Abstract
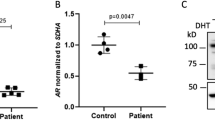
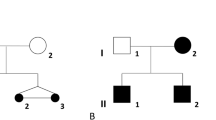
Despite the fact that genitourinary defects are among the most common birth defects in newborns, little is known about their etiology. Here we analyzed children born with congenital genitourinary tract masculinization disorders by array-comparative genomic hybridization, which revealed in 1.35% of cases the presence of de novo copy number gains at Xq28 encompassing the VAMP7 gene, which encodes a vesicle-trafficking protein that is part of the SNARE complex. Transgenic mice carrying a bacterial artificial chromosome encoding human VAMP7 mimicked the defective urogenital traits observed in boys with masculinization disorders such as cryptorchidism, urethral defects and hypospadias. Transgenic mice also exhibited reduced penile length, focal spermatogenic anomalies, diminished sperm motility and subfertility. VAMP7 colocalized with estrogen receptor α (ESR1) in the presence of its cognate ligand, 17β-estradiol. Elevated levels of VAMP7 markedly intensified ESR1-potentiated transcriptional activity by increasing ESR1 protein cellular content upon ligand stimulation and upregulated the expression of estrogen-responsive genes including ATF3, CYR61 and CTGF, all of which have been implicated in human hypospadias. Hence, increased gene dosage of VAMP7, and thus higher expression levels of its protein product, enhances estrogen receptor action in male genitourinary tissues, affects the virilization of the reproductive tract and results in genitourinary birth defects in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Referenced accessions
Gene Expression Omnibus
NCBI Reference Sequence
References
Sharpe, R.M. & Skakkebaek, N.E. Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract? Lancet 341, 1392–1395 (1993).
Wilhelm, D. & Koopman, P. The makings of maleness: towards an integrated view of male sexual development. Nat. Rev. Genet. 7, 620–631 (2006).
Vidaeff, A.C. & Sever, L.E. In utero exposure to environmental estrogens and male reproductive health: a systematic review of biological and epidemiologic evidence. Reprod. Toxicol. 20, 5–20 (2005).
Storgaard, L., Bonde, J.P. & Olsen, J. Male reproductive disorders in humans and prenatal indicators of estrogen exposure. A review of published epidemiological studies. Reprod. Toxicol. 21, 4–15 (2006).
McLachlan, J.A., Newbold, R.R. & Bullock, B. Reproductive tract lesions in male mice exposed prenatally to diethylstilbestrol. Science 190, 991–992 (1975).
Goyal, H.O. et al. Estrogen receptor α mediates estrogen-inducible abnormalities in the developing penis. Reproduction 133, 1057–1067 (2007).
Goyal, H.O., Braden, T.D., Williams, C.S. & Williams, J.W. Estrogen-induced developmental disorders of the rat penis involve both estrogen receptor (ESR)- and androgen receptor (AR)-mediated pathways. Biol. Reprod. 81, 507–516 (2009).
Simon, L. et al. Exposure of neonatal rats to anti-androgens induces penile mal-developments and infertility comparable to those induced by oestrogens. Int. J. Androl. 35, 364–376 (2012).
Li, X. et al. Altered structure and function of reproductive organs in transgenic male mice overexpressing human aromatase. Endocrinology 142, 2435–2442 (2001).
Kloepper, T.H., Kienle, C.N. & Fasshauer, D. An elaborate classification of SNARE proteins sheds light on the conservation of the eukaryotic endomembrane system. Mol. Biol. Cell 18, 3463–3471 (2007).
Pryor, P.R. et al. Molecular basis for the sorting of the SNARE VAMP7 into endocytic clathrin-coated vesicles by the ArfGAP Hrb. Cell 134, 817–827 (2008).
Pryor, P.R. et al. Combinatorial SNARE complexes with VAMP7 or VAMP8 define different late endocytic fusion events. EMBO Rep. 5, 590–595 (2004).
Advani, R.J. et al. VAMP-7 mediates vesicular transport from endosomes to lysosomes. J. Cell Biol. 146, 765–776 (1999).
Rao, S.K., Huynh, C., Proux-Gillardeaux, V., Galli, T. & Andrews, N.W. Identification of SNAREs involved in synaptotagmin VII–regulated lysosomal exocytosis. J. Biol. Chem. 279, 20471–20479 (2004).
Fader, C.M., Sanchez, D.G., Mestre, M.B. & Colombo, M.I. TI-VAMP/VAMP7 and VAMP3/cellubrevin: two v-SNARE proteins involved in specific steps of the autophagy/multivesicular body pathways. Biochim. Biophys. Acta 1793, 1901–1916 (2009).
Moreau, K., Ravikumar, B., Renna, M., Puri, C. & Rubinsztein, D.C. Autophagosome precursor maturation requires homotypic fusion. Cell 146, 303–317 (2011).
Tannour-Louet, M. et al. Identification of de novo copy number variants associated with human disorders of sexual development. PLoS ONE 5, e15392 (2010).
Shono, T. et al. Scanning electron microscopy shows inhibited gubernacular development in relation to undescended testes in oestrogen-treated mice. Int. J. Androl. 19, 263–270 (1996).
Nef, S., Shipman, T. & Parada, L.F. A molecular basis for estrogen-induced cryptorchidism. Dev. Biol. 224, 354–361 (2000).
Jiang, X.W., Li, J.H., Huang, T.H. & Deng, W.D. Effect of prenatal exposure to diethylstilbestrol on gubernacular development in fetal male mice. Asian J. Androl. 6, 325–329 (2004).
Cederroth, C.R. et al. Estrogen receptor α is a major contributor to estrogen-mediated fetal testis dysgenesis and cryptorchidism. Endocrinology 148, 5507–5519 (2007).
Kent, H.M. et al. Structural basis of the intracellular sorting of the SNARE VAMP7 by the AP3 adaptor complex. Dev. Cell 22, 979–988 (2012).
Magee, J.A., Chang, L.W., Stormo, G.D. & Milbrandt, J. Direct, androgen receptor–mediated regulation of the FKBP5 gene via a distal enhancer element. Endocrinology 147, 590–598 (2006).
Makkonen, H., Kauhanen, M., Paakinaho, V., Jaaskelainen, T. & Palvimo, J.J. Long-range activation of FKBP51 transcription by the androgen receptor via distal intronic enhancers. Nucleic Acids Res. 37, 4135–4148 (2009).
Chen, H. et al. Fkbp52 regulates androgen receptor transactivation activity and male urethra morphogenesis. J. Biol. Chem. 285, 27776–27784 (2010).
Nishida, H. et al. Gene expression analyses on embryonic external genitalia: identification of regulatory genes possibly involved in masculinization processes. Congenit. Anom. (Kyoto) 48, 63–67 (2008).
Miyagawa, S. et al. The role of sonic hedgehog–Gli2 pathway in the masculinization of external genitalia. Endocrinology 152, 2894–2903 (2011).
Rivas, A., Fisher, J.S., McKinnell, C., Atanassova, N. & Sharpe, R.M. Induction of reproductive tract developmental abnormalities in the male rat by lowering androgen production or action in combination with a low dose of diethylstilbestrol: evidence for importance of the androgen-estrogen balance. Endocrinology 143, 4797–4808 (2002).
Wang, Z. et al. Up-regulation of estrogen responsive genes in hypospadias: microarray analysis. J. Urol. 177, 1939–1946 (2007).
Liu, B. et al. Estradiol upregulates activating transcription factor 3, a candidate gene in the etiology of hypospadias. Pediatr. Dev. Pathol. 10, 446–454 (2007).
Liu, B. et al. Activating transcription factor 3 is up-regulated in patients with hypospadias. Pediatr. Res. 58, 1280–1283 (2005).
Liu, B., Agras, K., Willingham, E., Vilela, M.L. & Baskin, L.S. Activating transcription factor 3 is estrogen-responsive in utero and upregulated during sexual differentiation. Horm. Res. 65, 217–222 (2006).
Gurbuz, C. et al. Is activating transcription factor 3 up-regulated in patients with hypospadias? Korean J. Urol. 51, 561–564 (2010).
Kalfa, N. et al. Genomic variants of ATF3 in patients with hypospadias. J. Urol. 180, 2183–2188, discussion 2188 (2008).
Beleza-Meireles, A. et al. Activating transcription factor 3: a hormone responsive gene in the etiology of hypospadias. Eur. J. Endocrinol. 158, 729–739 (2008).
Thenot, S., Charpin, M., Bonnet, S. & Cavailles, V. Estrogen receptor cofactors expression in breast and endometrial human cancer cells. Mol. Cell. Endocrinol. 156, 85–93 (1999).
Lambard, S. et al. Aromatase in testis: expression and role in male reproduction. J. Steroid Biochem. Mol. Biol. 95, 63–69 (2005).
Song, W.C., Qian, Y., Sun, X. & Negishi, M. Cellular localization and regulation of expression of testicular estrogen sulfotransferase. Endocrinology 138, 5006–5012 (1997).
Hazary, S. & Gardner, W.U. Influence of sex hormones on abdominal musculature and the formation of inguinal and scrotal hernias in mice. Anat. Rec. 136, 437–443 (1960).
Reinert, R.B. et al. Tamoxifen-induced Cre-loxP recombination is prolonged in pancreatic islets of adult mice. PLoS ONE 7, e33529 (2012).
Li, X. & Rahman, N. Impact of androgen/estrogen ratio: lessons learned from the aromatase over-expression mice. Gen. Comp. Endocrinol. 159, 1–9 (2008).
Wang, H. et al. The stress response mediator ATF3 represses androgen signaling by binding the androgen receptor. Mol. Cell. Biol. 32, 3190–3202 (2012).
Girirajan, S., Campbell, C.D. & Eichler, E.E. Human copy number variation and complex genetic disease. Annu. Rev. Genet. 45, 203–226 (2011).
Ferlin, A. et al. Genetic alterations associated with cryptorchidism. J. Am. Med. Assoc. 300, 2271–2276 (2008).
Hadziselimovic, F. et al. EGR4 is a master gene responsible for fertility in cryptorchidism. Sex Dev. 3, 253–263 (2009).
Hadziselimovic, F., Hadziselimovic, N.O., Demougin, P. & Oakeley, E.J. Testicular gene expression in cryptorchid boys at risk of azoospermia. Sex Dev. 5, 49–59 (2011).
Mills, I.G. et al. Huntingtin interacting protein 1 modulates the transcriptional activity of nuclear hormone receptors. J. Cell Biol. 170, 191–200 (2005).
Ray, M.R. et al. Cyclin G–associated kinase: a novel androgen receptor–interacting transcriptional coactivator that is overexpressed in hormone refractory prostate cancer. Int. J. Cancer 118, 1108–1119 (2006).
Jasavala, R. et al. Identification of putative androgen receptor interaction protein modules: cytoskeleton and endosomes modulate androgen receptor signaling in prostate cancer cells. Mol. Cell. Proteomics 6, 252–271 (2007).
Kang, J. et al. A nuclear function of β-arrestin1 in GPCR signaling: regulation of histone acetylation and gene transcription. Cell 123, 833–847 (2005).
Yang, L. et al. APPL suppresses androgen receptor transactivation via potentiating Akt activity. J. Biol. Chem. 278, 16820–16827 (2003).
Enari, M., Ohmori, K., Kitabayashi, I. & Taya, Y. Requirement of clathrin heavy chain for p53-mediated transcription. Genes Dev. 20, 1087–1099 (2006).
Wei, X., Xu, H. & Kufe, D. MUC1 oncoprotein stabilizes and activates estrogen receptor α. Mol. Cell 21, 295–305 (2006).
Reid, G. et al. Cyclic, proteasome-mediated turnover of unliganded and liganded ERα on responsive promoters is an integral feature of estrogen signaling. Mol. Cell 11, 695–707 (2003).
Lonard, D.M., Nawaz, Z., Smith, C.L. & O'Malley, B.W. The 26S proteasome is required for estrogen receptor-α and coactivator turnover and for efficient estrogen receptor-α transactivation. Mol. Cell 5, 939–948 (2000).
Nawaz, Z., Lonard, D.M., Dennis, A.P., Smith, C.L. & O'Malley, B.W. Proteasome-dependent degradation of the human estrogen receptor. Proc. Natl. Acad. Sci. USA 96, 1858–1862 (1999).
Korolchuk, V.I., Menzies, F.M. & Rubinsztein, D.C. Mechanisms of cross-talk between the ubiquitin-proteasome and autophagy-lysosome systems. FEBS Lett. 584, 1393–1398 (2010).
Korolchuk, V.I., Mansilla, A., Menzies, F.M. & Rubinsztein, D.C. Autophagy inhibition compromises degradation of ubiquitin-proteasome pathway substrates. Mol. Cell 33, 517–527 (2009).
Ou, Z. et al. Branchiootorenal syndrome and oculoauriculovertebral spectrum features associated with duplication of SIX1, SIX6, and OTX2 resulting from a complex chromosomal rearrangement. Am. J. Med. Genet. A 146A, 2480–2489 (2008).
Ou, Z. et al. Bacterial artificial chromosome-emulation oligonucleotide arrays for targeted clinical array-comparative genomic hybridization analyses. Genet. Med. 10, 278–289 (2008).
Acknowledgements
We would like to thank all the referring physicians and especially E.T. Gonzales, L. Cisek, N. Janzen, D.R. Roth, and E. Jones, the affected individuals and their family members who participated in this study. We thank M. Marcelli (Baylor College of Medicine) for providing the foreskin fibroblasts for analysis. We thank L. White, C. Shaw and F. De Mayo for their assistance. This study was supported in part by US National Institutes of Health grant K12 DK0083014 (KURe to D.J.L.) and R01DK078121 from the National Institute of Diabetes and Digestive and Kidney Diseases (to D.J.L.).
Author information
Authors and Affiliations
Contributions
Both senior authors, M.T.-L. and D.J.L., conceived and supervised the study, conducted experiments, analyzed data and wrote and revised the manuscript. S.H. and J.-F.L. performed experiments and analyzed data. B.Z., K.R., J.A. and A.S. performed experiments. S.W.C. conducted the human CGH array studies.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Table 1 (PDF 17649 kb)
Rights and permissions
About this article
Cite this article
Tannour-Louet, M., Han, S., Louet, JF. et al. Increased gene copy number of VAMP7 disrupts human male urogenital development through altered estrogen action. Nat Med 20, 715–724 (2014). https://doi.org/10.1038/nm.3580
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3580
This article is cited by
-
Male infertility
Nature Reviews Disease Primers (2023)
-
Aphallie – zwei Fallbeispiele
Der Urologe (2020)
-
Regulation of masculinization: androgen signalling for external genitalia development
Nature Reviews Urology (2018)
-
Genomic copy number variation association study in Caucasian patients with nonsyndromic cryptorchidism
BMC Urology (2016)
-
Oestrogen action and male fertility: experimental and clinical findings
Cellular and Molecular Life Sciences (2015)