Abstract
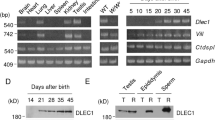
Severely reduced fertility is a common finding in cystic fibrosis (CF). We used in situ hybridization to examine the cell–specific expression of CFTR in the reproductive organs of rodents. In males CFTR mRNA is found in the round spermatids (spermatogenic stages V–X) and in the principal cells that line the initial segment of the epididymis. In both the testis and the epididymis, CFTR expression is developmentally regulated suggesting that the defect in the genital tract of male CF patients is of developmental origin. CFTR expression in the luminal and glandular epithelium of the uterus is regulated during the oestrous cycle and is maximal at pro–oestrus. Our results provide a biological rationale for the reduced fertility of CF patients, and suggest a possible cause for the comparatively poorer prognosis for women with CF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rommens, J.M. et al. Identification of the cystic fibrosis gene: Chromosome walking and jumping. Science 245, 1059–1065 (1989).
Kerem, B.-S. et al. Identification of the cystic fibrosis gene: Genetic analysis. Science 245, 1073–1080 (1989).
Cheng, S.H. et al. Defective intracellular transport and processing of CFTR is the molecular basis if most cystic fibrosis. Cell 63, 827–834 (1990).
Kartner, N., Augustinas, O., Jensen, T.J., Naismith, A.L. & Riordan, J.R. Mislocatisation of ΔF508 CFTR in cystic figrosis sweat gland. Nature Genet. 1, 321–327 (1992).
Anderson, M.P., Rich, D.P., Gregory, R.J., Smith, A.E. & Welsh, M.J. Generation of cAMP-activated chloride currents by expression of CFTR. Science 251, 679–682 (1991).
Kartner, N. et al. Expression of the cystic fibrosis gene in non-epithelial invertebrate cells produces a regulated anion conductance. Cell 64, 681–691 (1991).
Bear, C.E. et al. Purification and functional reconstitution of the cystic fibrosis transmembrane conductance regulator. Cell 68, 809–818 (1992).
Trezise, A.E.O. & Buchwald, M. In vivo cell-specific expression of the cystic fibrosis transmembrane conductance regulator. Nature 353, 434–437 (1991).
Brugman, S.M. & Taussig, L.M. The reproductive system. In Cystic Fibrosis. (ed. Taussig, L.M.) 323–337 (Thieme-Stratton, New York, 1984).
Clermont, Y. Kinetics of spermatogenesis in mammals: seminiferous epithelium cycle and spermatogonial renewal. Phys. Rev. 52, 198–236 (1972).
Leblond, C.P. & Clermont, Y. Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann. N.Y. Acad. Sci. 55, 548–573 (1952).
Hess, R.A. Quantitative and qualitative characteristics of the stages and transitions in the cycle of the rat seminiferous epithelium: light microscopic observations of perfusion-fixed and plastic-embedded testes. Biol. Reprod. 43, 525–542 (1990).
Morales, C.R., Alcivar, A.A., Hecht, N.B. & Griswold, M.D. Specific mRNAs in sertoli and germinal cells of testes from stage synchronised rats. Molec. Endocrin. 3, 725–733 (1989).
Russell, L.D., Alger, L.E. & Nequin, L.G. Hormonal control of pubertal spermatogenesis. Endocrinology 120, 1615–1632 (1987).
Robaire, B. & Hermo, L. Efferent ducts, epididymis, and vas deferens: structure, function and their regulation. In Physiology of Reproduction (eds Knobil, E. & Neill, J.) 999–1080 (Raven Press, New York 1988).
Heaton, N.D. & Pryor, J.P. Vasa aplasia and cystic fibrosis. Br. J. Urol. 66, 538–540 (1990).
Gottlieb, C., Ploen, L., Kvist, U. & Strandvik, B. The fertility potential of male cystic fibrosis patients. Int. J. Andro. 14, 437–440 (1991).
Stallard, B.J., Collard, M.W. & Griswold, M.D. A transferrinlike (hemiferrin) mRNA is expressed in the germ cells of rat testis. Molec. cell. Biol. 11, 1448–1453 (1991).
Crawford, I. et al. Immunocytochemical localisation of the cystic fibrosis gene product CFTR. Proc. natn. Acad. Sci. U.S.A. 88, 9262–9266 (1991).
Cohn, J.A., Melhus, O., Page, L.J., Dittrich, K.L. & Vigna, S.R. CFTR: development of high-affinity antibodies and localisation in sweat gland. Biochem. Biophys. Res. Comm. 181, 36–43 (1991).
Quinton, P.M. Cystic fibrosis: a disease in electrolyte transport. FASEB J. 4, 2709–2717 (1990).
Sprando, R.L. & Russell, L.D. Comparative study of cytoplasmic elimination in spermatids of selected mammalian species. Am. J. Anat. 178, 72–80 (1987).
Phillips, D.M. Insect sperm: their structure and morphogenesis. J. cell Biol. 44, 243–247 (1970).
Braun, R.E., Behringer, R.R., Peschon, J.J., Brinster, R.L. & Palmiter, R.D. Genetically haploid spermatids are phenotypically diploid. Nature 337, 373–376 (1989).
Caldwell, K.A. & Handel, M.A. Protamine transcipt sharing among postmeiotic spermatids. Proc. natn. Acad. Sci. U.S.A. 88, 2407–2411 (1991).
Murti, J.R., Bumbulis, M. & Schimenti, J.C. High-frequency germ line gene conversion in transgenic mice. Molec. cell Biol. 12, 2545–2552 (1992).
Schimenti, J. et al. A candidate gene family for the mouse t complex responder (Tcr) locus responsible for haploid effects on sperm function. Cell 55, 71–78 (1988).
Tsui, L.-C. & Buchwald, M. Biochemical and molecular genetics of cystic fibrosis. Adv. hum. Genet., 20, 153–266 (1991).
Riordan, J.R. et al. Identification of the cystic fibrosis gene: cloning and characterisation of complementary DNA. Science 245, 1066–1073 (1989).
Erikson, R.P. Post-meiotic gene expression. Trends Genet. 6, 264–269 (1990).
Wong, P.Y.D., Au, C.L. & Ngai, H.K. Electrolyte and water transport in rat epididymis; its possible role in sperm maturation. Int. J. Androl. Suppl2, 608–628 (1978).
Wong, P.Y.D. & Yeung, C.H. Absorptive and secretory functions of the perfused rat cauda epididymis. J. Physiol. 275, 13–26 (1978).
Harris, A., Chalkley, G., Goodman, S. & Coleman, L. Expression of the cystic fibrosis gene in human development. Development 113, 305–310 (1991).
Setchell, B.P. & Brooks, D.E. Anatomy, vasculature, innervation, and fluids of the male reproductive tract. In Physiology of Reproduction (eds Knobil, E. & Neill, J.) 753–836 (Raven Press, New York 1988).
Harper, M.J.K. Gamete and zygote transport. In Physiology of Reproduction (eds Knobil, E. & Neill, J.) 103–134 (Raven Press, New York, 1988).
Arceci, R.J., Croop, J.M., Horwitz, S.B. & Housman, D. The gene encoding multidrug resistance is induced and expressed at high levels during pregnancy in the secretory epithelium of the uterus. Proc. natn. Acad. Sci. U.S.A. 85, 4350–4354 (1988).
Trezise, A.E.O. et al. The multidrug resistance and cystic fibrosis genes have complementary patterns of epithelial expression. EMBO J. 11, 4291–4303 (1992).
Valverde, M.A. et al. Volume-regulated chloride channels associated with the human multidrug-resistance P-glycoprotein. Nature 355, 830–833 (1992).
Snouwaert, J.N. et al. An animal model for cystic fibrosis made by gene targeting. Science 257, 1083–1088 (1992).
Clarke, L.L. et al. Defective epithelial chloride transport in a gene-targeted mouse model of cystic fibrosis. Science 257, 1125–1128 (1992).
Colledge, W.H., Ratcliff, R., Foster, D., Williamson, R. & Evans, M.J. Cystic fibrosis mouse with intestinal obstruction. Lancet 340, 680 (1992).
Dorin, J.R. et al. Cystic fibrosis in the mouse by targeted insertional mutagenesis. Nature 359, 211–215 (1992).
Corey, M., McLaughlin, F.J., Williams, M. & Levison, H. A comparison of survival, growth and pulmonary function in patients with cystic fibrosis in Boston and Toronto. J. clin. Epidem. 41, 583–591 (1988).
Chalon, J., Loew, D.A.Y. & Orkin, L.R. Tracheobronchial cytologic changes during the menstrual cycle. JAMA 218, 1928–1931 (1971).
Badley, J.E., Bishop, G.A., St John, T. & Frelinger, J.A. A simple, rapid method for the purification of Poly A+ RNA. Biotechniques 6, 114–116 (1988).
Feinberg, A.P. & Vogelstein, B. A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 132, 6–13 (1983).
Trezise, A.E.O., Szpirer, C. & Buchwald, M. Localisation of the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR) in the rat to chromosome 4 and implications for the evolution of mammalian chromosomes. Genomics 14, 869–874 (1992).
Simmons, D.M., Arriza, J.L. & Swanson, L.W. A complete protocol for in situ hybridization of messenger RNAs in brain and other tissues with radiolabelled single-stranded RNA probes. J. Histotech. 12, 169–181 (1989).
Oakberg, E.F. A description of spermiogenesis in the mouse and its use in analysis of the cycle of the seminiferous epithelium and germ cell renewal. Am. J. Anat. 99, 391–413 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trezise, A., Linder, C., Grieger, D. et al. CFTR expression is regulated during both the cycle of the seminiferous epithelium and the oestrous cycle of rodents. Nat Genet 3, 157–164 (1993). https://doi.org/10.1038/ng0293-157
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0293-157
This article is cited by
-
Cystic fibrosis transmembrane conductance regulator—emerging regulator of cancer
Cellular and Molecular Life Sciences (2018)
-
In vitro and in vivo MMP gene expression localisation by In Situ-RT-PCR in cell culture and paraffin embedded human breast cancer cell line xenografts
BMC Cancer (2006)
-
Absence de corrélation génotype-phénotype dans les absences de canaux déférents
Andrologie (2001)
-
In Vivo Analysis of DNase I Hypersensitive Sites in the Human CFTR Gene
Molecular Medicine (1999)
-
Counseling and screening for cystic fibrosis in patients with congenital bilateral absence of the vas deferens: Patient perceptions
Journal of Genetic Counseling (1996)