Abstract
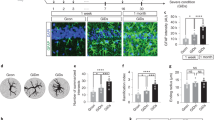
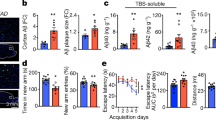
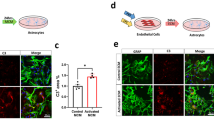
Alzheimer disease (AD) is a progressive neurodegenerative disorder characterized by excessive deposition of amyloid-β (Aβ) peptides in the brain. One of the earliest neuropathological changes in AD is the accumulation of astrocytes at sites of Aβ deposition1, but the cause or significance of this cellular response is unclear. Here we show that cultured adult mouse astrocytes migrate in response to monocyte chemoattractant protein-1 (MCP-1), a chemokine present in AD lesions1, and cease migration upon interaction with immobilized Aβ1–42. We also show that astrocytes bind and degrade Aβ1–42. Astrocytes plated on Aβ-laden brain sections from a mouse model of AD associate with the Aβ deposits and reduce overall Aβ levels in these sections. Our results suggest a novel mechanism for the accumulation of astrocytes around Aβ deposits, indicate a direct role for astrocytes in degradation of Aβ and implicate deficits in astroglial clearance of Aβ in the pathogenesis of AD. Treatments that increase removal of Aβ by astrocytes may therefore be a critical mechanism to reduce the neurodegeneration associated with AD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Role of inflammatory processes and microglial activation in Alzheimer's disease. In Alzheimer's Disease (ed. Terry, R.D.) chap. 26, 390 (Lippincott Williams & Wilkins, Philadelphia, Pennsylvania, 1999).
Heesen, M. et al. Mouse astrocytes respond to the chemokines MCP-1 and KC, but reverse transcriptase-polymerase chain reaction does not detect mRNA for the KC or new MCP-1 receptor. J. Neurosci. Res. 45, 382–391 (1996).
Hayashi, M., Luo, Y., Laning, J., Strieter, R.M. & Dorf, M.E. Production and function of monocyte chemoattractant protein-1 and other β-chemokines in murine glial cells. J. Neuroimmunol. 60, 143–150 (1995).
Johnstone, M., Gearing, A.J. & Miller, K.M. A central role for astrocytes in the inflammatory response to β-amyloid; chemokines, cytokines and reactive oxygen species are produced. J. Neuroimmunol. 93, 182–193 (1999).
Rezaie, P., Trillo-Pazos, G., Everall, I.P. & Male, D.K. Expression of β-chemokines and chemokine receptors in human fetal astrocyte and microglial co-cultures: potential role of chemokines in the developing CNS. Glia 37, 64–75 (2002).
Ishizuka, K. et al. Identification of monocyte chemoattractant protein-1 in senile plaques and reactive microglia of Alzheimer's disease. Psychiat. Clin. Neurosci. 51, 135–138 (1997).
Mehlhorn, G., Hollborn, M. & Schliebs, R. Induction of cytokines in glial cells surrounding cortical β-amyloid plaques in transgenic Tg2576 mice with Alzheimer pathology. Int. J. Dev. Neurosci. 18, 423–431 (2000).
Tezel, G., Hernandez, M.R. & Wax, M.B. In vitro evaluation of reactive astrocyte migration, a component of tissue remodeling in glaucomatous optic nerve head. Glia 34, 178–189 (2001).
Tawil, N.J. et al. α 1 β 1 integrin heterodimer functions as a dual laminin/collagen receptor in neural cells. Biochemistry 29, 6540–6544 (1990).
Husemann, J., Loike, J.D., Anankov, R., Febbraio, M. & Silverstein, S.C. Scavenger receptors in neurobiology and neuropathology: their role on microglia and other cells of the nervous system. Glia 40, 195–205 (2002).
Husemann, J., Loike, J.D., Kodama, T. & Silverstein, S.C. Scavenger receptor class B type I (SR-BI) mediates adhesion of neonatal murine microglia to fibrillar β-amyloid. J. Neuroimmunol. 114, 142–150 (2001).
Wegiel, J., Wang, K.C., Tarnawski, M. & Lach, B. Microglia cells are the driving force in fibrillar plaque formation, whereas astrocytes are a leading factor in plague degradation. Acta Neuropathol. (Berl.) 100, 356–364 (2000).
Kurt, M.A., Davies, D.C. & Kidd, M. β-amyloid immunoreactivity in astrocytes in Alzheimer's disease brain biopsies: an electron microscope study. Exp. Neurol. 158, 221–228 (1999).
Thal, D.R. et al. Amyloid β-protein (Aβ)-containing astrocytes are located preferentially near N-terminal-truncated Aβ deposits in the human entorhinal cortex. Acta Neuropathol. (Berl.) 100, 608–617 (2000).
Yamaguchi, H., Sugihara, S., Ogawa, A., Saido, T.C. & Ihara, Y. Diffuse plaques associated with astroglial amyloid β protein, possibly showing a disappearing stage of senile plaques. Acta Neuropathol. (Berl.) 95, 217–222 (1998).
Shaffer, L.M. et al. Amyloid β protein (A β) removal by neuroglial cells in culture. Neurobiol. Aging 16, 737–745 (1995).
Wyss-Coray, T. & Mucke, L. Inflammation in neurodegenerative disease—a double-edged sword. Neuron 35, 419–432 (2002).
DeWitt, D.A., Perry, G., Cohen, M., Doller, C. & Silver, J. Astrocytes regulate microglial phagocytosis of senile plaque cores of Alzheimer's disease. Exp. Neurol. 149, 329–340 (1998).
Bard, F. et al. Peripherally administered antibodies against amyloid β-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat. Med. 6, 916–919 (2000).
Savchenko, V.L., McKanna, J.A., Nikonenko, I.R. & Skibo, G.G. Microglia and astrocytes in the adult rat brain: comparative immunocytochemical analysis demonstrates the efficacy of lipocortin 1 immunoreactivity. Neuroscience 96, 195–203 (2000).
De Groot, C.J. et al. Establishment of human adult astrocyte cultures derived from postmortem multiple sclerosis and control brain and spinal cord regions: immunophenotypical and functional characterization. J. Neurosci. Res. 49, 342–354 (1997).
Coraci, I.S. et al. CD36, a class B scavenger receptor, is expressed on microglia in Alzheimer's disease brains and can mediate production of reactive oxygen species in response to β-amyloid fibrils. Am. J. Pathol. 160, 101–112 (2002).
Paresce, D.M., Ghosh, R.N. & Maxfield, F.R. Microglial cells internalize aggregates of the Alzheimer's disease amyloid β-protein via a scavenger receptor. Neuron 17, 553–565 (1996).
Wyss-Coray, T. et al. TGF-β1 promotes microglial amyloid-β clearance and reduces plaque burden in transgenic mice. Nat. Med. 7, 612–618 (2001).
Mucke, L. et al. High-level neuronal expression of aβ1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J. Neurosci. 20, 4050–4058 (2000).
Acknowledgements
This work was supported by National Institutes of Health grants AG-15871 (to T.W.-C.) and AG-19772 (to S.C.S.), the Alzheimer's Association (T.W.-C. and J.H.) and a Pilot Grant Award (to J.H.) from Columbia University's Alzheimer Disease Research Center (through National Institutes of Health grant AG-08702).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Wyss-Coray, T., Loike, J., Brionne, T. et al. Adult mouse astrocytes degrade amyloid-β in vitro and in situ. Nat Med 9, 453–457 (2003). https://doi.org/10.1038/nm838
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm838
This article is cited by
-
Physiological expression of mutated TAU impaired astrocyte activity and exacerbates β-amyloid pathology in 5xFAD mice
Journal of Neuroinflammation (2023)
-
BACE1 regulates expression of Clusterin in astrocytes for enhancing clearance of β-amyloid peptides
Molecular Neurodegeneration (2023)
-
Aß Pathology and Neuron–Glia Interactions: A Synaptocentric View
Neurochemical Research (2023)
-
FABP7 drives an inflammatory response in human astrocytes and is upregulated in Alzheimer’s disease
GeroScience (2023)
-
Nerve growth factor receptor (Ngfr) induces neurogenic plasticity by suppressing reactive astroglial Lcn2/Slc22a17 signaling in Alzheimer’s disease
npj Regenerative Medicine (2023)