Abstract
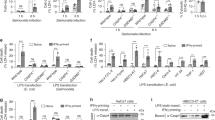
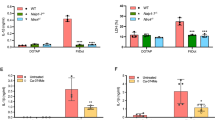
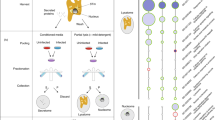
Macrophages respond to Salmonella typhimurium infection via Ipaf, a NACHT–leucine-rich repeat family member that activates caspase-1 and secretion of interleukin 1β. However, the specific microbial salmonella-derived agonist responsible for activating Ipaf is unknown. We show here that cytosolic bacterial flagellin activated caspase-1 through Ipaf but was independent of Toll-like receptor 5, a known flagellin sensor. Stimulation of the Ipaf pathway in macrophages after infection required a functional salmonella pathogenicity island 1 type III secretion system but not the flagellar type III secretion system; furthermore, Ipaf activation could be recapitulated by the introduction of purified flagellin directly into the cytoplasm. These observations raise the possibility that the salmonella pathogenicity island 1 type III secretion system cannot completely exclude 'promiscuous' secretion of flagellin and that the host capitalizes on this 'error' by activating a potent host-defense pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Poltorak, A. et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282, 2085–2088 (1998).
Hayashi, F. et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410, 1099–1103 (2001).
Martinon, F. & Tschopp, J. NLRs join TLRs as innate sensors of pathogens. Trends Immunol. 26, 447–454 (2005).
Inohara, N., Chamaillard, M., McDonald, C. & Nunez, G. NOD-LRR proteins: role in host-microbial interactions and inflammatory disease. Annu. Rev. Biochem. 74, 355–383 (2005).
Harton, J.A., Linhoff, M.W., Zhang, J. & Ting, J.P. Cutting edge: CATERPILLER: a large family of mammalian genes containing CARD, pyrin, nucleotide-binding, and leucine-rich repeat domains. J. Immunol. 169, 4088–4093 (2002).
Kufer, T.A., Fritz, J.H. & Philpott, D.J. NACHT-LRR proteins (NLRs) in bacterial infection and immunity. Trends Microbiol. 13, 381–388 (2005).
Chamaillard, M. et al. An essential role for NOD1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat. Immunol. 4, 702–707 (2003).
Girardin, S.E. et al. Nod1 detects a unique muropeptide from gram-negative bacterial peptidoglycan. Science 300, 1584–1587 (2003).
Girardin, S.E. et al. Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J. Biol. Chem. 278, 8869–8872 (2003).
Inohara, N. et al. Host recognition of bacterial muramyl dipeptide mediated through NOD2. Implications for Crohn's disease. J. Biol. Chem. 278, 5509–5512 (2003).
Kanneganti, T.D. et al. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature 440, 233–236 (2006).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol. Cell 10, 417–426 (2002).
Agostini, L. et al. NALP3 forms an IL-1β-processing inflammasome with increased activity in Muckle-Wells autoinflammatory disorder. Immunity 20, 319–325 (2004).
Martinon, F. & Tschopp, J. Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell 117, 561–574 (2004).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Poyet, J.L. et al. Identification of Ipaf, a human caspase-1-activating protein related to Apaf-1. J. Biol. Chem. 276, 28309–28313 (2001).
Geddes, B.J. et al. Human CARD12 is a novel CED4/Apaf-1 family member that induces apoptosis. Biochem. Biophys. Res. Commun. 284, 77–82 (2001).
Damiano, J.S., Stehlik, C., Pio, F., Godzik, A. & Reed, J.C. CLAN, a novel human CED-4-like gene. Genomics 75, 77–83 (2001).
Chen, L.M., Kaniga, K. & Galan, J.E. Salmonella spp. are cytotoxic for cultured macrophages. Mol. Microbiol. 21, 1101–1115 (1996).
Galan, J.E. Salmonella interactions with host cells: type III secretion at work. Annu. Rev. Cell Dev. Biol. 17, 53–86 (2001).
Hernandez, L.D., Pypaert, M., Flavell, R.A. & Galan, J.E. A Salmonella protein causes macrophage cell death by inducing autophagy. J. Cell Biol. 163, 1123–1131 (2003).
Chilcott, G.S. & Hughes, K.T. Coupling of flagellar gene expression to flagellar assembly in Salmonella enterica serovar typhimurium and Escherichia coli. Microbiol. Mol. Biol. Rev. 64, 694–708 (2000).
Aldridge, P. & Hughes, K.T. Regulation of flagellar assembly. Curr. Opin. Microbiol. 5, 160–165 (2002).
Eichelberg, K. & Galan, J.E. The flagellar sigma factor FliA (σ28) regulates the expression of Salmonella genes associated with the centisome 63 type III secretion system. Infect. Immun. 68, 2735–2743 (2000).
van Asten, F.J., Hendriks, H.G., Koninkx, J.F. & van Dijk, J.E. Flagella-mediated bacterial motility accelerates but is not required for Salmonella serotype Enteritidis invasion of differentiated Caco-2 cells. Int. J. Med. Microbiol. 294, 395–399 (2004).
Guo, L. et al. Regulation of lipid A modifications by Salmonella typhimurium virulence genes phoP-phoQ. Science 276, 250–253 (1997).
Pegues, D.A., Hantman, M.J., Behlau, I. & Miller, S.I. PhoP/PhoQ transcriptional repression of Salmonella typhimurium invasion genes: evidence for a role in protein secretion. Mol. Microbiol. 17, 169–181 (1995).
Means, T.K., Hayashi, F., Smith, K.D., Aderem, A. & Luster, A.D. The Toll-like receptor 5 stimulus bacterial flagellin induces maturation and chemokine production in human dendritic cells. J. Immunol. 170, 5165–5175 (2003).
Applequist, S.E., Wallin, R.P. & Ljunggren, H.G. Variable expression of Toll-like receptor in murine innate and adaptive immune cell lines. Int. Immunol. 14, 1065–1074 (2002).
Mariathasan, S., Weiss, D.S., Dixit, V.M. & Monack, D.M. Innate immunity against Francisella tularensis is dependent on the ASC/caspase-1 axis. J. Exp. Med. 202, 1043–1049 (2005).
Srinivasula, S.M. et al. The PYRIN-CARD protein ASC is an activating adaptor for caspase-1. J. Biol. Chem. 277, 21119–21122 (2002).
Wang, L. et al. PYPAF7, a novel PYRIN-containing Apaf1-like protein that regulates activation of NF-κB and caspase-1-dependent cytokine processing. J. Biol. Chem. 277, 29874–29880 (2002).
Weber, C.H. & Vincenz, C. The death domain superfamily: a tale of two interfaces? Trends Biochem. Sci. 26, 475–481 (2001).
Hersh, D. et al. The Salmonella invasin SipB induces macrophage apoptosis by binding to caspase-1. Proc. Natl. Acad. Sci. USA 96, 2396–2401 (1999).
Hilbi, H. et al. Shigella-induced apoptosis is dependent on caspase-1 which binds to IpaB. J. Biol. Chem. 273, 32895–32900 (1998).
Roy, D. et al. A process for controlling intracellular bacterial infections induced by membrane injury. Science 304, 1515–1518 (2004).
Andrei, C. et al. Phospholipases C and A2 control lysosome-mediated IL-1 β secretion: Implications for inflammatory processes. Proc. Natl. Acad. Sci. USA 101, 9745–9750 (2004).
Lee, S.H. & Galan, J.E. Salmonella type III secretion-associated chaperones confer secretion-pathway specificity. Mol. Microbiol. 51, 483–495 (2004).
Lloyd, S.A., Norman, M., Rosqvist, R. & Wolf-Watz, H. Yersinia YopE is targeted for type III secretion by N-terminal, not mRNA, signals. Mol. Microbiol. 39, 520–531 (2001).
Miao, E.A. & Miller, S.I. A conserved amino acid sequence directing intracellular type III secretion by Salmonella typhimurium. Proc. Natl. Acad. Sci. USA 97, 7539–7544 (2000).
Miao, E.A. et al. Salmonella typhimurium leucine-rich repeat proteins are targeted to the SPI1 and SPI2 type III secretion systems. Mol. Microbiol. 34, 850–864 (1999).
Miao, E.A. et al. Salmonella effectors translocated across the vacuolar membrane interact with the actin cytoskeleton. Mol. Microbiol. 48, 401–415 (2003).
Acknowledgements
We thank V. Dixit and Genentech for Ipaf- and ASC-deficient mice; S. Akira for TLR5- and MyD88-deficient mice; G. Chilcott, H. Bonnifield, O. Nanassy, J. Karlinsey and K. Hughes for flagellar mutant strains and advice regarding flagellar biology; C. Carlson, M. Ohl and K. Smith for contributions and discussions; H. Bonnifield, C. Carlson and members of the Aderem lab for critical review of this manuscript; and E. Andersen-Nissen for providing flagellin and ovalbumin protein.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Schematic of the structures synthesized by flagellar mutants. (PDF 41 kb)
Supplementary Fig. 2
Mutant strains over expressing flagellin display increased cytotoxicity. (PDF 28 kb)
Supplementary Fig. 3
S. typhimurium repress flagellin transcription through PhoP/PhoQ. (PDF 23 kb)
Supplementary Fig. 4
Ipaf sensitizes macrophages to salmonella infection. (PDF 40 kb)
Supplementary Table 1
Strain list: Chromosomal insertion elements and plasmids. (PDF 27 kb)
Rights and permissions
About this article
Cite this article
Miao, E., Alpuche-Aranda, C., Dors, M. et al. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1β via Ipaf. Nat Immunol 7, 569–575 (2006). https://doi.org/10.1038/ni1344
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1344
This article is cited by
-
Exploiting bacterial-origin immunostimulants for improved vaccination and immunotherapy: current insights and future directions
Cell & Bioscience (2024)
-
Revealing novel pyroptosis-related therapeutic targets for sepsis based on machine learning
BMC Medical Genomics (2023)
-
LNCGM1082-mediated NLRC4 activation drives resistance to bacterial infection
Cellular & Molecular Immunology (2023)
-
The distinct translational landscapes of gram-negative Salmonella and gram-positive Listeria
Nature Communications (2023)
-
Squaramides enhance NLRP3 inflammasome activation by lowering intracellular potassium
Cell Death Discovery (2023)