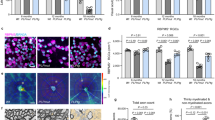
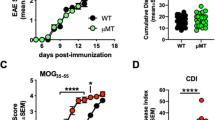
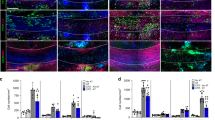
Abstract
Ever since the use of interferon-γ to treat patients with multiple sclerosis resulted in enhanced disease, the role of IFN-γ in demyelination has been under question. To address this issue directly, transgenic mice were generated that expressed the cDNA of murine IFN-γ in the central nervous system by using an oligodendrocyte-specific promoter. Expression of the transgene occurred after 8 weeks of age, at which time the murine immune and central nervous systems are both fully developed. Directly associated with transgene expression, primary demyelination occurred and was accompanied by clinical abnormalities consistent with CNS disorders. Additionally, multiple hallmarks of immune-mediated CNS disease were observed including upregulation of MHC molecules, gliosis and lymphocytic Infiltration. These results demonstrate a direct role for IFN-γ as an inducer of CNS demyelination leading to disease and provide new opportunities for dissecting the mechanism of demyelination.
Similar content being viewed by others
Article PDF
References
Cannella, B. & Raine, C.S. . The adhesion molecule and cytokine profile of multiple sclerosis lesions. Ann. Neural. 37, 424–435 (1995).
Renno, T., Lin, J.Y., Piccirillo, C., Antel, J. & Owens, T. Cytokine production by Cells in cerebrospinal fluid during experimental allergic encephalomyelitis in SJL/J mice. J. Neuroimmunol. 49, 1–7 (1994).
Pearce, B.D., Hobbs, M.V., McGraw, T.S. & Buchmeier, M.J. Cytokine induction during T-Cell-mediated clearance of mouse hepatitis virus from neurons in vivo . J. Virol. 68, 5483–5495 (1994).
Rodriguez, M., Pierce, M.L. & Howie, E. Immune response gene products (la antigens) on glial and endothelial Cells in virus-induced demyelination. J. Immunol. 138, 3438–3442 (1987).
Traugott, U. & Lebon, P. Interferon-gamma and la antigen are present on astrocytes in active chronic multiple sclerosis lesions. J. Neurol. Sci. 84, 257–264 (1988).
Rubio, N. & Torres, C. IL-1, IL-2 and IFN-gamma production by Theiler's virus-induced encephalomyelitic SJL/J mice. Immunology 74, 284–289 (1991).
Woodroofe, M.N. & Cuzner, M.L. Cytokine mRNA expression in inflammatory multiple sclerosis lesions: Detection by non-radioactive in situ hybridization. Cytokine 5, 583–588 (1993).
Sobel, R.A. The pathology of multiple sclerosis. Neurol. Clin. 13, 1–21 (1995).
Hauser, S.L. et al. Immunohistochemical analysis of the Cellular infiltrate in multiple sclerosis lesions. Ann. Neural. 19, 578–587 (1986).
Lassmann, H., Suchanek, G. & Ozawa, K. Histopathology and the bloodcerebrospinal fluid barrier in multiple sclerosis. Ann. Neural. 36, S42–S46 (1994).
Benveniste, E.N. & Benos, D.J., TNF-alpha - and IFN-gamma-mediated signal transduction pathways: Effects on glial Cell gene expression and function. FASEB J. 9, 1577–1584 (1995).
Sethna, M.P. & Lampson, L.A. Immune modulation within the brain: Recruitment of inflammatory Cells and increased major histocompatibility antigen expression following intracerebral injection of interferon-gamma. J. Neuroimmunol. 34, 121–132 (1991).
Lublin, F.D. et al. Monoclonal anti-gamma interferon antibodies enhance experimental allergic encephalomyelitis. Autoimmunity 16, 267–274 (1993).
Rodriguez, M., Pavelko, K. & Coffman, R. Gamma interferon is critical for resistance to Theiler's virus-induced demyelination. J. Virol. 69, 7286–7290 (1995).
Shalaby, M.R., Hamilton, E.B., Benninger, A.H. & Marafino, B.J.J. In vivo antiviral activity of recombinant murine gamma interferon. J. Interferon Res. 5, 339–345 (1985).
Young, H.A. & Hardy, K.J. Role of interferon-gamma in immune Cell regulation. J. Leukocyte Biol. 58, 373–381 (1995).
Linthicum, D.S., Munoz, J.J. & Blaskett, A. Acute experimental autoimmune en- cephalomyelitis in mice. I. Adjuvant action of Bordetella pertussis is due to vasoactive amine sensitization and increased vascular permeability of the central nervous system. Cell. Immunol. 73, 299–310 (1982).
Paterson, P.Y. Transfer of allergoencephalomyelitis in rats by means of lymph node Cells J. Exp. Med. 111, 119–133 (1960).
Sarvetnick, N. et al. Loss of pancreatic islet tolerance induced by beta-Cell expression of interferon-gamma. Nature 346, 844–847 (1990).
Hanafusa, T. et al. The NOD mouse. Diabetes Res. Clin. Pract. 24 (Suppl.), S307–S311 (1994).
Gow, A., Friedrich, V.L. & Lazzarini, R.A. Myelin basic protein gene contains separate enhancers for oligodendrocyte and Schwann Cell expression. J. Cell Biol. 119, 605–616 (1992).
Evans, C.F., Horwitz, M.S., Hobbs, M.V. & Oldstone, M.B.A., Viral infection of transgenic mice expressing a viral protein in oligodendrocytes leads to chronic central nervous system autoimmune disease J. Exp. Med. 184, 2371–2384 (1996).
Corbin, J.G. et al. Targetted CNS expression of interferon-gamma in transgenic mice leads to hypomyelination, reactive gliosis, and abnormal cerebellar development. Mol. Cell. Neurosci. 7, 354–370 (1996).
Turnley, A.M. et al. Dysmyelination in transgenic mice resulting from expression of class I histocompatibility molecules in oligodendrocytes. Nature 353, 566–569 (1991).
Neumann, H., Cavalie, A., Jenne, D.E. & Wekerle, H. Induction of MHC class I genes in neurons. Science 269, 549–552 (1995).
Joly, E., Mucke, L. & Oldstone, M.B. Viral persistence in neurons explained by lack of major histocompatibility class I expression. Science 253, 1283–1285 (1991).
Bergsteindottir, K., Brennan, A., Jessen, K.R. & Mirsky, R., In the presence of dexamethasone, gamma interferon induces rat oligodendrocytes to express major histocompatibility complex class II molecules. Proc. Natl. Acad. Sci. USA 89, 9054–9058 (1992).
Massa, P.T., Ozato, K. & McFarlin, D.E. Cell type-specific regulation of major histocompatibility complex (MHC) class I gene expression in astrocytes, oligodendrocytes, and neurons. Glia 8, 201–207 (1993).
Suzumura, A. et al. Induction of glial Cell MHC antigen expression in neurotropic coronavirus infections: Characterization of the H-2-inducing soluble factor elaborated by infected brain Cells J. Immunol. 140, 2068–2072 (1988).
Vartanian, T., Li, Y., Zhao, M. & Stefansson, K. Interferon-gamma-induced oligodendrocyte Cell death: Implications for the pathogenesis of multiple sclerosis. Mol. Med. 1, 732–743 (1995).
Merrill, J.E., Ignarro, L.J., Sherman, M.P., Melinek, J. & Lane, T.E. Microglial cell cytotoxicity of oligodendrocytes is mediated through nitric oxide. J. Immunol. 151, 2132–2141 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Horwitz, M., Evans, C., Mcgavern, D. et al. Primary demyelination in transgenic mice expressing interferon-γ. Nat Med 3, 1037–1041 (1997). https://doi.org/10.1038/nm0997-1037
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0997-1037
This article is cited by
-
Therapeutic Potential of Cytokines in Demyelinating Lesions After Stroke
Journal of Molecular Neuroscience (2021)
-
Matrix metalloproteinases in the CNS: interferons get nervous
Cellular and Molecular Life Sciences (2019)
-
Interferon-γ induces progressive nigrostriatal degeneration and basal ganglia calcification
Nature Neuroscience (2011)
-
Transgenic mouse models of multiple sclerosis
Cellular and Molecular Life Sciences (2010)
-
CB2 cannabinoid receptors as an emerging target for demyelinating diseases: from neuroimmune interactions to cell replacement strategies
British Journal of Pharmacology (2008)