Abstract
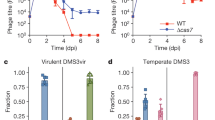
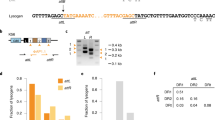
Temperate phages are common, and prophages are abundant residents of sequenced bacterial genomes. Mycobacteriophages are viruses that infect mycobacterial hosts including Mycobacterium tuberculosis and Mycobacterium smegmatis, encompass substantial genetic diversity and are commonly temperate. Characterization of ten Cluster N temperate mycobacteriophages revealed at least five distinct prophage-expressed viral defence systems that interfere with the infection of lytic and temperate phages that are either closely related (homotypic defence) or unrelated (heterotypic defence) to the prophage. Target specificity is unpredictable, ranging from a single target phage to one-third of those tested. The defence systems include a single-subunit restriction system, a heterotypic exclusion system and a predicted (p)ppGpp synthetase, which blocks lytic phage growth, promotes bacterial survival and enables efficient lysogeny. The predicted (p)ppGpp synthetase coded by the Phrann prophage defends against phage Tweety infection, but Tweety codes for a tetrapeptide repeat protein, gp54, which acts as a highly effective counter-defence system. Prophage-mediated viral defence offers an efficient mechanism for bacterial success in host–virus dynamics, and counter-defence promotes phage co-evolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hambly, E. & Suttle, C. A. The viriosphere, diversity, and genetic exchange within phage communities. Curr. Opin. Microbiol. 8, 444–450 (2005).
Abedon, S. T. Phage evolution and ecology. Adv. Appl. Microbiol. 67, 1–45 (2009).
Jacobs-Sera, D. et al. On the nature of mycobacteriophage diversity and host preference. Virology 434, 187–201 (2012).
Hendrix, R. W. Bacteriophages: evolution of the majority. Theor. Popul. Biol. 61, 471–480 (2002).
Suttle, C. A. Marine viruses—major players in the global ecosystem. Nat. Rev. Microbiol. 5, 801–812 (2007).
Barrangou, R. et al. CRISPR provides acquired resistance against viruses in prokaryotes. Science 315, 1709–1712 (2007).
Arber, W. & Dussoix, D. Host specificity of DNA produced by Escherichia coli. I. Host controlled modification of bacteriophage lambda. J. Mol. Biol. 5, 18–36 (1962).
Dussoix, D. & Arber, W. Host specificity of DNA produced by Escherichia coli. II. Control over acceptance of DNA from infecting phage lambda. J. Mol. Biol. 5, 37–49 (1962).
Tock, M. R. & Dryden, D. T. The biology of restriction and anti-restriction. Curr. Opin. Microbiol. 8, 466–472 (2005).
Swarts, D. C. et al. DNA-guided DNA interference by a prokaryotic Argonaute. Nature 507, 258–261 (2014).
Goldfarb, T. et al. BREX is a novel phage resistance system widespread in microbial genomes. EMBO J. 34, 169–183 (2015).
Fineran, P. C. et al. The phage abortive infection system, ToxIN, functions as a protein–RNA toxin–antitoxin pair. Proc. Natl Acad. Sci. USA 106, 894–899 (2009).
Chopin, M.-C., Chopin, A. & Bidnenko, E. Phage abortive infection in lactococci: variations on a theme. Curr. Opin. Microbiol. 8, 473–479 (2005).
Bondy-Denomy, J., Pawluk, A., Maxwell, K. L. & Davidson, A. R. Bacteriophage genes that inactivate the CRISPR/Cas bacterial immune system. Nature 493, 429–432 (2013).
Blower, T. R., Evans, T. J., Przybilski, R., Fineran, P. C. & Salmond, G. P. C. Viral evasion of a bacterial suicide system by RNA-based molecular mimicry enables infectious altruism. PLoS Genet. 8, e1003023 (2012).
Casjens, S. Prophages and bacterial genomics: what have we learned so far? Mol. Microbiol. 49, 277–300 (2003).
Knowles, B. et al. Lytic to temperate switching of viral communities. Nature 531, 466–470 (2016).
Fortier, L.-C. & Sekulovic, O. Importance of prophages to evolution and virulence of bacterial pathogens. Virulence 4, 354–365 (2013).
Kita, K., Kawakami, H. & Tanaka, H. Evidence for horizontal transfer of the EcoT38I restriction-modification gene to chromosomal DNA by the P2 phage and diversity of defective P2 prophages in Escherichia coli TH38 strains. J. Bacteriol. 185, 2296–2305 (2003).
Hofer, B., Ruge, M. & Dreiseikelmann, B. The superinfection exclusion gene (sieA) of bacteriophage P22: identification and overexpression of the gene and localization of the gene product. J. Bacteriol. 177, 3080–3086 (1995).
Cumby, N., Edwards, A. M., Davidson, A. R. & Maxwell, K. L. The bacteriophage HK97 gp15 moron element encodes a novel superinfection exclusion protein. J. Bacteriol. 194, 5012–5019 (2012).
Pope, W. H. et al. Whole genome comparison of a large collection of mycobacteriophages reveals a continuum of phage genetic diversity. eLife 4, e06416 (2015).
Hatfull, G. F. et al. Comparative genomic analysis of 60 mycobacteriophage genomes: genome clustering, gene acquisition, and gene size. J. Mol. Biol. 397, 119–143 (2010).
Hendrix, R. W., Smith, M. C., Burns, R. N., Ford, M. E. & Hatfull, G. F. Evolutionary relationships among diverse bacteriophages and prophages: all the world's a phage. Proc. Natl Acad. Sci. USA 96, 2192–2197 (1999).
Pedulla, M. L. et al. Origins of highly mosaic mycobacteriophage genomes. Cell 113, 171–182 (2003).
Hatfull, G. F. The secret lives of mycobacteriophages. Adv. Virus Res. 82, 179–288 (2012).
Jordan, T. C. et al. A broadly implementable research course in phage discovery and genomics for first-year undergraduate students. mBio 5, e01051-13 (2014).
Hatfull, G. F. et al. Complete genome sequences of 63 mycobacteriophages. Genome Announc. 1, e00847-13 (2013).
Hatfull, G. F. et al. Complete genome sequences of 61 mycobacteriophages. Genome Announc. 4, e00389-16 (2016).
Broussard, G. W. et al. Integration-dependent bacteriophage immunity provides insights into the evolution of genetic switches. Mol. Cell. 49, 237–248 (2013).
Oliveira, P. H., Touchon, M. & Rocha, E. P. The interplay of restriction-modification systems with mobile genetic elements and their prokaryotic hosts. Nucleic Acids Res. 42, 10618–10631 (2014).
Pham, T. T., Jacobs-Sera, D., Pedulla, M. L., Hendrix, R. W. & Hatfull, G. F. Comparative genomic analysis of mycobacteriophage Tweety: evolutionary insights and construction of compatible site-specific integration vectors for mycobacteria. Microbiology 153, 2711–2723 (2007).
Tahar, R., Ringwald, P. & Basco, L. K. Heterogeneity in the circumsporozoite protein gene of Plasmodium malariae isolates from sub-Saharan Africa. Mol. Biochem. Parasitol. 92, 71–78 (1998).
Booth, H. A. & Holland, P. W. H. Annotation, nomenclature and evolution of four novel homeobox genes expressed in the human germ line. Gene 387, 7–14 (2007).
Soding, J., Biegert, A. & Lupas, A. N. The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res. 33, W244–W248 (2005).
Hogg, T., Mechold, U., Malke, H., Cashel, M. & Hilgenfeld, R. Conformational antagonism between opposing active sites in a bifunctional RelA/SpoT homolog modulates (p)ppGpp metabolism during the stringent response [corrected]. Cell 117, 57–68 (2004).
Steinchen, W. & Bange, G. The magic dance of the alarmones (p)ppGpp. Mol. Microbiol. 101, 531–544 (2016).
Yang, J. & Zhang, Y. Protein structure and function prediction using I-TASSER. Curr. Protoc. Bioinformatics 52, 5.8.1–5.8.15 (2015).
Kelley, L. A., Mezulis, S., Yates, C. M., Wass, M. N. & Sternberg, M. J. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 10, 845–858 (2015).
Potrykus, K. & Cashel, M. (P)ppGpp still magical? Annu. Rev. Microbiol. 62, 35–51 (2008).
Bondy-Denomy, J. et al. Prophages mediate defense against phage infection through diverse mechanisms. ISME J. 10, 2854–2866 (2016).
Pope, W. H. et al. Cluster M mycobacteriophages Bongo, PegLeg, and Rey with unusually large repertoires of tRNA isotypes. J. Virol. 88, 2461–2480 (2014).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Cresawn, S. G. et al. Phamerator: a bioinformatic tool for comparative bacteriophage genomics. BMC Bioinformatics 12, 395 (2011).
Marinelli, L. J. et al. BRED: a simple and powerful tool for constructing mutant and recombinant bacteriophage genomes. PLoS ONE 3, e3957 (2008).
Lee, M. H., Pascopella, L., Jacobs, W. R. Jr & Hatfull, G. F. Site-specific integration of mycobacteriophage L5: integration-proficient vectors for Mycobacterium smegmatis, Mycobacterium tuberculosis, and bacille Calmette-Guérin. Proc. Natl Acad. Sci. USA 88, 3111–3115 (1991).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Li, H. et al. The sequence alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Thorvaldsdottir, H., Robinson, J. T. & Mesirov, J. P. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform. 14, 178–192 (2013).
Krumsiek, J., Arnold, R. & Rattei, T. Gepard: a rapid and sensitive tool for creating dotplots on genome scale. Bioinformatics 23, 1026–1028 (2007).
Huson, D. H. SplitsTree: analyzing and visualizing evolutionary data. Bioinformatics 14, 68–73 (1998).
Hatfull, G. F. et al. Exploring the mycobacteriophage metaproteome: phage genomics as an educational platform. PLoS Genet. 2, e92 (2006).
Acknowledgements
The authors thank the many students in the SEA-PHAGES programme that contributed to the isolation, annotation and characterization of the phages described here. Specific contributions are noted at http://phagesdb.org. The authors thank J. Schiebel, A. Jonas, T. Stoner, D. Green, R. Rush and L. Lin for help with escape mutant isolation, C.-C. Ko for help with plasmid construction and D. Asai, V. Sivanathan, K. Bradley and L. Barker for support of the SEA-PHAGES programme. This work was supported by grants from the National Institutes of Health (GM116884) and the Howard Hughes Medical Institute (54308198) to G.F.H. and a National Science Foundation pre-doctoral fellowship to T.N.M. (no. 1247842).
Author information
Authors and Affiliations
Contributions
R.M.D., D.J.-S., C.A.G.B., T.N.M., W.H.P., V.C.W., J.W. and G.F.H. conceived and designed the experiments. R.M.D., D.J.-S., C.A.G.B., R.A.G., W.H.P., J.C.C.R., D.A.R., B.R.B., C.F.H., C.M.M., M.T.M. and J.N.T. performed the experiments. R.M.D., D.J.-S., C.A.G.B., R.A.G., T.N.M., W.H.P., B.R.B., J.C.C.R., D.A.R., T.A., R.A., J.A.B., J.S.B., D.B., S.G.C., W.B.D., L.A.D., N.P.E., A.M.F., U.G., J.H.G., C.F.H., L.E.H., K.W.H., S.I., A.A.J., M.A.K., K.K.K., C.M.M., S.F.M., S.D.M., M.T.M., J.N., S.T.P., M.C.P., M.K.P., C.A.R., C.J.R., M.R.R., J.N.T., E.V., V.C.W., J.W. and G.F.H. analysed the data. S.G.C. contributed material/analysis tools. R.M.D., D.J.-S., T.N.M., W.H.P., D.A.R., T.A., R.A., J.A.B., J.S.B., D.B., S.G.C., W.B.D., L.A.D., N.P.E., A.M.F., U.G., J.H.G., L.E.H., K.W.H., S.I., A.A.J., M.A.K., K.K.K., C.M.M., S.F.M., S.D.M., J.N., S.T.P., M.C.P., M.K.P., C.A.R., C.J.R., M.R.R., E.V., V.C.W., J.W. and G.F.H. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Discussion; Supplementary References; Supplementary Tables 1–4; Supplementary Figures 1–22 (PDF 10476 kb)
Supplementary Table 5
Oligonucleotides used in this study (XLSX 30 kb)
Rights and permissions
About this article
Cite this article
Dedrick, R., Jacobs-Sera, D., Bustamante, C. et al. Prophage-mediated defence against viral attack and viral counter-defence. Nat Microbiol 2, 16251 (2017). https://doi.org/10.1038/nmicrobiol.2016.251
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.251
This article is cited by
-
Anti-phage defence through inhibition of virion assembly
Nature Communications (2024)
-
Bacteriophages avoid autoimmunity from cognate immune systems as an intrinsic part of their life cycles
Nature Microbiology (2024)
-
Genetic diversity of RNA viruses infecting invertebrate pests of rice
Science China Life Sciences (2024)
-
Benzo[a]pyrene stress impacts adaptive strategies and ecological functions of earthworm intestinal viromes
The ISME Journal (2023)
-
Prediction and characterization of prophages of Stenotrophomonas maltophilia reveals a remarkable phylogenetic diversity of prophages
Scientific Reports (2023)