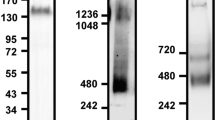
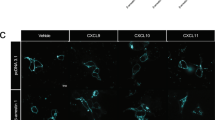
Abstract
Regulation of the affinity of the β2 integrin LFA-1 by chemokines is critical to lymphocyte trafficking, but the signaling mechanisms that control this process are not well understood. Here we investigated the signaling events controlling LFA-1 affinity triggering by chemokines in human primary T lymphocytes. We found that the small GTPase Rac1 mediated chemokine-induced LFA-1 affinity triggering and lymphocyte arrest in high endothelial venules. Unexpectedly, another Rho family member, Cdc42, negatively regulated LFA-1 activation. The Rho effectors PLD1 and PIP5KC were also critical to LFA-1 affinity modulation. Notably, PIP5KC was found to specifically control the transition of LFA-1 from an extended low–intermediate state to a high-affinity state, which correlated with lymphocyte arrest. Thus, chemokines control lymphocyte trafficking by triggering a Rho-dependent signaling cascade leading to conformer-specific modulation of LFA-1 affinity
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Butcher, E.C. Leukocyte-endothelial cell recognition: three (or more) steps to specificity and diversity. Cell 67, 1033–1036 (1991).
Alon, R. & Dustin, M.L. Force as a facilitator of integrin conformational changes during leukocyte arrest on blood vessels and antigen-presenting cells. Immunity 26, 17–27 (2007).
Ley, K., Laudanna, C., Cybulsky, M.I. & Nourshargh, S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat. Rev. Immunol. 7, 678–689 (2007).
Laudanna, C. & Alon, R. Right on the spot. Chemokine triggering of integrin-mediated arrest of rolling leukocytes. Thromb. Haemost. 95, 5–11 (2006).
Kinashi, T. Intracellular signalling controlling integrin activation in lymphocytes. Nat. Rev. Immunol. 5, 546–559 (2005).
Carman, C.V. & Springer, T.A. Integrin avidity regulation: are changes in affinity and conformation underemphasized? Curr. Opin. Cell Biol. 15, 547–556 (2003).
Constantin, G. et al. Chemokines trigger immediate beta2 integrin affinity and mobility changes: differential regulation and roles in lymphocyte arrest under flow. Immunity 13, 759–769 (2000).
Cairo, C.W., Mirchev, R. & Golan, D.E. Cytoskeletal regulation couples LFA-1 conformational changes to receptor lateral mobility and clustering. Immunity 25, 297–308 (2006).
Giagulli, C. et al. RhoA and zeta PKC control distinct modalities of LFA-1 activation by chemokines: critical role of LFA-1 affinity triggering in lymphocyte in vivo homing. Immunity 20, 25–35 (2004).
Kim, M., Carman, C.V., Yang, W., Salas, A. & Springer, T.A. The primacy of affinity over clustering in regulation of adhesiveness of the integrin αLβ2. J. Cell Biol. 167, 1241–1253 (2004).
Shamri, R. et al. Lymphocyte arrest requires instantaneous induction of an extended LFA-1 conformation mediated by endothelium-bound chemokines. Nat. Immunol. 6, 497–506 (2005).
Giagulli, C. et al. The Src family kinases Hck and Fgr are dispensable for inside-out, chemoattractant-induced signaling regulating beta 2 integrin affinity and valency in neutrophils, but are required for beta 2 integrin-mediated outside-in signaling involved in sustained adhesion. J. Immunol. 177, 604–611 (2006).
D'Ambrosio, D. et al. Quantitative differences in chemokine receptor engagement generate diversity in integrin-dependent lymphocyte adhesion. J. Immunol. 169, 2303–2312 (2002).
Pasvolsky, R. et al. RhoA is involved in LFA-1 extension triggered by CXCL12 but not in a novel outside-in LFA-1 activation facilitated by CXCL9. J. Immunol. 180, 2815–2823 (2008).
Shimonaka, M. et al. Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J. Cell Biol. 161, 417–427 (2003).
Katagiri, K., Maeda, A., Shimonaka, M. & Kinashi, T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat. Immunol. 4, 741–748 (2003).
Prochiantz, A. Homeodomain-derived peptides. In and out of the cells. Ann. NY Acad. Sci. 886, 172–179 (1999).
Zhao, X., Carnevale, K.A. & Cathcart, M.K. Human monocytes use Rac1, not Rac2, in the NADPH oxidase complex. J. Biol. Chem. 278, 40788–40792 (2003).
Hall, A. Rho GTPases and the actin cytoskeleton. Science 279, 509–514 (1998).
Zigmond, S.H. et al. Mechanism of Cdc42-induced actin polymerization in neutrophil extracts. J. Cell Biol. 142, 1001–1012 (1998).
Powner, D.J. & Wakelam, M.J. The regulation of phospholipase D by inositol phospholipids and small GTPases. FEBS Lett. 531, 62–64 (2002).
Henage, L.G., Exton, J.H. & Brown, H.A. Kinetic analysis of a mammalian phospholipase D: allosteric modulation by monomeric GTPases, protein kinase C, and polyphosphoinositides. J. Biol. Chem. 281, 3408–3417 (2006).
Bae, C.D., Min, D.S., Fleming, I.N. & Exton, J.H. Determination of interaction sites on the small G protein RhoA for phospholipase D. J. Biol. Chem. 273, 11596–11604 (1998).
Brown, H.A., Henage, L.G., Preininger, A.M., Xiang, Y. & Exton, J.H. Biochemical analysis of phospholipase D. Methods Enzymol. 434, 49–87 (2007).
Yamazaki, M. et al. Interaction of the small G protein RhoA with the C terminus of human phospholipase D1. J. Biol. Chem. 274, 6035–6038 (1999).
Cai, S. & Exton, J.H. Determination of interaction sites of phospholipase D1 for RhoA. Biochem. J. 355, 779–785 (2001).
Weernink, P.A. et al. Activation of type I phosphatidylinositol 4-phosphate 5-kinase isoforms by the Rho GTPases, RhoA, Rac1, and Cdc42. J. Biol. Chem. 279, 7840–7849 (2004).
Jarquin-Pardo, M., Fitzpatrick, A., Galiano, F.J., First, E.A. & Davis, J.N. Phosphatidic acid regulates the affinity of the murine phosphatidylinositol 4-phosphate 5-kinase-Ibeta for phosphatidylinositol-4-phosphate. J. Cell. Biochem. 100, 112–128 (2007).
Kanaho, Y., Kobayashi-Nakano, A. & Yokozeki, T. The phosphoinositide kinase PIP5K that produces the versatile signaling phospholipid PI4,5P(2). Biol. Pharm. Bull. 30, 1605–1609 (2007).
Martel, V. et al. Conformation, localization, and integrin binding of talin depend on its interaction with phosphoinositides. J. Biol. Chem. 276, 21217–21227 (2001).
Wegener, K.L. et al. Structural basis of integrin activation by talin. Cell 128, 171–182 (2007).
Ling, K., Doughman, R.L., Firestone, A.J., Bunce, M.W. & Anderson, R.A. Type I gamma phosphatidylinositol phosphate kinase targets and regulates focal adhesions. Nature 420, 89–93 (2002).
Di Paolo, G. et al. Recruitment and regulation of phosphatidylinositol phosphate kinase type 1 gamma by the FERM domain of talin. Nature 420, 85–89 (2002).
Barsukov, I.L. et al. Phosphatidylinositol phosphate kinase type 1gamma and beta1-integrin cytoplasmic domain bind to the same region in the talin FERM domain. J. Biol. Chem. 278, 31202–31209 (2003).
Battistini, L. et al. CD8+ T cells from patients with acute multiple sclerosis display selective increase of adhesiveness in brain venules: a critical role for P-selectin glycoprotein ligand-1. Blood 101, 4775–4782 (2003).
Piccio, L. et al. Efficient recruitment of lymphocytes in inflamed brain venules requires expression of cutaneous lymphocyte antigen and fucosyltransferase-VII. J. Immunol. 174, 5805–5813 (2005).
Garcia-Bernal, D. et al. Vav1 and Rac control chemokine-promoted T lymphocyte adhesion mediated by the integrin alpha4beta1. Mol. Biol. Cell 16, 3223–3235 (2005).
Colley, W.C. et al. Phospholipase D2, a distinct phospholipase D isoform with novel regulatory properties that provokes cytoskeletal reorganization. Curr. Biol. 7, 191–201 (1997).
Sampath, R., Gallagher, P.J. & Pavalko, F.M. Cytoskeletal interactions with the leukocyte integrin beta2 cytoplasmic tail. Activation-dependent regulation of associations with talin and alpha-actinin. J. Biol. Chem. 273, 33588–33594 (1998).
Stanley, P. et al. Intermediate-affinity LFA-1 binds alpha-actinin-1 to control migration at the leading edge of the T cell. EMBO J. 27, 62–75 (2008).
Iyer, S.S. & Kusner, D.J. Association of phospholipase D activity with the detergent-insoluble cytoskeleton of U937 promonocytic leukocytes. J. Biol. Chem. 274, 2350–2359 (1999).
Kam, Y. & Exton, J.H. Phospholipase D activity is required for actin stress fiber formation in fibroblasts. Mol. Cell. Biol. 21, 4055–4066 (2001).
Isenberg, G. & Goldmann, W.H. Peptide-specific antibodies localize the major lipid binding sites of talin dimers to oppositely arranged N-terminal 47 kDa subdomains. FEBS Lett. 426, 165–170 (1998).
Woolf, E. et al. Lymph node chemokines promote sustained T lymphocyte motility without triggering stable integrin adhesiveness in the absence of shear forces. Nat. Immunol. 8, 1076–1085 (2007).
Zarbock, A., Lowell, C.A. & Ley, K. Spleen tyrosine kinase Syk is necessary for E-selectin-induced alpha(L)beta(2) integrin-mediated rolling on intercellular adhesion molecule-1. Immunity 26, 773–783 (2007).
Hartwell, L.H., Hopfield, J.J., Leibler, S. & Murray, A.W. From molecular to modular cell biology. Nature 402, C47–C52 (1999).
Bhalla, U.S. & Iyengar, R. Functional modules in biological signalling networks. Novartis Found. Symp. 239, 4–13 discussion 13–5, 45–51 (2001).
Pereira-Leal, J.B., Levy, E.D. & Teichmann, S.A. The origins and evolution of functional modules: lessons from protein complexes. Phil. Trans. R. Soc. Lond. B 361, 507–517 (2006).
Segal, E., Friedman, N., Kaminski, N., Regev, A. & Koller, D. From signatures to models: understanding cancer using microarrays. Nat. Genet. 37 Suppl, S38–S45 (2005).
Robinson, M.K. et al. Antibody against the Leu-CAM beta-chain (CD18) promotes both LFA-1- and CR3-dependent adhesion events. J. Immunol. 148, 1080–1085 (1992).
Shimaoka, M. et al. AL-57, a ligand-mimetic antibody to integrin LFA-1, reveals chemokine-induced affinity up-regulation in lymphocytes. Proc. Natl. Acad. Sci. USA 103, 13991–13996 (2006).
Lum, A.F., Green, C.E., Lee, G.R., Staunton, D.E. & Simon, S.I. Dynamic regulation of LFA-1 activation and neutrophil arrest on intercellular adhesion molecule 1 (ICAM-1) in shear flow. J. Biol. Chem. 277, 20660–20670 (2002).
Acknowledgements
Supported by the Italian Association for Cancer Research, the Italian Ministry of University and Scientific Research, Fondazione Cariverona (Verona, Italy), the National Multiple Sclerosis Society of New York and Fondazione Italiana Sclerosi Multipla.
Author information
Authors and Affiliations
Contributions
M.B.-V. developed the Tat technology, set up the siRNA technology and did the adhesion assays; A.M. set up the siRNA technology and did adhesion assays and biochemical studies, ImageStream experiments and analysis; C.G. developed the P1 technology; D.S. provided monoclonal antibody 327C; B.R. and M.M. did the intravital microscopy studies; G.C. provided expertise in intravital microscopy and assistance with writing; and C.L. designed the study, analyzed the data and wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Methods (PDF 718 kb)
Rights and permissions
About this article
Cite this article
Bolomini-Vittori, M., Montresor, A., Giagulli, C. et al. Regulation of conformer-specific activation of the integrin LFA-1 by a chemokine-triggered Rho signaling module. Nat Immunol 10, 185–194 (2009). https://doi.org/10.1038/ni.1691
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1691
This article is cited by
-
LFA-1 nanoclusters integrate TCR stimulation strength to tune T-cell cytotoxic activity
Nature Communications (2024)
-
Intravascular adhesion and recruitment of neutrophils in response to CXCL1 depends on their TRPC6 channels
Journal of Molecular Medicine (2020)
-
PLD-dependent phosphatidic acid microdomains are signaling platforms for podosome formation
Scientific Reports (2019)
-
Rac1 activation links tau hyperphosphorylation and Aβ dysmetabolism in Alzheimer’s disease
Acta Neuropathologica Communications (2018)
-
Nanovesicles from adipose-derived mesenchymal stem cells inhibit T lymphocyte trafficking and ameliorate chronic experimental autoimmune encephalomyelitis
Scientific Reports (2018)