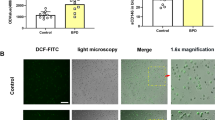
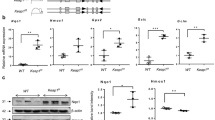
Abstract
Respiratory distress syndrome (RDS) due to insufficient production of surfactant is a common and severe complication of preterm delivery. Here, we report that loss of the hypoxia-inducible transcription factor-2α (HIF-2α) caused fatal RDS in neonatal mice due to insufficient surfactant production by alveolar type 2 cells. VEGF, a target of HIF-2α, regulates fetal lung maturation: because VEGF levels in alveolar cells were reduced in HIF-2α-deficient fetuses; mice with a deficiency of the VEGF164 and VEGF188 isoforms or of the HIF-binding site in the VEGF promotor died of RDS; intrauterine delivery of anti-VEGF-receptor-2 antibodies caused RDS and VEGF stimulated production of surfactant proteins by cultured type 2 pneumocytes. Intrauterine delivery or postnatal intratracheal instillation of VEGF stimulated conversion of glycogen to surfactant and protected preterm mice against RDS. The pneumotrophic effect of VEGF may have therapeutic potential for lung maturation in preterm infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Goldenberg, R.L., Hauth, J.C. & Andrews, W.W. Intrauterine infection and preterm delivery. N. Engl. J. Med. 342, 1500–1507 (2000).
Bhakoo, O.N., Narang, A., Karthikeyan, G. & Kumar, P. Spectrum of respiratory distress in very low birthweight neonates. Indian J. Pediatr. 67, 803–804 (2000).
Bourbon, J.R., Rieutort, M., Engle, M.J. & Farrell, P.M. Utilization of glycogen for phospholipid synthesis in fetal rat lung. Biochim. Biophys. Acta 712, 382–389 (1982).
Kennedy, J.D. Lung function outcome in children of premature birth. J. Paediatr. Child Health 35, 516–521 (1999).
Jakkula, M. et al. Inhibition of angiogenesis decreases alveolarization in the developing rat lung. Am. J. Physiol. Lung Cell. Mol. Physiol. 279, L600–607 (2000).
Ferrara, N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am. J. Physiol. Cell. Physiol. 280, C1358–1366 (2001).
Healy, A.M., Morgenthau, L., Zhu, X., Farber, H.W. & Cardoso, W.V. VEGF is deposited in the subepithelial matrix at the leading edge of branching airways and stimulates neovascularization in the murine embryonic lung. Dev. Dyn. 219, 341–352 (2000).
Klekamp, J.G., Jarzecka, K. & Perkett, E.A. Exposure to hyperoxia decreases the expression of vascular endothelial growth factor and its receptors in adult rat lungs. Am. J. Pathol. 154, 823–831 (1999).
Kaner, R.J. & Crystal, R.G. Compartmentalization of vascular endothelial growth factor to the epithelial surface of the human lung. Mol. Med. 7, 240–246 (2001).
Lassus, P. et al. Pulmonary vascular endothelial growth factor and Flt-1 in fetuses, in acute and chronic lung disease, and in persistent pulmonary hypertension of the newborn. Am. J. Respir. Crit. Care Med. 164, 1981–1987 (2001).
Bhatt, A.J. et al. Disrupted pulmonary vasculature and decreased vascular endothelial growth factor, Flt-1, and TIE-2 in human infants dying with bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 164, 1971–1980 (2001).
Lassus, P., Ristimaki, A., Ylikorkala, O., Viinikka, L. & Andersson, S. Vascular endothelial growth factor in human preterm lung. Am. J. Respir. Crit. Care Med. 159, 1429–1433 (1999).
D'Angio, C. et al. Vascular endothelial growth factor in pulmonary lavage fluid from premature infants: Effects of age and postnatal dexamethasone. Biol. Neonate 76, 266–273 (1999).
Brown, K.R., England, K.M., Goss, K.L., Snyder, J.M. & Acarregui, M.J. VEGF induces airway epithelial cell proliferation in human fetal lung in vitro. Am. J. Physiol. Lung Cell. Mol. Physiol. 281, L1001–1010 (2001).
Tian, H., McKnight, S.L. & Russell, D.W. Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev. 11, 72–82 (1997).
Ema, M. et al. A novel bHLH-PAS factor with close sequence similarity to hypoxia-inducible factor 1 α regulates VEGF expression and is potentially involved in lung and vascular development. Proc. Natl. Acad. Sci. USA 94, 4273–4278 (1997).
Flamme, I. et al. HRF, a putative basic helix-loop-helix-PAS-domain transcription factor is closely related to hypoxia-inducible factor-1α and developmentally expressed in blood vessels. Mech. Dev. 63, 51–63 (1997).
Brusselmans, K. et al. Hypoxia-inducible factor-2α (HIF-2α) is involved in the apoptotic response to hypoglycemia but not to hypoxia. J. Biol. Chem. 276, 39192–39196 (2001).
Botas, C. et al. Altered surfactant homeostasis and alveolar type II cell morphology in mice lacking surfactant protein D. Proc. Natl. Acad. Sci. USA 95, 11869–11874 (1998).
Jain, S., Maltepe, E., Lu, M.M., Simon, C. & Bradfield, C.A. Expression of ARNT, ARNT2, HIF-1α, HIF-2α, and Ah receptor mRNAs in the developing mouse. Mech. Dev. 73, 117–123 (1998).
Carmeliet, P. et al. Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF164 and VEGF188. Nature Med. 5, 495–502 (1999).
Stalmans, I. et al. Arteriolar and venular patterning in retinas of mice selectively expressing VEGF isoforms. J. Clin. Invest. 109, 327–336 (2002).
Carmeliet, P. et al. Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nature Med. 7, 575–583 (2001).
Oosthuyse, B. et al. Deletion of the hypoxia-response element in the vascular endothelial growth factor promoter causes motor neuron degeneration. Nature Genet. 28, 131–138 (2001).
Rannels, S.R. Impaired surfactant synthesis in fetal type II lung cells from gsd/gsd rats. Exp. Lung Res. 22, 213–29 (1996).
Rayani, H.H., Gewolb, I.H. & Floros, J. Glucose decreases steady state mRNA content of hydrophobic surfactant proteins B and C in fetal rat lung explants. Exp. Lung Res. 25, 69–79 (1999).
Gilden, C., Sevanian, A., Tierney, D.F., Kaplan, S.A. & Barrett, C.T. Regulation of fetal lung phosphatidyl choline synthesis by cortisol: role of glycogen and glucose. Pediatr. Res. 11, 845–848 (1977).
Ren, J.M., Gulve, E.A., Cartee, G.D. & Holloszy, J.O. Hypoxia causes glycogenolysis without an increase in percent phosphorylase a in rat skeletal muscle. Am. J. Physiol. 263, E1086–1091 (1992).
Semenza, G.L. HIF-1 and mechanisms of hypoxia sensing. Curr. Opin. Cell. Biol. 13, 167–171 (2001).
Yu, A.Y. et al. Temporal, spatial, and oxygen-regulated expression of hypoxia-inducible factor-1 in the lung. Am. J. Physiol. 275, L818–826 (1998).
Liang, Y. et al. Activation of vascular endothelial growth factor A transcription in tumorigenic glioblastoma cell lines by an enhancer with cell-type specific DNAse 1 accessibility. J. Biol. Chem. Mar 23 (2002) online publication: http://www.jbc.org/cgi/content/abstract/M201766200v1
Bernatchez, P.N., Winstead, M.V., Dennis, E.A. & Sirois, M.G. VEGF stimulation of endothelial cell PAF synthesis is mediated by group V 14 kDa secretory phospholipase A2. Br. J. Pharmacol. 134, 197–205 (2001).
Bourbon, J.R., Hoffman, D.R. & Johnston, J.M. Effect of platelet-activating factor on glycogen metabolism in fetal rat lung. Exp. Lung Res. 17, 789–801 (1991).
Rooney, S.A. Regulation of surfactant secretion. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 129, 233–243 (2001).
Pugazhenthi, S. & Khandelwal, R.L. Regulation of glycogen synthase activation in isolated hepatocytes. Mol. Cell. Biochem. 149–150, 95–101 (1995).
Tian, H., Hammer, R.E., Matsumoto, A.M., Russell, D.W. & McKnight, S.L. The hypoxia-responsive transcription factor EPAS1 is essential for catecholamine homeostasis and protection against heart failure during embryonic development. Genes Dev. 12, 3320–3324 (1998).
Peng, J., Zhang, L., Drysdale, L. & Fong, G.H. The transcription factor EPAS-1/hypoxia-inducible factor 2α plays an important role in vascular remodeling. Proc. Natl. Acad. Sci. USA 97, 8386–8391 (2000).
Ng, Y.S., Rohan, R., Sunday, M.E., Demello, D.E. & D'Amore, P.A. Differential expression of VEGF isoforms in mouse during development and in the adult. Dev. Dyn. 220, 112–121 (2001).
Corne, J. et al. IL-13 stimulates vascular endothelial cell growth factor and protects against hyperoxic acute lung injury. J. Clin. Invest. 106, 783–791 (2000).
Walfisch, A., Hallak, M. & Mazor, M. Multiple courses of antenatal steroids: risks and benefits. Obstet. Gynecol. 98, 491–497 (2001).
Bhatt, A.J., Amin, S.B., Chess, P.R., Watkins, R.H. & Maniscalco, W.M. Expression of vascular endothelial growth factor and Flk-1 in developing and glucocorticoid-treated mouse lung. Pediatr. Res. 47, 606–613 (2000).
Shweiki, D., Itin, A., Soffer, D. & Keshet, E. Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359, 843–845 (1992).
Hoet, P.H., Gilissen, L. & Nemery, B. Polyanions protect against the in vitro pulmonary toxicity of polycationic paint components associated with the Ardystil syndrome. Toxicol. Appl. Pharmacol. 175, 184–190 (2001).
Acknowledgements
We thank R. Verbesselt, K. Desmet, C. Van Geet and H. Devlieger for helpful discussion; and A. Bouché, M. De Mol, B. Hermans, S. Jansen, L. Kieckens, W.Y. Man, A. Manderveld, K. Maris, W. Martens, M. Nijs, S. Terclavers, A. Vandenhoeck, B. Vanwetswinkel, P. Van Wezemael and S. Wyns for technical assistance. This work was supported by an FWO-fellowship to V.C. and S.P. and by grants from the Research Fund K.U.Leuven (GOA/2001/09) and the BIOMED (#PL963380) to P.C. and D.C.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Compernolle, V., Brusselmans, K., Acker, T. et al. Loss of HIF-2α and inhibition of VEGF impair fetal lung maturation, whereas treatment with VEGF prevents fatal respiratory distress in premature mice. Nat Med 8, 702–710 (2002). https://doi.org/10.1038/nm721
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm721
This article is cited by
-
Pericytes contribute to pulmonary vascular remodeling via HIF2α signaling
EMBO Reports (2024)
-
The inhibition of protein translation promotes tumor angiogenic switch
Molecular Biomedicine (2022)
-
Appropriate dose of intravitreal ranibizumab for ROP: a retrospective study
BMC Ophthalmology (2022)
-
Molecular regulation of lung maturation in near-term fetal sheep by maternal daily vitamin C treatment in late gestation
Pediatric Research (2022)
-
VEGFR1-tyrosine kinase signaling in pulmonary fibrosis
Inflammation and Regeneration (2021)