thioredoxin in the heart increases oxidative stress and cardiac hypertrophy. J. Clin. Invest 112, 1395–1406 (2003).
Matsushima, S. et al. Overexpression of mitochondrial peroxiredoxin-3 prevents left ventricular remodeling and failure after myocardial infarction in mice. Circulation 113, 1779–1786 (2006).
Zhang, X. et al. Cardiac-specific overexpression of catalase rescues ventricular myocytes from ethanol-induced cardiac contractile defect. J. Mol. Cell. Cardiol. 35, 645–652 (2003).
Berry, C. E. & Hare, J. M. Xanthine oxidoreductase and cardiovascular disease: molecular mechanisms and pathophysiological implications. J. Physiol. 555, 589–606 (2004).
Cappola, T. P. et al. Allopurinol improves myocardial efficiency in patients with idiopathic dilated cardiomyopathy. Circulation 104, 2407–2411 (2001).
Engberding, N. et al. Allopurinol attenuates left ventricular remodeling and dysfunction after experimental myocardial infarction: a new action for an old drug? Circulation 110, 2175–2179 (2004).
Saavedra, W. F. et al. Imbalance between xanthine oxidase and nitric oxide synthase signaling pathways underlies mechanoenergetic uncoupling in the failing heart. Circ. Res. 90, 297–304 (2002).
Minhas, K. M. et al. Xanthine oxidoreductase inhibition causes reverse remodeling in rats with dilated cardiomyopathy. Circ. Res. 98, 271–279 (2006).
Amado, L. C. et al. Xanthine oxidase inhibition ameliorates cardiovascular dysfunction in dogs with pacing-induced heart failure. J. Mol. Cell. Cardiol. 39, 531–536 (2005).
Baldus, S. et al. Inhibition of xanthine oxidase improves myocardial contractility in patients with ischemic cardiomyopathy. Free Radic. Biol. Med. 41, 1282–1288 (2006).
Reyes, A. J. & Leary, W. P. Allopurinol or oxypurinol in heart failure therapy — a promising new development or end of story? Cardiovasc. Drugs Ther. 19, 311–313 (2005).
Byrne, J. A., Grieve, D. J., Cave, A. C. & Shah, A. M. Oxidative stress and heart failure. Arch. Mal Coeur Vaiss. 96, 214–221 (2003).
Li, J. M., Gall, N. P., Grieve, D. J., Chen, M. & Shah, A. M. Activation of NADPH oxidase during progression of cardiac hypertrophy to failure. Hypertension 40, 477–484 (2002).
Murdoch, C. E., Grieve, D. J., Cave, A. C., Looi, Y. H. & Shah, A. M. NADPH oxidase and heart failure. Curr. Opin. Pharmacol. 6, 148–153 (2006).
Lambeth, J. D. NOX enzymes and the biology of reactive oxygen. Nature Rev. Immunol. 4, 181–189 (2004).
Nakagami, H., Takemoto, M. & Liao, J. K. NADPH oxidase-derived superoxide anion mediates angiotensin II-induced cardiac hypertrophy. J. Mol. Cell. Cardiol. 35, 851–859 (2003).
Byrne, J. A. et al. Contrasting roles of NADPH oxidase isoforms in pressure-overload versus angiotensin II-induced cardiac hypertrophy. Circ. Res. 93, 802–805 (2003).
Maytin, M. et al. Pressure overload-induced myocardial hypertrophy in mice does not require gp91phox. Circulation 109, 1168–1171 (2004).
Bendall, J. K., Cave, A. C., Heymes, C., Gall, N. & Shah, A. M. Pivotal role of a gp91(phox)-containing NADPH oxidase in angiotensin II-induced cardiac hypertrophy in mice. Circulation 105, 293–296 (2002).
bu-Soud, H. M. & Stuehr, D. J. Nitric oxide synthases reveal a role for calmodulin in controlling electron transfer. Proc. Natl Acad. Sci. USA 90, 10769–10772 (1993).
Schmidt, H. H. et al. No ·NO from NO synthase. Proc. Natl Acad. Sci. USA 93, 14492–14497 (1996).
Wever, R. M., van, D. T., van Rijn, H. J., de, G. F. & Rabelink, T. J. Tetrahydrobiopterin regulates superoxide and nitric oxide generation by recombinant endothelial nitric oxide synthase. Biochem. Biophys. Res. Commun. 237, 340–344 (1997).
Takimoto, E. et al. Oxidant stress from nitric oxide synthase-3 uncoupling stimulates cardiac pathologic remodeling from chronic pressure load. J. Clin. Invest. 115, 1221–1231 (2005). This demonstrated that oxidant stress from NOS uncoupling is important in hypertrophy and dilation of hearts subjected to pressure overload, and that its prevention by exogenous BH 4 also blunts pathological remodelling.
Alp, N. J. et al. Tetrahydrobiopterin-dependent preservation of nitric oxide-mediated endothelial function in diabetes by targeted transgenic GTP-cyclohydrolase I overexpression. J Clin. Invest. 112, 725–735 (2003).
Landmesser, U. et al. Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J. Clin. Invest. 111, 1201–1209 (2003).
Dixon, L. J. et al. Functional consequences of endothelial nitric oxide synthase uncoupling in congestive cardiac failure. Circulation 107, 1725–1728 (2003).
Alp, N. J. & Channon, K. M. Regulation of endothelial nitric oxide synthase by tetrahydrobiopterin in vascular disease. Arterioscler. Thromb. Vasc. Biol. 24, 413–420 (2004).
Madeddu, P. Correction of endothelial dysfunction by tetrahydrobiopterin: new hope for the treatment of arterial hypertension? J. Hypertens. 23, 1335–1336 (2005).
Kase, H., Hashikabe, Y., Uchida, K., Nakanishi, N. & Hattori, Y. Supplementation with tetrahydrobiopterin prevents the cardiovascular effects of angiotensin II-induced oxidative and nitrosative stress. J. Hypertens. 23, 1375–1382 (2005).
Moreno, J. M. et al. Cardiac and renal antioxidant enzymes and effects of tempol in hyperthyroid rats. Am. J. Physiol. Endocrinol. Metab. 289, E776–E783 (2005).
Zhang, G. X. et al. Cardiac oxidative stress in acute and chronic isoproterenol-infused rats. Cardiovasc. Res. 65, 230–238 (2005).
Hoshida, S. et al. Ebselen protects against ischemia-reperfusion injury in a canine model of myocardial infarction. Am. J. Physiol. 267, H2342–H2347 (1994).
Li, H. L. et al. Isorhapontigenin, a new resveratrol analog, attenuates cardiac hypertrophy via blocking signaling transduction pathways. Free Radic. Biol. Med. 38, 243–257 (2005).
Clarke, R. & Armitage, J. Antioxidant vitamins and risk of cardiovascular disease. Review of large-scale randomised trials. Cardiovasc. Drugs Ther. 16, 411–415 (2002).
Hoffmann, F., Feil, R., Kleppisch, T. & Scholssman, J. Function of cGMP-dependent protein kinases as revealed by gene deletion. Physiol. Rev. 86, 1–23 (2006).
Calderone, A., Thaik, C. M., Takahashi, N., Chang, D. F. & Colucci, W. S. Nitric oxide, atrial natriuretic peptide, and cyclic GMP inhibit the growth-promoting effects of norepinephrine in cardiac myocytes and fibroblasts. J. Clin. Invest. 101, 812–818 (1998).
Pilz, R. B. & Casteel, D. E. Regulation of gene expression by cyclic GMP. Circ. Res. 93, 1034–1046 (2003).
Balligand, J. L. Regulation of cardiac β-adrenergic response by nitric oxide. Cardiovasc. Res. 43, 607–620 (1999).
Maurice, D. H. Cyclic nucleotide phosphodiesterase-mediated integration of cGMP and cAMP signaling in cells of the cardiovascular system. Front. Biosci. 10, 1221–1228 (2005).
Mongillo, M. et al. Compartmentalized phosphodiesterase-2 activity blunts β-adrenergic cardiac inotropy via an NO/cGMP-dependent pathway. Circ. Res. 98, 226–234 (2006).
Silberbach, M. et al. Extracellular signal-regulated protein kinase activation is required for the anti-hypertrophic effect of atrial natriuretic factor in neonatal rat ventricular myocytes. J. Biol. Chem. 274, 24858–24864 (1999).
Silberbach, M. & Roberts, C. T. Jr. Natriuretic peptide signalling: molecular and cellular pathways to growth regulation. Cell. Signal. 13, 221–231 (2001).
Koller, K. J. et al. Selective activation of the B natriuretic peptide receptor by C-type natriuretic peptide (CNP). Science 252, 120–123 (1991).
Layland, J., Li, J. M. & Shah, A. M. Role of cyclic GMP-dependent protein kinase in the contractile response to exogenous nitric oxide in rat cardiac myocytes. J. Physiol. 540, 457–467 (2002).
Wollert, K. C. et al. Increased effects of C-type natriuretic peptide on contractility and calcium regulation in murine hearts overexpressing cyclic GMP-dependent protein kinase I. Br. J. Pharmacol. 140, 1227–1236 (2003).
MacLennan, D. H. & Kranias, E. G. Phospholamban: a crucial regulator of cardiac contractility. Nature Rev. Mol. Cell Biol. 4, 566–577 (2003).
Murthy, K. S., Zhou, H., Grider, J. R. & Makhlouf, G. M. Inhibition of sustained smooth muscle contraction by PKA and PKG preferentially mediated by phosphorylation of RhoA. Am. J. Physiol. Gastrointest. Liver Physiol. 284, G1006–G1016 (2003).
Sauzeau, V. et al. Cyclic GMP-dependent protein kinase signaling pathway inhibits RhoA-induced Ca2+ sensitization of contraction in vascular smooth muscle. J. Biol. Chem. 275, 21722–21729 (2000).
Tang, K. M. et al. Regulator of G-protein signaling-2 mediates vascular smooth muscle relaxation and blood pressure. Nature Med 9, 1506–1512 (2003).
Zou, M. X. et al. RGS2 is upregulated by and attenuates the hypertrophic effect of α(1)-adrenergic activation in cultured ventricular myocytes. Cell. Signal. 18, 1655–1663 (2006).
Zhang, W. et al. Selective loss of fine tuning of Gq/11 signaling by RGS2 protein exacerbates cardiomyocyte hypertrophy. J. Biol. Chem. 281, 5811–5820 (2006).
Costa, A. D. et al. Protein kinase G transmits the cardioprotective signal from cytosol to mitochondria. Circ. Res. 97, 329–336 (2005).
Das, A., Xi, L. & Kukreja, R. C. Phosphodiesterase-5 inhibitor sildenafil preconditions adult cardiac myocytes against necrosis and apoptosis. Essential role of nitric oxide signaling. J. Biol. Chem. 280, 12944–12955 (2005).
Nisoli, E. et al. Mitochondrial biogenesis in mammals: the role of endogenous nitric oxide. Science 299, 896–899 (2003).
Holtwick, R. et al. Pressure-independent cardiac hypertrophy in mice with cardiomyocyte-restricted inactivation of the atrial natriuretic peptide receptor guanylyl cyclase-A. J. Clin. Invest. 111, 1399–1407 (2003).
Ogawa, Y. et al. Brain natriuretic peptide appears to act locally as an antifibrotic factor in the heart. Can. J. Physiol. Pharmacol. 79, 723–729 (2001).
Ritchie, R. H., Schiebinger, R. J., LaPointe, M. C. & Marsh, J. D. Angiotensin II-induced hypertrophy of adult rat cardiomyocytes is blocked by nitric oxide. Am. J. Physiol. 275, H1370–H1374 (1998).
Sackner-Bernstein, J. D., Kowalski, M., Fox, M. & Aaronson, K. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA 293, 1900–1905 (2005).
Cataliotti, A. & Burnett, J. C. Jr. Natriuretic peptides: novel therapeutic targets in heart failure. J. Investig. Med. 53, 378–384 (2005).
Evgenov, O. V. et al. NO-independent stimulators and activators of soluble guanylate cyclase: discovery and therapeutic potential. Nature Rev. Drug Discov. 5, 755–768 (2006).
Stasch, J. P. et al. Targeting the heme-oxidized nitric oxide receptor for selective vasodilatation of diseased blood vessels. J. Clin. Invest. 116, 2552–2561 (2006).
Lugnier, C. Cyclic nucleotide phosphodiesterase (PDE) superfamily: a new target for the development of specific therapeutic agents. Pharmacol. Ther. 109, 366–398 (2006).
Bender, A. T. & Beavo, J. A. Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol. Rev. 58, 488–520 (2006).
Castro, L. R., Verde, I., Cooper, D. M. & Fischmeister, R. Cyclic guanosine monophosphate compartmentation in rat cardiac myocytes. Circulation 113, 2221–2228 (2006).
Galie, N. et al. Sildenafil citrate therapy for pulmonary arterial hypertension. N. Engl. J. Med. 353, 2148–2157 (2005).
Goldstein, I. et al. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N. Engl. J. Med. 338, 1397–1404 (1998).
Senzaki, H. et al. Cardiac phosphodiesterase 5 (cGMP-specific) modulates β-adrenergic signaling in vivo and is down-regulated in heart failure. FASEB J. 15, 1718–1726 (2001).
Takimoto, E. et al. cGMP catabolism by phosphodiesterase 5A regulates cardiac adrenergic stimulation by NOS3-dependent mechanism. Circ. Res. 96, 100–109 (2005).
Borlaug, B. A., Melenovsky, V., Marhin, T., Fitzgerald, P. & Kass, D. A. Sildenafil inhibits β-adrenergic-stimulated cardiac contractility in humans. Circulation 112, 2642–2649 (2005).
Takimoto, E. et al. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nature Med. 11, 214–222 (2005). This showed that the chronic inhibition of PDE5a with sildenafil blunted progression of hypertrophy, while enhancing heart function, and reversed pre-existing early stage hypertrophy in pressure-overloaded hearts.
Aramburu, J., Rao, A. & Klee, C. B. Calcineurin: from structure to function. Curr. Top. Cell. Regul. 36, 237–295 (2000).
Crabtree, G. R. & Olson, E. N. NFAT signaling: choreographing the social lives of cells. Cell 109 (Suppl 1), 67–79 (2002).
Molkentin, J. D. et al. A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93, 215–228 (1998).
Bueno, O. F. et al. Impaired cardiac hypertrophic response in calcineurin Aβ-deficient mice. Proc. Natl Acad. Sci. USA 99, 4586–4591 (2002).
De Windt, L. J. et al. Targeted inhibition of calcineurin attenuates cardiac hypertrophy in vivo. Proc. Natl Acad. Sci. USA 98, 3322–3327 (2001).
Rothermel, B. A. et al. Myocyte-enriched calcineurin-interacting protein, MCIP1, inhibits cardiac hypertrophy in vivo. Proc. Natl Acad. Sci. USA 98, 3328–3333 (2001).
van Rooij, E. et al. MCIP1 overexpression suppresses left ventricular remodeling and sustains cardiac function after myocardial infarction. Circ. Res. 94, e18–e26 (2004).
Wilkins, B. J. et al. Targeted disruption of NFATc3, but not NFATc4, reveals an intrinsic defect in calcineurin-mediated cardiac hypertrophic growth. Mol. Cell. Biol. 22, 7603–7613 (2002).
Zou, Y. et al. Calcineurin plays a critical role in the development of pressure overload-induced cardiac hypertrophy. Circulation 104, 97–101 (2001).
Frey, N. & Olson, E. N. Cardiac hypertrophy: the good, the bad, and the ugly. Annu. Rev. Physiol. 65, 45–79 (2003).
Wilkins, B. J. & Molkentin, J. D. Calcium-calcineurin signaling in the regulation of cardiac hypertrophy. Biochem. Biophys. Res. Commun. 322, 1178–1191 (2004).
Chan, B., Greenan, G., McKeon, F. & Ellenberger, T. Identification of a peptide fragment of DSCR1 that competitively inhibits calcineurin activity in vitro and in vivo. Proc. Natl Acad. Sci. USA 102, 13075–13080 (2005).
Bush, E. et al. A small molecular activator of cardiac hypertrophy uncovered in a chemical screen for modifiers of the calcineurin signaling pathway. Proc. Natl Acad. Sci. USA 101, 2870–2875 (2004).
Bush, E. & Hood, D. Use of modulators of a novel form of muscle selective calcineurin interacting protein (MCIP-1-38) as a treatment for cardiovascular diseases, and modulator screening methods. Patent WO200533461 (2005).
Hilioti, Z. et al. GSK-3 kinases enhance calcineurin signaling by phosphorylation of RCNs. Genes Dev. 18, 35–47 (2004).
Antos, C. L. et al. Activated glycogen synthase kinase-3β suppresses cardiac hypertrophy in vivo. Proc. Natl Acad. Sci. USA 99, 907–912 (2002).
Abbasi, S. et al. Protein kinase-mediated regulation of calcineurin through the phosphorylation of modulatory calcineurin-interacting protein 1. J. Biol. Chem. 281, 7717–7726 (2006).
Frey, N., Richardson, J. A. & Olson, E. N. Calsarcins, a novel family of sarcomeric calcineurin-binding proteins. Proc. Natl Acad. Sci. USA 97, 14632–14637 (2000).
Frey, N. & Olson, E. N. Calsarcin-3, a novel skeletal muscle-specific member of the calsarcin family, interacts with multiple Z-disc proteins. J. Biol. Chem. 277, 13998–14004 (2002).
Frey, N. et al. Mice lacking calsarcin-1 are sensitized to calcineurin signaling and show accelerated cardiomyopathy in response to pathological biomechanical stress. Nature Med. 10, 1336–1343 (2004).
Gomes, M. D., Lecker, S. H., Jagoe, R. T., Navon, A. & Goldberg, A. L. Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy. Proc. Natl Acad. Sci. USA 98, 14440–14445 (2001).
Li, H. H. et al. Atrogin-1/muscle atrophy F-box inhibits calcineurin-dependent cardiac hypertrophy by participating in an SCF ubiquitin ligase complex. J. Clin. Invest. 114, 1058–1071 (2004).
Sandri, M. et al. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117, 399–412 (2004).
Ni, Y. G. et al. Foxo transcription factors blunt cardiac hypertrophy by inhibiting calcineurin signaling. Circulation 114, 1159–1168 (2006).
Skurk, C. et al. The FOXO3 a transcription factor regulates cardiac myocyte size downstream of AKT signaling. J. Biol. Chem. 280, 20814–20823 (2005).
Kau, T. R. et al. A chemical genetic screen identifies inhibitors of regulated nuclear export of a Forkhead transcription factor in PTEN-deficient tumor cells. Cancer Cell 4, 463–476 (2003).
Kuriyama, M. et al. A cell-permeable NFAT inhibitor peptide prevents pressure-overload cardiac hypertrophy. Chem. Biol. Drug Des. 67, 238–243 (2006).
Roehrl, M. H. et al. Selective inhibition of calcineurin-NFAT signaling by blocking protein–protein interaction with small organic molecules. Proc. Natl Acad. Sci. USA 101, 7554–7559 (2004).
Kang, S., Li, H., Rao, A. & Hogan, P. G. Inhibition of the calcineurin-NFAT interaction by small organic molecules reflects binding at an allosteric site. J. Biol. Chem. 280, 37698–37706 (2005).
Ramsey, I. S., Delling, M. & Clapham, D. E. An introduction to TRP channels. Annu. Rev. Physiol. 68, 619–647 (2006).
Vazquez, G., Wedel, B. J., Aziz, O., Trebak, M. & Putney, J. W. Jr. The mammalian TRPC cation channels. Biochim. Biophys. Acta 1742, 21–36 (2004).
Bush, E. W. et al. Canonical transient receptor potential channels promote cardiomyocyte hypertrophy through activation of calcineurin signaling. J. Biol. Chem. 281, 33487–33496 (2006).
Seth, M. et al. Sarco(endo)plasmic reticulum Ca2+ ATPase (SERCA) gene silencing and remodeling of the Ca2+ signaling mechanism in cardiac myocytes. Proc. Natl Acad. Sci. USA 101, 16683–16688 (2004).
Onohara, N. et al. TRPC3 and TRPC6 are essential for angiotensin II-induced cardiac hypertrophy. EMBO J. 25, 5305–5316 (2006).
Nakayama, H., Wilkin, B. J., Bodi, I. & Molkentin, J. D. Calcineurin-dependent cardiomyopathy is activated by TRPC in the adult mouse heart. FASEB J. 20, 1660–1670 (2006).
Kuwahara, K. et al. TRPC6 fulfills a calcineurin signaling circuit during pathologic cardiac remodeling. J. Clin. Invest. 116, 3114–3126 (2006).
Maroto, R. et al. TRPC1 forms the stretch-activated cation channel in vertebrate cells. Nature Cell Biol. 7, 179–185 (2005).
Rosenberg, P. et al. TRPC3 channels confer cellular memory of recent neuromuscular activity. Proc. Natl Acad. Sci. USA 101, 9387–9392 (2004).
Lievremont, J. P., Bird, G. S. & Putney, J. W. Jr. Mechanism of inhibition of TRPC cation channels by 2-aminoethoxydiphenylborane. Mol. Pharmacol. 68, 758–762 (2005).
Djuric, S. W. et al. 3, 5-Bis(trifluoromethyl)pyrazoles: a novel class of NFAT transcription factor regulator. J. Med. Chem. 43, 2975–2981 (2000).
Trevillyan, J. M. et al. Potent inhibition of NFAT activation and T cell cytokine production by novel low molecular weight pyrazole compounds. J. Biol. Chem. 276, 48118–48126 (2001).
He, L. P., Hewavitharana, T., Soboloff, J., Spassova, M. A. & Gill, D. L. A functional link between store-operated and TRPC channels revealed by the 3, 5-bis (trifluoromethyl)pyrazole derivative, BTP2. J. Biol. Chem. 280, 10997–11006 (2005).
Zitt, C. et al. Potent inhibition of Ca2+ release-activated Ca2+ channels and T-lymphocyte activation by the pyrazole derivative BTP2. J. Biol. Chem. 279, 12427–12437 (2004).
Venkatesh, N. et al. Chemical genetics to identify NFAT inhibitors: potential of targeting calcium mobilization in immunosuppression. Proc. Natl Acad. Sci. USA 101, 8969–8974 (2004).
Tobimatsu, T. & Fujisawa, H. Tissue-specific expression of four types of rat calmodulin-dependent protein kinase II mRNAs. J. Biol. Chem. 264, 17907–17912 (1989).
Means, A. R. Regulatory cascades involving calmodulin-dependent protein kinases. Mol. Endocrinol. 14, 4–13 (2000).
Schulman, H. The multifunctional Ca2+/calmodulin-dependent protein kinases. Curr. Opin. Cell Biol. 5, 247–253 (1993).
Kato, T. et al. Calmodulin kinases II and IV and calcineurin are involved in leukemia inhibitory factor-induced cardiac hypertrophy in rats. Circ. Res. 87, 937–945 (2000).
Passier, R. et al. CaM kinase signaling induces cardiac hypertrophy and activates the MEF2 transcription factor in vivo. J. Clin. Invest. 105, 1395–1406 (2000).
Ramirez, M. T., Zhao, X. L., Schulman, H. & Brown, J. H. The nuclear δB isoform of Ca2+/calmodulin-dependent protein kinase II regulates atrial natriuretic factor gene expression in ventricular myocytes. J. Biol. Chem. 272, 31203–31208 (1997).
Zhang, T., Miyamoto, S. & Brown, J. H. Cardiomyocyte calcium and calcium/calmodulin-dependent protein kinase II: friends or foes? Recent Prog. Horm. Res. 59, 141–168 (2004).
Zhu, W. et al. Ca2+/calmodulin-dependent kinase II and calcineurin play critical roles in endothelin-1-induced cardiomyocyte hypertrophy. J. Biol. Chem. 275, 15239–15245 (2000).
Boknik, P. et al. Enhanced protein phosphorylation in hypertensive hypertrophy. Cardiovasc. Res. 51, 717–728 (2001).
Colomer, J. M., Mao, L., Rockman, H. A. & Means, A. R. Pressure overload selectively up-regulates Ca2+/calmodulin-dependent protein kinase II in vivo. Mol. Endocrinol. 17, 183–192 (2003).
Currie, S. & Smith, G. L. Calcium/calmodulin-dependent protein kinase II activity is increased in sarcoplasmic reticulum from coronary artery ligated rabbit hearts. FEBS Lett. 459, 244–248 (1999).
Hagemann, D., Bohlender, J., Hoch, B., Krause, E. G. & Karczewski, P. Expression of Ca2+/calmodulin-dependent protein kinase II δ-subunit isoforms in rats with hypertensive cardiac hypertrophy. Mol. Cell. Biochem. 220, 69–76 (2001).
Zhang, T. et al. The δC isoform of CaMKII is activated in cardiac hypertrophy and induces dilated cardiomyopathy and heart failure. Circ. Res. 92, 912–919 (2003).
Kirchhefer, U., Schmitz, W., Scholz, H. & Neumann, J. Activity of cAMP-dependent protein kinase and Ca2+/calmodulin-dependent protein kinase in failing and nonfailing human hearts. Cardiovasc. Res. 42, 254–261 (1999).
Zhang, T. et al. The cardiac-specific nuclear δB isoform of Ca2+/calmodulin-dependent protein kinase II induces hypertrophy and dilated cardiomyopathy associated with increased protein phosphatase 2A activity. J. Biol. Chem. 277, 1261–1267 (2002).
Zhang T. et al. The δC isoform of CaMKII is activated in cardiac hypertrophy and induces dilated cardiomyopathy and heart failure. Circ. Res. 92, 912–919 (2003).
Zhang, R. et al. Calmodulin kinase II inhibition protects against structural heart disease. Nature Med. 11, 409–417 (2005). This demonstrated that CaM kinase inhibition in the heart, using either a peptide or a small molecule, is well tolerated and suppresses adverse cardiac remodelling in animal models.
Yang, Y. et al. Calmodulin kinase II inhibition protects against myocardial cell apoptosis in vivo. Am. J. Physiol. Heart Circ. Physiol. 291, H3065–H3075 (2006).
Zhu, W. Z. et al. Linkage of β1-adrenergic stimulation to apoptotic heart cell death through protein kinase A-independent activation of Ca2+/calmodulin kinase II. J. Clin. Invest. 111, 617–625 (2003).
Bers, D. M. & Guo, T. Calcium signaling in cardiac ventricular myocytes. Ann. N. Y. Acad. Sci. 1047, 86–98 (2005).
Dzhura, I., Wu, Y., Colbran, R. J., Balser, J. R. & Anderson, M. E. Calmodulin kinase determines calcium-dependent facilitation of L-type calcium channels. Nature Cell Biol. 2, 173–177 (2000).
Witcher, D. R., Kovacs, R. J., Schulman, H., Cefali, D. C. & Jones, L. R. Unique phosphorylation site on the cardiac ryanodine receptor regulates calcium channel activity. J. Biol. Chem. 266, 11144–11152 (1991).
Kohlhaas, M. et al. Increased sarcoplasmic reticulum calcium leak but unaltered contractility by acute CaMKII overexpression in isolated rabbit cardiac myocytes. Circ. Res. 98, 235–244 (2006).
Maier, L. S. et al. Transgenic CaMKIIδC overexpression uniquely alters cardiac myocyte Ca2+ handling: reduced SR Ca2+ load and activated SR Ca2+ release. Circ. Res. 92, 904–911 (2003).
Simmerman, H. K., Collins, J. H., Theibert, J. L., Wegener, A. D. & Jones, L. R. Sequence analysis of phospholamban. Identification of phosphorylation sites and two major structural domains. J. Biol. Chem. 261, 13333–13341 (1986).
Wagner, S. & Maier, L. S. Modulation of cardiac Na(+) and Ca(2+) currents by CaM and CaMKII. J. Cardiovasc. Electrophysiol. 17 (Suppl. 1), 26–33 (2006).
Ai, X., Curran, J. W., Shannon, T. R., Bers, D. M. & Pogwizd, S. M. Ca2+/calmodulin-dependent protein kinase modulates cardiac ryanodine receptor phosphorylation and sarcoplasmic reticulum Ca2+ leak in heart failure. Circ. Res. 97, 1314–1322 (2005).
Anderson, M. E. The fire from within: the biggest Ca2+ channel erupts and dribbles. Circ. Res. 97, 1213–1215 (2005).
Wu, Y. et al. Calmodulin kinase II and arrhythmias in a mouse model of cardiac hypertrophy. Circulation 106, 1288–1293 (2002).
Wu, X. et al. Local InsP3-dependent perinuclear Ca2+ signaling in cardiac myocyte excitation-transcription coupling. J. Clin. Invest. 116, 675–682 (2006).
Backs, J., Song, K., Bezprozvannaya, S., Chang, S. & Olson, E. N. CaM kinase II selectively signals to histone deacetylase 4 during cardiomyocyte hypertrophy. J. Clin. Invest. 116, 1853–1864 (2006).
Anderson, M. E., Higgins, L. S. & Schulman, H. Disease mechanisms and emerging therapies: protein kinases and their inhibitors in myocardial disease. Nature Clin. Pract. Cardiovasc. Med. 3, 437–445 (2006).
Hashimoto, Y. et al. Potent and preferential inhibition of Ca2+/calmodulin-dependent protein kinase II by K252a and its derivative, KT5926. Biochem. Biophys. Res. Commun. 181, 423–429 (1991).
Rosenberg, O. S., Deindl, S., Sung, R. J., Nairn, A. C. & Kuriyan, J. Structure of the autoinhibited kinase domain of CaMKII and SAXS analysis of the holoenzyme. Cell 123, 849–860 (2005).
Witte, S. et al. Inhibition of the c-Jun N-terminal kinase/AP-1 and NF-κB pathways by PICOT, a novel protein kinase C-interacting protein with a thioredoxin homology domain. J. Biol. Chem. 275, 1902–1909 (2000).
Jeong, D. et al. PICOT inhibits cardiac hypertrophy and enhances ventricular function and cardiomyocyte contractility. Circ. Res. 99, 307–314 (2006).
Chen, L. et al. Opposing cardioprotective actions and parallel hypertrophic effects of δPKC and εPKC. Proc. Natl Acad. Sci. USA 98, 11114–11119 (2001).
Mochly-Rosen, D. et al. Cardiotrophic effects of protein kinase C ε: analysis by in vivo modulation of PKCε translocation. Circ. Res. 86, 1173–1179 (2000).
Wu, G., Toyokawa, T., Hahn, H. & Dorn, G. W. Epsilon protein kinase C in pathological myocardial hypertrophy. Analysis by combined transgenic expression of translocation modifiers and Gα9. J. Biol. Chem. 275, 29927–29930 (2000).
Hahn, H. S. et al. Ischemic protection and myofibrillar cardiomyopathy: dose-dependent effects of in vivo δPKC inhibition. Circ. Res. 91, 741–748 (2002).
Klein, G. et al. Increased collagen deposition and diastolic dysfunction but preserved myocardial hypertrophy after pressure overload in mice lacking PKCε. Circ. Res. 96, 748–755 (2005).
Tuttle, K. R. et al. The effect of ruboxistaurin on nephropathy in type 2 diabetes. Diabetes Care 28, 2686–2690 (2005).
The PKC-DRS Study Group. The effect of ruboxistaurin on visual loss in patients with moderately severe to very severe nonproliferative diabetic retinopathy: initial results of the protein kinase C β inhibitor diabetic retinopathy study (PKC-DRS) multicenter randomized clinical trial. Diabetes 54, 2188–2197 (2005).
Boyle, A. J. et al. Inhibition of protein kinase C reduces left ventricular fibrosis and dysfunction following myocardial infarction. J. Mol. Cell. Cardiol. 39, 213–221 (2005). This demonstrated a beneficial effect of a small-molecule inhibitor of PKC in the prevention of post-MI remodelling in rats.
Davies, S. P., Reddy, H., Caivano, M. & Cohen, P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem. J. 351, 95–105 (2000).
Roman, B. B., Geenen, D. L., Leitges, M. & Buttrick, P. M. PKC-β is not necessary for cardiac hypertrophy. Am. J. Physiol Heart Circ. Physiol. 280, H2264–H2270 (2001).
Hahn, H. S. et al. Protein kinase Cα negatively regulates systolic and diastolic function in pathological hypertrophy. Circ. Res. 93, 1111–1119 (2003).
Braz, J. C. et al. PKC-α regulates cardiac contractility and propensity toward heart failure. Nature Med. 10, 248–254 (2004).
Bowling, N. et al. Increased protein kinase C activity and expression of Ca2+-sensitive isoforms in the failing human heart. Circulation 99, 384–391 (1999).
Hambleton, M. et al. Pharmacological- and gene therapy-based inhibition of protein kinase Cα/β enhances cardiac contractility and attenuates heart failure. Circulation 114, 574–582 (2006). This demonstrated that small-molecule inhibitors of PKC enhance cardiac contractility in animal models.
McKinsey, T. A. & Olson, E. N. Toward transcriptional therapies for the failing heart: chemical screens to modulate genes. J. Clin. Invest. 115, 538–546 (2005).
Verdin, E., Dequiedt, F. & Kasler, H. G. Class II histone deacetylases: versatile regulators. Trends Genet. 19, 286–293 (2003).
Chang, S. et al. Histone deacetylases 5 and 9 govern responsiveness of the heart to a subset of stress signals and play redundant roles in heart development. Mol. Cell. Biol. 24, 8467–8476 (2004).
Zhang, C. L. et al. Class II histone deacetylases act as signal-responsive repressors of cardiac hypertrophy. Cell 110, 479–488 (2002).
Han, A., He, J., Wu, Y., Liu, J. O. & Chen, L. Mechanism of recruitment of class II histone deacetylases by myocyte enhancer factor-2. J. Mol. Biol. 345, 91–102 (2005).
Dai, Y. S., Xu, J. & Molkentin, J. D. The DnaJ-related factor Mrj interacts with nuclear factor of activated T cells c3 and mediates transcriptional repression through class II histone deacetylase recruitment. Mol. Cell Biol. 25, 9936–9948 (2005).
Xing, W. et al. Myocardin induces cardiomyocyte hypertrophy. Circ. Res. 98, 1089–1097 (2006).
Sucharov, C. C., Langer, S. J., Bristow, M. R. & Leinwand, L. A. Shuttling of HDAC5 in H9C2 cells regulates YY1 function through CaMKIV/PKD and PP2A. Am. J. Physiol. Cell. Physiol. 291, C1029–C1037 (2006).
Song, K. et al. The transcriptional coactivator CAMTA2 stimulates cardiac growth by opposing class II histone deacetylases. Cell 125, 453–466 (2006).
Grozinger, C. M. & Schreiber, S. L. Regulation of histone deacetylase 4 and 5 and transcriptional activity by 14-3-3-dependent cellular localization. Proc. Natl Acad. Sci. USA 97, 7835–7840 (2000).
McKinsey, T. A., Zhang, C. L. & Olson, E. N. Activation of the myocyte enhancer factor-2 transcription factor by calcium/calmodulin-dependent protein kinase-stimulated binding of 14-3-3 to histone deacetylase 5. Proc. Natl Acad. Sci. USA 97, 14400–14405 (2000).
McKinsey, T. A., Zhang, C. L., Lu, J. & Olson, E. N. Signal-dependent nuclear export of a histone deacetylase regulates muscle differentiation. Nature 408, 106–111 (2000).
McKinsey, T. A., Zhang, C. L. & Olson, E. N. Identification of a signal-responsive nuclear export sequence in class II histone deacetylases. Mol. Cell. Biol. 21, 6312–6321 (2001).
Wang, A. H. et al. Regulation of histone deacetylase 4 by binding of 14-3-3 proteins. Mol. Cell. Biol. 20, 6904–6912 (2000).
Harrison, B. C. et al. The CRM1 nuclear export receptor controls pathological cardiac gene expression. Mol. Cell. Biol. 24, 10636–10649 (2004).
Vega, R. B. et al. Protein kinases C and D mediate agonist-dependent cardiac hypertrophy through nuclear export of histone deacetylase 5. Mol. Cell. Biol. 24, 8374–8385 (2004).
Harrison, B. C. et al. Regulation of cardiac stress signaling by protein kinase D1. Mol. Cell. Biol. 26, 3875–3888 (2006).
Gschwendt, M. et al. Inhibition of protein kinase C μ by various inhibitors. Differentiation from protein kinase c isoenzymes. FEBS Lett. 392, 77–80 (1996).
Cohen, P. Protein kinases — the major drug targets of the twenty-first century? Nature Rev. Drug Discov. 1, 309–315 (2002).
Rozengurt, E., Rey, O. & Waldron, R. T. Protein kinase D signaling. J. Biol. Chem. 280, 13205–13208 (2005).
Antos, C. L. et al. Dose-dependent blockade to cardiomyocyte hypertrophy by histone deacetylase inhibitors. J. Biol. Chem. 278, 28930–28937 (2003).
Kee, H. J. et al. Inhibition of histone deacetylation blocks cardiac hypertrophy induced by angiotensin II infusion and aortic banding. Circulation 113, 51–59 (2006). Together with reference 265, demonstrated that small-molecule inhibitors of HDACs are efficacious in the prevention and reversal of pathological cardiac hypertrophy in animal models.
Kong, Y. et al. Suppression of class I and II histone deacetylases blunts pressure-overload cardiac hypertrophy. Circulation 113, 2579–2588 (2006).
Kook, H. et al. Cardiac hypertrophy and histone deacetylase-dependent transcriptional repression mediated by the atypical homeodomain protein Hop. J. Clin. Invest 112, 863–871 (2003).
Danzi, S., Dubon, P. & Klein, I. Effect of serum triiodothyronine on regulation of cardiac gene expression: role of histone acetylation. Am. J. Physiol Heart Circ. Physiol. 289, H1506–H1511 (2005).
Davis, F. J., Pillai, J. B., Gupta, M. & Gupta, M. P. Concurrent opposite effects of trichostatin A, an inhibitor of histone deacetylases, on expression of α-MHC and cardiac tubulins: implication for gain in cardiac muscle contractility. Am. J. Physiol Heart Circ. Physiol. 288, H1477–H1490 (2005).
Braunwald, E. & Bristow, M. R. Congestive heart failure: fifty years of progress. Circulation 102, IV14–IV23 (2000).
Fischle, W. et al. Enzymatic activity associated with class II HDACs is dependent on a multiprotein complex containing HDAC3 and SMRT/N-CoR. Mol. Cell 9, 45–57 (2002).
Zhang, C. L., McKinsey, T. A., Lu, J. R. & Olson, E. N. Association of COOH-terminal-binding protein (CtBP) and MEF2-interacting transcription repressor (MITR) contributes to transcriptional repression of the MEF2 transcription factor. J. Biol. Chem. 276, 35–39 (2001).
Zhang, C. L., McKinsey, T. A. & Olson, E. N. Association of class II histone deacetylases with heterochromatin protein 1: potential role for histone methylation in control of muscle differentiation. Mol. Cell. Biol. 22, 7302–7312 (2002).
Han, A. et al. Sequence-specific recruitment of transcriptional co-repressor Cabin1 by myocyte enhancer factor-2. Nature 422, 730–734 (2003).
Youn, H. D., Grozinger, C. M. & Liu, J. O. Calcium regulates transcriptional repression of myocyte enhancer factor 2 by histone deacetylase 4. J. Biol. Chem. 275, 22563–22567 (2000).
Alcendor, R. R., Kirshenbaum, L. A., Imai, S., Vatner, S. F. & Sadoshima, J. Silent information regulator 2α, a longevity factor and class III histone deacetylase, is an essential endogenous apoptosis inhibitor in cardiac myocytes. Circ. Res. 95, 971–980 (2004).
Trivedi, C. M. et al. HDAC2 regulates the cardiac hypertrophic response by modulating GSK3β activity. Nature Med. 13, 324–331 (2007).
Minagawa, T., Ijuin, T., Mochizuki, Y. & Takenawa, T. Identification and characterization of a sac domain-containing phosphoinositide 5-phosphatase. J. Biol. Chem. 276, 22011–22015 (2001).
Glozak, M. A., Sengupta, N., Zhang, X. & Seto, E. Acetylation and deacetylation of non-histone proteins. Gene 363, 15–23 (2005).
Heineke, J. & Molkentin, J. D. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nature Rev. Mol. Cell Biol. 7, 589–600 (2006).
Behr, T. M. et al. Hypertensive end-organ damage and premature mortality are p38 mitogen-activated protein kinase-dependent in a rat model of cardiac hypertrophy and dysfunction. Circulation 104, 1292–1298 (2001).
Kyoi, S. et al. Opposing effect of p38 MAP kinase and JNK inhibitors on the development of heart failure in the cardiomyopathic hamster. Cardiovasc. Res. 69, 888–898 (2006).
Liu, Y. H. et al. Inhibition of p38 mitogen-activated protein kinase protects the heart against cardiac remodeling in mice with heart failure resulting from myocardial infarction. J. Card. Fail. 11, 74–81 (2005).
Zhang, S. et al. Role of 14-3-3-mediated p38 mitogen-activated protein kinase inhibition in cardiac myocyte survival. Circ. Res. 93, 1026–1028 (2003).
Beckles, D. L., Mascareno, E. & Siddiqui, M. A. Inhibition of Jak2 phosphorylation attenuates pressure overload cardiac hypertrophy. Vascul. Pharmacol. 45, 350–357 (2006).
Palfi, A. et al. PARP inhibition prevents postinfarction myocardial remodeling and heart failure via the protein kinase C/glycogen synthase kinase-3β pathway. J. Mol. Cell. Cardiol. 41, 149–159 (2006).
Sano, M. et al. Activation and function of cyclin T-Cdk9 (positive transcription elongation factor-b) in cardiac muscle-cell hypertrophy. Nature Med. 8, 1310–1317 (2002).
Sausville, E. A. Cyclin-dependent kinase modulators studied at the NCI: pre-clinical and clinical studies. Curr. Med. Chem. Anticancer Agents 3, 47–56 (2003).
Oh, H. et al. Telomere attrition and CHK2 activation in human heart failure. Proc. Natl Acad. Sci. USA 100, 5378–5383 (2003).
Arienti, K. L. et al. Checkpoint kinase inhibitors: SAR and radioprotective properties of a series of 2-arylbenzimidazoles. J. Med. Chem. 48, 1873–1885 (2005).
Harris, I. S. et al. Raf-1 kinase is required for cardiac hypertrophy and cardiomyocyte survival in response to pressure overload. Circulation 110, 718–723 (2004).
Schreck, R. & Rapp, U. R. Raf kinases: oncogenesis and drug discovery. Int. J. Cancer 119, 2261–2271 (2006).
Davidson, S. M. et al. The transcriptional coactivator p300 plays a critical role in the hypertrophic and protective pathways induced by phenylephrine in cardiac cells but is specific to the hypertrophic effect of urocortin. Chembiochem. 6, 162–170 (2005).
Lau, O. D. et al. HATs off: selective synthetic inhibitors of the histone acetyltransferases p300 and PCAF. Mol. Cell 5, 589–595 (2000).
Owan, T. E. et al. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N. Engl. J. Med. 355, 251–259 (2006).
Klapholz, M. et al. Hospitalization for heart failure in the presence of a normal left ventricular ejection fraction: results of the New York Heart Failure Registry. J. Am. Coll. Cardiol. 43, 1432–1438 (2004).
Fatkin, D. et al. An abnormal Ca(2+) response in mutant sarcomere protein-mediated familial hypertrophic cardiomyopathy. J. Clin. Invest. 106, 1351–1359 (2000).
Acknowledgements
T.A.M. thanks E. Bush for helpful discussions and L. Castonguay for assistance with the compound structures. D.A.K. is supported by NIH-PO1-HL-077180 and PO1-HL-59408 grants, and the Peter Belfer Laboratory Research Fund.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
T.A.M. is an employee of Gilead Sciences and owns shares of Gilead stock. He has been involved in the assessment of a number of the small-molecule inhibitors described in the Review.
D.A.K's laboratory receives funding from BioMarin, who manufacture tetrahydrobiopterin, which is discussed in this Review.
Related links
Glossary
- Sarcomere
-
The basic functional contractile unit of muscle. It is composed largely of the proteins actin and myosin.
- Myosin
-
An ATPase that regulates contraction through association with actin.
- Crossbridges
-
A mysosin–actin junction.
- Actin
-
A primary thin filament contractile protein in the sarcomere.
- Z bands
-
Delimit the sarcomere.
Rights and permissions
About this article
Cite this article
McKinsey, T., Kass, D. Small-molecule therapies for cardiac hypertrophy: moving beneath the cell surface. Nat Rev Drug Discov 6, 617–635 (2007). https://doi.org/10.1038/nrd2193
Issue Date:
DOI: https://doi.org/10.1038/nrd2193
This article is cited by
-
Astragaloside IV derivative HHQ16 ameliorates infarction-induced hypertrophy and heart failure through degradation of lncRNA4012/9456
Signal Transduction and Targeted Therapy (2023)
-
Deubiquitinase JOSD2 improves calcium handling and attenuates cardiac hypertrophy and dysfunction by stabilizing SERCA2a in cardiomyocytes
Nature Cardiovascular Research (2023)
-
Current and emerging drug targets in heart failure treatment
Heart Failure Reviews (2022)
-
NRF1-enhanced miR-4458 alleviates cardiac hypertrophy through releasing TTP-inhibited TFAM
In Vitro Cellular & Developmental Biology - Animal (2020)
-
C1q-TNF-related protein-3 attenuates pressure overload-induced cardiac hypertrophy by suppressing the p38/CREB pathway and p38-induced ER stress
Cell Death & Disease (2019)