Abstract
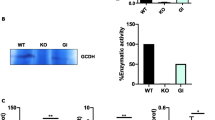
Aspartylglycosaminuria (AGU), the most common disorder of glycoprotein degradation in humans, is caused by mutations in the gene encoding the lysosomal enzyme glycosylasparaginase (Aga)1. The resulting enzyme deficiency allows aspartylglucosamine (GlcNAc–Asn) and other glycoasparagines to accumulate in tissues and body fluids, from early fetal life onward1. The clinical course is characterized by normal early development, slowly progressing to severe mental and motor retardation in early adulthood2,3. The exact pathogenesis of AGU in humans is unknown and neither therapy nor an animal model for this debilitating and ultimately fatal disease exists. Through targeted disruption of the mouse Aga gene in embryonic stem cells, we generated mice that completely lack Aga activity. At the age of 5–10 months a massive accumulation of GlcNAc–Asn was detected along with lysosomal vacuolization, axonal swelling in the gracile nucleus and impaired neuromotor coordination. A significant number of older male mice had massively swollen bladders, which was not caused by obstruction, but most likely related to the impaired function of the nervous system. These findings are consistent with the pathogenesis of AGU and provide further data explaining the impaired neurological function in AGU patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mononen, I., Fisher, K.J., Kaartinen, V. & Aronson, N.N., Jr. Aspartylglycosaminuria: Protein chemistry and molecular biology of the most common lysosomal storage disorder of glycoprotein degradation. FASEB. J. 7, 1247–1256 (1993).
Aula, P., Autio, S., Raivio, K.O. & Rapola, J., Aspartylglucosaminuria. in Genetic Errors of Glycoprotein Metabolism (eds. Durand, P. & O'Brien, J.S.) 123–152 (Springer, Heidelberg, 1982).
Thomas, G.H. & Beaudet, A.L. Disorders of glycoprotein degradation and structure: α-Mannosidosis, sialidosis, aspartylglucosaminuria, and carbohydrate-deficient glycoprotein syndrome. in The Metabolic and Molecular Bases of Inherited Disease (eds. Scriver, C.R., Beaudet, A.L., Sly, W.S. & Valle, D.) 2545–2548 (McGraw-Hill, New York, 1995).
Tenhunen, K. et al. Molecular cloning, chromosomal assignment, and expression of the mouse aspartylglucosaminidase gene. Genomics 30, 244–250 (1995).
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J.C. Derivatization of completely cell-culture-derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 90, 8424–8428 (1993).
Fisher, K.J. & Aronson, N.N., Jr. Characterization of the mutation responsible for aspartylglucosaminuria in three Finnish patients. J. Biol. Chem. 266, 12105–12113 (1991).
Kaartinen, V. & Mononen, I. Analysis of aspartylglucosamine at the picomole level by high-performance liquid chromatography. J. Chromatogr. 490, 292–299 (1989).
Haltia, M., Palo, J. & Autio, S. Aspartylglucosaminuria: A generalized storage disease. Acta Neuropathol. 31, 243–255 (1975).
Arstila, A.U., Palo, J., Haltia, M., Riekkinen, P. & Autio, S., Aspartylglucosaminuria, I. Fine structural studies on liver, kidney and brain. Acta Neuropathol. 20, 207–216 (1972).
Otterbach, B. & Stoffel, W. Acid sphingomyelinase-deficient mice mimic the neurovisceral form of human lysosomal storage disease (Nieman-Pick disease). Cell 81, 1053–1061 (1995).
Cases, O. et al. Aggressive behavior and altered amounts of brain serotonin and norepinephrine in mice lacking MAOA. Science 268, 1763–1766 (1995).
Guastavino, J-M., Larsson, K. & Jaisson, P. Neurological murine mutants as models for single-gene effects on behavior. in Techniques for the Genetic Analysis of Brain and Behavior: Focus on the Mouse, (eds. Goldowitz, G., Wahlsten, D. & Wimer, R.E.) 375–390 (Elsevier, Amsterdam, 1992).
Yamazaki, K. et al. Gracile axonal dystrophy (GAD), a new neurological mutant in the mouse. Proc. Soc. Exp. Biol. Med. 187, 209–215 (1988).
Indravasu, S. & Dexter, R.A. Infantile neuroaxonal dystrophy and its relationship to Hallervorden-Spatz disease. Neurology 18, 693–699 (1968).
Oynagi, K. et al. Galactosialidosis: Neuropathological findings in a case of the late-infantile type. Acta Neuropathol. 85, 331–339 (1991).
Mononen, I. et al. Recombinant glycosylasparaginase and in vitro correction of aspartylglycosaminuria. FASEB J. 9, 428–433 (1995).
Enomaa, N., Danos, O., Peltonen, L. & Jalanko, A. Correction of deficient enzyme activity in a lysosomal storage disease, aspartylglucosaminuria by enzyme replacement and retroviral gene transfer. Hum. Gene Ther. 6, 723–731 (1995).
Wurst, W. & Joyner, A. Production of targeted embryonic stem cell clones. in Gene Targeting: A Practical Approach (ed. Joyner, A.L.) 31–62 (Oxford Univ. Press, Oxford, 1993).
Papaioannou, V.P. & Johnson, R. Production of chimeras and genetically defined offspring from targeted ES cells in Gene Targeting: A Practical Approach (ed. Joyner, A.L.) 107–146 (Oxford Univ. Press, Oxford, 1993).
Kaartinen, V. & Mononen, I. Assay of aspartylglucosylaminase by highperformance liquid chromatography. Anal. Biochem. 190, 98–101 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaartinen, V., Mononen, I., Voncken, J. et al. A mouse model for the human lysosomal disease aspartylglycosaminuria. Nat Med 2, 1375–1378 (1996). https://doi.org/10.1038/nm1296-1375
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1296-1375
This article is cited by
-
Tiered analysis of whole-exome sequencing for epilepsy diagnosis
Molecular Genetics and Genomics (2020)
-
Aspartylglycosaminuria: a review
Orphanet Journal of Rare Diseases (2016)
-
Identification of Small Molecule Compounds for Pharmacological Chaperone Therapy of Aspartylglucosaminuria
Scientific Reports (2016)
-
Early initiation of enzyme replacement therapy improves metabolic correction in the brain tissue of aspartylglycosaminuria mice
Journal of Inherited Metabolic Disease (2010)
-
The Finnish disease heritage III: the individual diseases
Human Genetics (2003)