Abstract
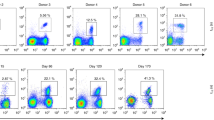
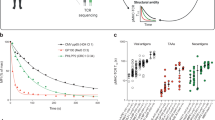
Unlike B cells, T cells lack the capacity to improve the affinity of their antigen receptors by somatic mutation. It is, therefore, believed that optimization of cellular immunity is mediated almost exclusively through selective expansion of T cells bearing receptors with the highest affinity for antigen. We show here that T cell responsiveness to peptide (termed “functional avidity”) increased>50-fold during the early stages of viral infection. This indicated that T cells, like B cells, undergo extensive functional maturation in vivo. However, in contrast to B cells, maturation of the T cell response can occur without any appreciable change in T cell receptor affinity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Eisen, H. N., Sykulev, Y. & Tsomides, T. J. Antigen-specific T-cell receptors and their reactions with complexes formed by peptides with major histocompatibility complex proteins. Adv. Protein Chem. 49, 1–56 (1996).
Valitutti, S. & Lanzavecchia, A. Serial triggering of TCRs: a basis for the sensitivity and specificity of antigen recognition. Immunol. Today 18, 299–304 (1997).
Busch, D. H. & Pamer, E. G. T cell affinity maturation by selective expansion during infection. J. Exp. Med. 189, 701–710 (1999).
Savage, P. A., Boniface, J. J. & Davis, M. M. A kinetic basis for T cell receptor repertoire selection during an immune response. Immunity 10, 485–492 (1999).
Sykulev, Y., Cohen, R. J. & Eisen, H. N. The law of mass action governs antigen-stimulated cytolytic activity of CD8+ cytotoxic T lymphocytes. Proc. Natl Acad. Sci. USA 92, 11990–11992 (1995).
Salzmann, M. & Bachmann, M. F. Estimation of maximal affinities between T-cell receptors and MHC/peptide complexes. Mol. Immunol. 35, 65–71 (1998).
Zinkernagel, R. M. & Doherty, P. C. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248, 701–702 (1974).
Oldstone, M. B. A. et al. Virus and immune responses: lymphocytic choriomeningitis virus as a prototype model of viral pathogenesis. Br. Med. Bull. 41, 70–74 (1985).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
Slifka, M. K. & Whitton, J. L. Antigen-specific regulation of T cell-mediated cytokine production. Immunity 12, 451–457 (2000).
Buchmeier, M. J. & Zajac, A. in Persistent Viral Infections (eds Ahmed, R. & Chen, I.) 575–605 (John Wiley & Sons, New York, 1999).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Whitton, J. L. Lymphocytic choriomeningitis virus CTL. Semin. Virol. 1, 257–262 (1990).
Valitutti, S., Muller, S., Dessing, M. & Lanzavecchia, A. Different responses are elicited in cytotoxic T lymphocytes by different levels of T cell receptor occupancy. J. Exp. Med. 183, 1917–1921 (1996).
Bachmann, M. F. et al. Developmental regulation of lck targeting to the CD8 coreceptor controls signaling in naïve and memory T cells. J. Exp. Med. 189, 1521–1530 (1999).
Nakayama, E. Blocking of effector cell cytotoxicity and T-cell proliferation by Lyt antisera. Immunol. Rev. 68, 117–134 (1982).
Hollander, N. Effects of anti-Lyt antibodies on T-cell functions. Immunol. Rev. 68, 43–66 (1982).
von Herrath, M. G., Dockter, J. & Oldstone, M. B. A. How virus induces a rapid or slow onset insulin-dependent diabetes mellitus in a transgenic model. Immunity 1, 231–242 (1994).
Slifka, M. K. & Whitton, J. L. Activated and memory CD8+ T cells can be distinguished by their cytokine profiles and phenotypic markers. J. Immunol. 164, 208–216 (2000).
Crawford, F. et al. Detection of antigen-specific T cells with multivalent soluble class II MHC covalent peptide complexes. Immunity 8, 675–682 (1998).
Yee, C. et al. Isolation of high avidity melanoma-reactive CTL from heterogeneous populations using peptide-MHC tetramers. J. Immunol. 162, 2227–2234 (1999).
Nugent, C. T. et al. Characterization of CD8+ T lymphocytes that persist after peripheral tolerance to a self antigen expressed in the pancreas. J. Immunol. 164, 191–200 (2000).
Daniels, M. A. & Jameson, S. C. Critical role for CD8 in T cell receptor binding and activation by peptide/major histocompatibility complex multimers. J. Exp. Med. 191, 335–346 (2000).
Whelan, J. A. et al. Specificity of CTL interactions with peptide-MHC class I tetrameric complexes is temperature dependent. J. Immunol. 163, 4342–4348 (1999).
Reichstetter, S. et al. Distinct T cell interactions with HLA class II tetramers characterize a spectrum of TCR affinities in the human antigen-specific T cell response. J. Immunol. 165, 6994–6998 (2000).
Sourdive, D. J. et al. Conserved T cell receptor repertoire in primary and memory CD8 T cell responses to an acute viral infection. J. Exp. Med. 188, 71–82 (1998).
Lin, M. Y. & Welsh, R. M. Stability and diversity of T cell receptor repertoire usage during lymphocytic choriomeningitis virus infection of mice. J. Exp. Med. 188, 1993–2005 (1998).
Pircher, H. et al. Tolerance induction in double specific T-cell receptor transgenic mice varies with antigen. Nature 342, 559–561 (1989).
Fitzpatrick, D. R. et al. Distinct methylation of the interferon gamma (IFN-γ) and interleukin 3 (IL-3) genes in newly activated primary CD8+ T lymphocytes: regional IFN-γ promoter demethylation and mRNA expression are heritable in CD44hiCD8+ T cells. J. Exp. Med. 188, 103–117 (1998).
Agarwal, S. & Rao, A. Modulation of chromatin structure regulates cytokine gene expression during T cell differentiation. Immunity 9, 765–775 (1998).
Oehen, S. & Brduscha-Riem, K. Differentiation of naïve CTL to effector and memory CTL: correlation of effector function with phenotype and cell division. J. Immunol. 161, 5338–5346 (1998).
Ahmed, R. Virus persistence and immune memory. Semin. Virol. 5, 319–324 (1994).
Bachmann, M. F. et al. Distinct roles for LFA-1 and CD28 during activation of naïve T cells: adhesion versus costimulation. Immunity 7, 549–557 (1997).
Bachmann, M. F., Barner, M. & Kopf, M. CD2 sets quantitative thresholds in T cell activation. J. Exp. Med. 190, 1383–1392 (1999).
Harrington, L. E. et al. Differentiating between memory and effector CD8 T cells by altered expression of cell surface O-glycans. J. Exp. Med. 191, 1241–1246 (2000).
Slifka, M. K., Pagarigan, R. R. & Whitton, J. L. NK markers are expressed on a high percentage of virus-specific CD8+ and CD4+ T cells. J. Immunol. 164, 2009–2015 (2000).
Weiss, A. & Littman, D. R. Signal transduction by lymphocyte antigen receptors. Cell 76, 263–274 (1994).
Chan, A. C. & Shaw, A. S. Regulation of antigen receptor signal transduction by protein tyrosine kinases. Curr. Opin. Immunol. 8, 394–401 (1996).
Wange, R. L. & Samelson, L. E. Complex complexes: signaling at the TCR. Immunity 5, 197–205 (1996).
Horwitz, M. S., Yanagi, Y. & Oldstone, M. B. A. T-cell receptors from virus-specific cytotoxic T lymphocytes recognizing a single immunodominant nine-amino-acid viral epitope show marked diversity. J. Virol. 68, 352–357 (1994).
Myung, P. S., Boerthe, N. J. & Koretzky, G. A. Adapter proteins in lymphocyte antigen-receptor signaling. Curr. Opin. Immunol. 12, 256–266 (2000).
Zhang, W. & Samelson, L. E. The role of membrane-associated adaptors in T cell receptor signalling. Semin. Immunol. 12, 35–41 (2000).
Kane, L. P., Lin, J. & Weiss, A. Signal transduction by the TCR for antigen. Curr. Opin. Immunol. 12, 242–249 (2000).
Fahmy, T. M., Bieler, J. G., Edidin, M. & Schneck, J. P. Increased TCR avidity after T cell activation: a mechanism for sensing low-density antigen. Immunity 14, 135–143 (2001).
Holler, P. D. et al. In vitro evolution of a T cell receptor with high affinity for peptide/MHC. Proc. Natl Acad. Sci. USA 97, 5387–5392 (2000).
Alexander-Miller, M. A., Leggatt, G. R., Sarin, A. & Berzofsky, J. A. Role of antigen, CD8, and cytotoxic T lymphocyte (CTL) avidity in high dose antigen induction of apoptosis of effector CTL. J. Exp. Med. 184, 485–492 (1996).
Acknowledgements
We thank A. Lord for secretarial support, R. Pagarigan for technical assistance and C. T. Nugent, R. Soloff and D. Parker for helpful discussions. NP(118–126) and GP(33–41) tetramers were prepared by the NIH Tetramer Core Facility in Atlanta, GA. Supported by NIH grant AI-27028.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Slifka, M., Whitton, J. Functional avidity maturation of CD8+ T cells without selection of higher affinity TCR. Nat Immunol 2, 711–717 (2001). https://doi.org/10.1038/90650
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/90650
This article is cited by
-
Interferon-γ couples CD8+ T cell avidity and differentiation during infection
Nature Communications (2023)
-
The pursuit of transplantation tolerance: new mechanistic insights
Cellular & Molecular Immunology (2019)
-
T-cell receptor-engineered T cells for cancer treatment: current status and future directions
Protein & Cell (2018)
-
Immune Signature of Enhanced Functional Avidity CD8+ T Cells in vivo Induced by Vaccinia Vectored Vaccine
Scientific Reports (2017)
-
Heterogeneity assessment of functional T cell avidity
Scientific Reports (2017)