Abstract
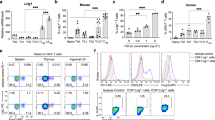
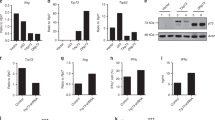
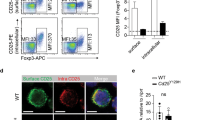
We investigated the in vivo function of the B7 family member B7-H3 (also known as B7RP-2) by gene targeting. B7-H3 inhibited T cell proliferation mediated by antibody to T cell receptor or allogeneic antigen-presenting cells. B7-H3-deficient mice developed more severe airway inflammation than did wild-type mice in conditions in which T helper cells differentiated toward type 1 (TH1) rather than type 2 (TH2). B7-H3 expression was consistently enhanced by interferon-γ but suppressed by interleukin 4 in dendritic cells. B7-H3-deficient mice developed experimental autoimmune encephalomyelitis several days earlier than their wild-type littermates, and accumulated higher concentrations of autoantibodies to DNA. Thus, B7-H3 is a negative regulator that preferentially affects TH1 responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lenschow, D.J., Walunas, T.L. & Bluestone, J.A. CD28/B7 system of T cell costimulation. Annu. Rev. Immunol. 14, 233–258 (1996).
Watts, T.H. & DeBenedette, M.A. T cell co-stimulation molecules other that CD28. Curr. Opin. Immunol. 11, 286–293 (1999).
Carreno, B. & Collins, M. The B7 family of ligands and its receptors: New pathways for costimulation and inhibition of immune responses. Annu. Rev. Immunol. 20, 29–53 (2002).
Sharpe, A.H. & Freeman, G.J. The B7-CD28 superfamily. Nat. Rev. Immunol. 2, 116–126 (2002).
Coyle, A.J. & Gutierrez-Ramos, J.-C. The expanding B7 superfamily: Increasing complexity in costimulatory signals regulating T cell function. Nat. Immunol. 2, 203–209 (2001).
Liang, L. & Sha, W.C. The right place at the right time: novel B7 family members regulate effector T cell responses. Curr. Opin. Immunol. 14, 384–390 (2002).
Lanzavecchia, A., Lezzi, G. & Viola, A. From TCR engagement to T cell activation: a kinetic view of T cell behavior. Cell 96, 1–4 (1999).
Murphy, K.M. & Reiner, S.L. Decision making in the immune system: The lineage decisions of helper T cells. Nat. Rev. Immunol. 2, 933–944 (2002).
Brunner, M., Chambers, C. & Allison, J. CTLA-4 mediates inhibition of early events of T cell proliferation. J. Immunol. 162, 5813–5820 (1999).
Waterhouse, P. et al. Lymphoproliferative disorders with early lethality in mice deficient in CTLA-4. Science 270, 985–988 (1995).
Tivol, E. et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan destruction, revealing a critical negative regulatory role of CTLA-4. Immunity 3, 541–547 (1995).
Mandelbrot, D.A., McAdam, A.J. & Sharpe, A.H. B7-1 or B7-2 is required to produce the lymphoproliferative phenotype in mice lacking cytotoxic T lymphocyte-associated antigen 4 (CTLA-4). J. Exp. Med. 189, 435–440 (1999).
Freeman, G.J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000).
Dong, H., Zhu, G., Tamada, K. & Chen, L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat. Med. 5, 1365–1369 (1999).
Latchman, Y. et al. PD-L2, a novel B7 homologue, is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2, 261–268 (2001).
Tseng, S.Y. et al. B7-DC, a new dendritic cell molecule with potent costimulatory properties for T cells. J. Exp. Med. 193, 839–846 (2001).
Carter, L.L. et al. PD-1:PD-L inhibitory pathway affects both CD4+ and CD8+ T cells and is overcome by IL-2. Eur. J. Immunol. 32, 634–643 (2002).
Mazanet, M.M. & Hughes, C.C.W. B7-H1 is expressed by human endothelial cells and suppresses T cell cytokine synthesis. J. Immunol. 169, 3581–3588 (2002).
Nishimura, H., Nose, M., Hiai, H., Minato, N. & Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 11, 141–151 (1999).
Nishimura, H. et al. Autoimmune dilated cardiomyopathy in PD-1 receptor deficient mice. Science 291, 319–322 (2001).
Yoshinaga, S.K. et al. T-cell co-stimulation through B7RP-1 and ICOS. Nature 402, 827–832 (1999).
Swallow, M.M., Wallin, J.J. & Sha, W.C. B7h, a novel costimultory homolog of B7.1 and B7.2, is induced by TNF-α. Immunity 11, 423–432 (1999).
Wang, S. et al. Costimulation of T cells by B7-H2, a B7-like molecule that binds ICOS. Blood 96, 2808–2813 (2000).
Ling, V. et al. Cutting edge: identification of GL50, a novel B7-like protein that functionally binds to ICOS receptor. J. Immunol. 164, 1653–1657 (2000).
Brodie, D. et al. LICOS, a primordial costimulatory ligand? Curr. Biol. 10, 333–336 (2000).
Hutloff, A. et al. ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature 397, 263–266 (1999).
Coyle, A.J. et al. The CD28-related molecule ICOS is required for effective T cell-dependent immune responses. Immunity 13, 95–105 (2000).
Tafuri, A. et al. ICOS is essential for effective T helper cell responses. Nature 409, 105–109 (2001).
McAdam, A. et al. ICOS is critical for CD40 mediated antibody class switching. Nature 409, 102–105 (2001).
Dong, C. et al. ICOS co-stimulatory receptor is essential for T cell activation and function. Nature 409, 97–101 (2001).
Watanabe, N. et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat. Immunol. 4, 670–679 (2003).
Sica, G.L. et al. B7-H4, a molecule of the B7 family, negatively regulates T cell immunity. Immunity 18, 849–861 (2003).
Prasad, D.V.R., Richards, S., Mai, X.M., & Dong, C. B7S1, a novel B7 family member that negatively regulates T cell activation. Immunity 18, 863–873 (2003).
Chapoval, A.I. et al. B7-H3: A costimulatory molecule for T cell activation and IFN-γ production. Nat. Immunol. 2, 269–274 (2001).
Sun, M. et al. Characterization of mouse and human B7-H3 genes. J. Immunol. 168, 6294–6297 (2002).
Stämpfli, M.R. et al. Regulation of allergic mucosal sensitization by interleukin-12 gene transfer to the airway. Am. J. Respir. Cell Mol. Biol. 21, 317–326 (1999).
Stämpfli, M.R. et al. GM-CSF transgene expression in the airway allows aerosolized ovalbumin to induce allergic sensitization in mice. J. Clin. Invest. 102, 1704–1714 (1998).
Wiley, R.E. et al. Expression of the TH1 chemokine IFN-γ-inducible protein 10 in the airway alters mucosal allergic sensitization in mice. J. Immunol. 166, 2750–2759 (2001).
Fung-Leung, W.-P., Kundig, T.M., Zinkernagel, R.M. & Mak, T.W. Immune response against lymphocytic choriomeningitis virus infection in mice without CD8 expression. J. Exp. Med. 174, 1425–1429 (1991).
Segal, B.M., Dwyer, B.K. & Shevach, E.M. An interleukin (IL)-10/IL-12 immunoregulatory circuit controls susceptibility to autoimmune disease. J. Exp. Med. 187, 537–546 (1998).
Suen, W.E., Bergman, C.M., Hjelmstrom, P. & Ruddle, N.H. A critical role for lymphotoxin in experimental allergic encephalomyelitis. J. Exp. Med. 186, 1233–1240 (1997).
Shahinian, A. et al. Differential T cell costimulatory requirements in CD28-deficient mice. Science 261, 609–612 (1993).
Kündig, T.M. et al. Duration of TCR stimulation determines costimulatory requirements. Immunity 5, 41–52 (1996).
Bertram, E.M., Lau, P. & Watts, T.H. Temporal segregation of 4-1BB versus CD28 mediated costimulation: 4-1BBL influences T cell numbers late in the primary response and regulates the size of the T cell memory response following influenza infection. J. Immunol. 168, 3777–3785 (2002).
Dong, H. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nat. Med. 8, 793–800 (2002).
Inaba, K. et al. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J. Exp. Med. 176, 1693–1702 (1992).
Raptis, L. et al. Cellular ras gene activity is required for full neoplastic transformation by the large tumor antigen of SV40. Cell Growth Differ. 8, 891–901 (1997).
Acknowledgements
We thank A. Tafuri and A. Shahinian for their preliminary data; T. Walker and S. Goncharova for technical assistance with airway inflammation experiments; A. Ho for help with EAE experiments; Z. Hao for advice on autoantibody analysis; D. Bouchard for cell sorting and preparation of the manuscript; and M. Saunders for scientific editing. This work was supported by the Canadian Network for Vaccines and Immunotherapeutics of Cancer and Chronic Viral Diseases. W.-K.S. is the recipient of a postdoctoral fellowship from the Cancer Research Institute.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Suh, WK., Gajewska, B., Okada, H. et al. The B7 family member B7-H3 preferentially down-regulates T helper type 1–mediated immune responses. Nat Immunol 4, 899–906 (2003). https://doi.org/10.1038/ni967
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni967
This article is cited by
-
Multi-omics analyses of CD276 in pan-cancer reveals its clinical prognostic value in glioblastoma and other major cancer types
BMC Cancer (2023)
-
The effects of MYC on tumor immunity and immunotherapy
Cell Death Discovery (2023)
-
Bispecific antibody targeting both B7-H3 and PD-L1 exhibits superior antitumor activities
Acta Pharmacologica Sinica (2023)
-
Switchable CAR T cell strategy against osteosarcoma
Cancer Immunology, Immunotherapy (2023)
-
Immunohistochemical analysis of B7-H3 expression in patients with lung cancer following various anti-cancer treatments
Investigational New Drugs (2023)