Abstract
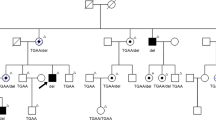
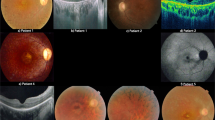
The gene RPGR was previously identified in the RP3 region of Xp21.1 and shown to be mutated in 10–20% of patients with the progressive retinal degeneration X-linked retinitis pigmentosa1,2 (XLRP). The mutations predominantly affected a domain homologous to RCC1, a guanine nucleotide exchange factor for the small GTPase Ran, although they were present in fewer than the 70–75% of XLRP patients predicted from linkage studies3,4,5,6. Mutations in the RP2 locus at Xp11.3 were found in a further 10–20% of XLRP patients, as predicted from linkage studies6,7,8. Because the mutations in the remainder of the XLRP patients may reside in undiscovered exons of RPGR, we sequenced a 172-kb region containing the entire gene. Analysis of the sequence disclosed a new 3′ terminal exon that was mutated in 60% of XLRP patients examined. This exon encodes 567 amino acids, with a repetitive domain rich in glutamic acid residues. The sequence is conserved in the mouse, bovine and Fugu rubripes genes. It is preferentially expressed in mouse and bovine retina, further supporting its importance for retinal function. Our results suggest that mutations in RPGR are the only cause of RP3 type XLRP and account for the disease in over 70% of XLRP patients and an estimated 11% of all retinitis pigmentosa patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Meindl, A. et al. A gene (RPGR) with homology to the RCC1 guanine nucleotide exchange factor is mutated in X-linked retinitis pigmentosa (RP3). Nature Genet. 13, 35–42 (1996).
Roepman, R. et al. Positional cloning of the gene for retinitis pigmentosa 3: homology with the guanine-nucleotide-exchange factor RCC1. Hum. Mol. Genet. 5, 1035–1041 (1996).
Buraczynska, M. et al. Spectrum of mutations in the RPGR gene that are identified in 20% of families with X-linked retinitis pigmentosa. Am. J. Hum. Genet. 61, 1287–1292 (1997).
Miano, M.G. et al. Mutation analysis of the RPGR gene reveals novel mutations in South European patients with X-linked retinitis pigmentosa (RP3). Eur. J. Hum. Genet. 7, 687–694 (1999).
Ott, J. et al. Localizing multiple X chromosome-linked retinitis pigmentosa loci using multilocus homogeneity tests. Proc. Natl Acad. Sci. USA 87, 701–704 (1990).
Teague, P.W. et al. Heterogeneity analysis in 40 X-linked retinitis pigmentosa families. Am. J. Hum. Genet. 55, 105–111 (1994).
Schwahn, U. et al. Positional cloning of the gene for X-linked retinitis pigmentosa 2. Nature Genet. 19, 327–332 (1998).
Hardcastle, A.J. et al. Mutations in the RP2 gene cause disease in 10% of families with familial X-linked retinitis pigmentosa assessed in this study. Am. J. Hum. Genet. 64, 1210–1215 (1999).
Dry, K.L. et al. Identification of a novel gene, ETX1 from Xp21.1, a candidate gene for X-linked retintis pigmentosa (RP3). Hum. Mol. Genet. 4, 2347–2353 (1995).
Meindl, A. et al. A gene (SRPX) encoding a sushi-repeat-containing protein is deleted in patients with X-linked retinitis pigmentosa. Hum. Mol. Genet. 4, 2339–2346 (1995).
Kirschner, R. et al. RPGR transcription studies in mouse and human tissues reveal a retina-specific isoform that is disrupted in a patient with X-linked retinitis pigmentosa. Hum. Mol. Genet. 8, 1571–1578 (1999).
Wootton, J.C. Sequences with ‘unusual’ amino acid compositions. Curr. Opin. Struct. Biol. 4, 413–421 (1994).
Wang, G., Seidman, M.M. & Glazer, P.M. Mutagenesis in mammalian cells induced by triple helix formation and transcription-coupled repair. Science 271, 802–805 (1996).
Cooper, D.N. & Krawczak, M. Human Gene Mutation (Bios, Oxford, 1993).
Weaver, D.T. & DePamphilis, M.L. Specific sequences in native DNA that arrest synthesis by DNA polymerase a. J. Biol. Chem. 257, 2075–2086 (1982).
Roepman, R. et al. Identification of a gene disrupted by a microdeletion in a patient with X-linked retinitis pigmentosa (XLRP). Hum. Mol. Genet. 5, 827–833 (1996).
Jurka, J., Klonowski, P., Dagman, V. & Pelton, P. CENSORæa program for identification and elimination of repetitive elements from DNA sequences. Comput. Chem. 20, 119–121 (1996).
Zhang, M.Q. Identification of protein coding regions in the human genome by quadratic discriminant analysis. Proc. Natl Acad. Sci. USA 94, 565–568 (1997).
Xu, Y., Mural, R., Shah, M. & Uberbacher, E. Recognizing exons in genomic sequence using GRAIL II. Genet. Eng. 16, 241–253 (1994).
Burge, C. & Karlin, S. Prediction of complete gene structures in human genomic DNA. J. Mol. Biol. 268, 78–94 (1997).
Reese, M.G., Eeckman, F.H., Kulp, D. & Haussler, D. Improved splice site detection in Genie. J. Comput. Biol. 4, 311–323 (1997).
Dunn, K.C., Aotaki-Keen, A.E., Putkey, F.R. & Hjelmeland, L.M. ARPE-19, a human retinal pigment epithelial cell line with differentiated properties. Exp. Eye Res. 62, 155–169 (1996).
Dry, K. et al. Identification of a 5′ splice site mutation in the RPGR gene in a family with X-linked retinitis pigmentosa (RP3). Hum. Mutat. 13, 141–145 (1999).
Acknowledgements
We thank M. Boulton and V. Smith for human retinas; J. Kenyon for the mouse PAC clones; M. Warren and L. Sawyer for comments; the Sequencing Unit Core of Area di Ricerca di Napoli, CNR, for cosmid sequencing; and M. D'Urso for support. We acknowledge the generous support of The Foundation Fighting Blindness, the British Retinitis Pigmentosa Society, Guide Dogs for the Blind Association and the UK Medical Research Council. A.C. was supported by Telethon, Italy grant E1093.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Vervoort, R., Lennon, A., Bird, A. et al. Mutational hot spot within a new RPGR exon in X-linked retinitis pigmentosa. Nat Genet 25, 462–466 (2000). https://doi.org/10.1038/78182
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/78182
This article is cited by
-
Ciliary tip actin dynamics regulate photoreceptor outer segment integrity
Nature Communications (2024)
-
Restoration of RPGR expression in vivo using CRISPR/Cas9 gene editing
Gene Therapy (2022)
-
Extending the phenotypic spectrum of PRPF8, PRPH2, RP1 and RPGR, and the genotypic spectrum of early-onset severe retinal dystrophy
Orphanet Journal of Rare Diseases (2021)
-
X-linked dominant RPGR gene mutation in a familial Coats angiomatosis
BMC Ophthalmology (2021)
-
The genetics of rod-cone dystrophy in Arab countries: a systematic review
European Journal of Human Genetics (2021)