Abstract
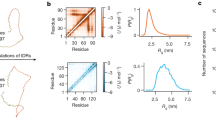
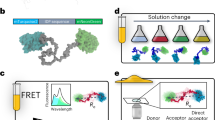
Thousands of human proteins lack recognizable tertiary structure in most of their chains. Here we hypothesize that some use their structured N-terminal domains (SNTDs) to organise the remaining protein chain via intramolecular interactions, generating partially structured proteins. This model has several attractive features: as protein chains emerge, SNTDs form spontaneously and serve as nucleation points, creating more compact shapes. This reduces the risk of protein degradation or aggregation. Moreover, an interspersed pattern of SNTD-docked regions and free loops can coordinate assembly of sub-complexes in defined loop-sections and enables novel regulatory mechanisms, for example through posttranslational modifications of docked regions.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simister, P., Schaper, F., O'Reilly, N. et al. Self-organization of intrinsically disordered proteins with folded N-termini . Nat Prec (2010). https://doi.org/10.1038/npre.2010.5124.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2010.5124.1