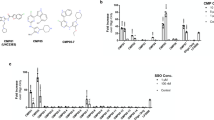
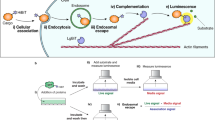
Abstract
Developing efficient delivery vectors for bioactive molecules is of great importance within both traditional and novel drug development, such as oligonucleotide (ON)-based therapeutics. To address delivery efficiency using cell-penetrating peptides (CPPs), we here present a protocol based on splice correction utilizing both neutral and anionic antisense ONs, either covalently conjugated via a disulfide bridge or non-covalently complexed, respectively, that generates positive readout in the form of luciferase expression. The decisive advantage of using splice correction for evaluation of CPPs is that the ON induces a biological response in contrast to traditionally used methods, for example, fluorescently labeled peptides. An emerging number of studies emphasize the role of endocytosis in translocation of CPPs, and this protocol is also utilized to determine the relative contribution of different endocytic pathways in the uptake of CPPs, which provides valuable information for future design of novel, more potent CPPs for bioactive cargoes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Langel, Ü. Handbook of Cell-Penetrating Peptides 2nd edn. (CRC Press, Boca Raton, FL, 2006).
El-Andaloussi, S., Holm, T. & Langel, Ü. Cell-penetrating peptides: mechanisms and applications. Curr. Pharm. Des. 11, 3597–3611 (2005).
Wadia, J.S., Stan, R.V. & Dowdy, S.F. Transducible TAT-HA fusogenic peptide enhances escape of TAT-fusion proteins after lipid raft macropinocytosis. Nat. Med. 10, 310–315 (2004).
El-Andaloussi, S., Johansson, H.J., Lundberg, P. & Langel, Ü. Induction of splice correction by cell-penetrating peptide nucleic acids. J. Gene Med. 8, 1262–1273 (2006).
Richard, J.P. et al. Cell-penetrating peptides. A reevaluation of the mechanism of cellular uptake. J. Biol. Chem. 278, 585–590 (2003).
Kang, S.H., Cho, M.J. & Kole, R. Up-regulation of luciferase gene expression with antisense oligonucleotides: implications and applications in functional assay development. Biochemistry 37, 6235–6239 (1998).
Sazani, P. & Kole, R. Therapeutic potential of antisense oligonucleotides as modulators of alternative splicing. J. Clin. Invest. 112, 481–486 (2003).
Faustino, N.A. & Cooper, T.A. Pre-mRNA splicing and human disease. Genes Dev. 17, 419–437 (2003).
Astriab-Fisher, A., Sergueev, D., Fisher, M., Shaw, B.R. & Juliano, R.L. Conjugates of antisense oligonucleotides with the Tat and antennapedia cell-penetrating peptides: effects on cellular uptake, binding to target sequences, and biologic actions. Pharm. Res. 19, 744–754 (2002).
Bendifallah, N. et al. Evaluation of cell-penetrating peptides (CPPs) as vehicles for intracellular delivery of antisense peptide nucleic acid (PNA). Bioconjug. Chem. 17, 750–758 (2006).
Holm, T. et al. Studying the uptake of cell-penetrating peptides. Nat. Protoc. 1, 1001–1005 (2006).
Shiraishi, T. & Nielsen, P.E. Enhanced delivery of cell-penetrating peptide-peptide nucleic acid conjugates by endosomal disruption. Nat. Protoc. 1, 633–636 (2006).
Horonchik, L. et al. Heparan sulfate is a cellular receptor for purified infectious prions. J. Biol. Chem. 280, 17062–17067 (2005).
Moulton, H.M., Nelson, M.H., Hatlevig, S.A., Reddy, M.T. & Iversen, P.L. Cellular uptake of antisense morpholino oligomers conjugated to arginine-rich peptides. Bioconjug. Chem. 15, 290–299 (2004).
Abes, S. et al. Vectorization of morpholino oligomers by the (R-Ahx-R)4 peptide allows efficient splicing correction in the absence of endosomolytic agents. J. Control. Rel. 116, 304–313 (2006).
Nelson, M.H. et al. Arginine-rich peptide conjugation to morpholino oligomers: effects on antisense activity and specificity. Bioconjug. Chem. 16, 959–966 (2005).
Hällbrink, M., Oehlke, J., Papsdorf, G. & Bienert, M. Uptake of cell-penetrating peptides is dependent on peptide-to-cell ratio rather than on peptide concentration. Biochim. Biophys. Acta 1667, 222–228 (2004).
EL Andaloussi, S., Johansson, H., Holm, T. & Langel, Ü. A novel cell-penetrating peptide, M918, for efficient delivery of proteins and peptide nucleic acids. Mol. Ther. (in the press).
Simeoni, F., Morris, M.C., Heitz, F. & Divita, G. Insight into the mechanism of the peptide-based gene delivery system MPG: implications for delivery of siRNA into mammalian cells. Nucleic Acids Res. 31, 2717–2724 (2003).
Acknowledgements
This work was funded by the Swedish Research Council (VR-NT, VR-Med), Swedish Governmental Agency for Innovation Systems (VINNOVA SAMBIO) and Swedish Center for Biomembrane Research. HeLa pLuc 705 cells were kindly provided by R. Kole and B. Lebleu. We thank Henrik Johansson for assistance with statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Andaloussi, S., Guterstam, P. & Langel, Ü. Assessing the delivery efficacy and internalization route of cell-penetrating peptides. Nat Protoc 2, 2043–2047 (2007). https://doi.org/10.1038/nprot.2007.302
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.302
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.