Abstract
Efficient lead halide perovskite solar cells use hole-blocking layers to help collection of photogenerated electrons and to achieve high open-circuit voltages. Here, we report the realization of efficient perovskite solar cells grown directly on fluorine-doped tin oxide-coated substrates without using any hole-blocking layers. With ultraviolet–ozone treatment of the substrates, a planar Au/hole-transporting material/CH3NH3PbI3-xClx/substrate cell processed by a solution method has achieved a power conversion efficiency of over 14% and an open-circuit voltage of 1.06 V measured under reverse voltage scan. The open-circuit voltage is as high as that of our best reference cell with a TiO2 hole-blocking layer. Besides ultraviolet–ozone treatment, we find that involving Cl in the synthesis is another key for realizing high open-circuit voltage perovskite solar cells without hole-blocking layers. Our results suggest that TiO2 may not be the ultimate interfacial material for achieving high-performance perovskite solar cells.
Similar content being viewed by others
Introduction
Organic–inorganic lead halide perovskite solar cells have attracted much attention in recent years. The power conversion efficiency (PCE) of lead halide perovskite-based solar cells has rapidly increased from 3.8 to ~20.1% (certified) in just 5 years1,2,3,4,5,6,7,8. Lead halide perovskite solar cells use either the mesoscopic or the planar cell architecture. Regardless of the cell architecture, high-efficiency lead halide perovskite solar cells exclusively use hole-blocking layers (HBLs)/electron-transporting layers (ETLs) and hole-transporting layers (HTLs)/electron-blocking layers (EBLs). These intrinsic interfacial layers are believed critically necessary for achieving high open-circuit voltages (VOC’s) and efficiencies because they promote effective carrier separations and charge recombination reduction at the front contacts, typically transparent conducting oxides such as fluorine-doped tin oxide (FTO) or indium-doped tin oxide-coated glass, and back contacts, typically metals such as Ag or Au. In the inverted structures, efficient perovskite solar cells have been achieved using organic HBLs and EBLs9. In the regular structures, the most used HBL/ETL and HTL/EBL materials reported in literature are TiO2 and 2,2′,7,7′-tetrakis-(N,N-di-p-methoxyphenylamine)-9,9′-spirobifluorene (spiro-OMeTAD), respectively. A report has also shown that Y doping of TiO2 ETL can improve the cell performance6. The reason why spiro-OMeTAD is a good HTL material for achieving high-efficiency cells has been investigated10. Recently, perovskite solar cells using poly(triaryl amine) HTLs have achieved the highest certified efficiency7. Significant efforts have been paid to search for alternative HBL and HTL materials or even to eliminate HTL layer11,12,13,14. Some lead halide perovskite solar cells without HTLs have achieved reasonable conversion efficiencies. It has been shown that the remnant PbI2 layer in a CH3NH3PbI3 film prepared by a two-step method can act as a HBL15. There are many oxides, for example, ZnO, In2O3 and SnO2, that exhibit similar or even better electronic and optical properties than TiO2. Theoretically, these oxides should be as good HBL materials as TiO2. For example, perovskite solar cells using ZnO as HBL have achieved PCEs as high as 15.7%. To further understand the operation mechanism of lead halide perovskite solar cells and to further improve the cell performance, it is important to understand whether or not TiO2 is the ultimate HBL for achieving high-efficiency cells in the regular structure.
In this paper, we report on the realization of efficient planar lead halide perovskite solar cells grown directly on FTO substrates without any HBLs. By applying ultraviolet–ozone (UVO) treatment to the FTO substrates, a simple planar Au/spiro-OMeTAD/CH3NH3PbI3-xClx/FTO cell processed by a low-temperature solution method has achieved a PCE of over 14% and a VOC of 1.06 V measured under the reverse voltage scan with a scan rate of 0.1 V s−1. The VOC is as high as our best reference device using a TiO2 HBL (PCE=16.07%, VOC=1.06 V) measured with the same voltage scan and rate. The perovskite absorbers were deposited by the one-step process and there are no obvious unreacted PbI2 layers at FTO/perovskite interfaces to act as HBLs. Our results, therefore, strongly suggest that TiO2 may not be an ultimate HBL material for achieving high-efficiency lead halide perovskite solar cells. We show that involving Cl in the synthesis process and applying UVO treatment to the FTO substrates are two key factors for achieving high VOC lead halide perovskite solar cells without any HBLs. The UVO treatment improves the perovskite film coverage, and, therefore, the fill factor (FF) and the VOC. Cl may segregate to and passivate FTO/CH3NH3PbI3-xClx interfaces and, thus, improves the VOC. Our results reveal that alternative HBL materials or cell structures should be explored to further improve the performance of lead halide perovskite solar cells.
Results
Device structure
Figure 1a shows the device structure of our perovskite solar cells grown directly on FTO substrates without any HBLs. The perovskite films were directly coated on FTO substrates by a one-step spin-coating method. Following the deposition of the perovskite layer, a spiro-OMeTAD HTL (~250 nm) was deposited by spin coating. The cells were finished with thermally evaporated Au back contacts. The FTO substrates have a sheet resistance of 14 Ohms per square. Therefore, the FTO layers are the n++ electrodes. Reports in literature have shown that some lead halide perovskite solar cells behave as p-i-n cells3,16,17,18. Our lead halide perovskites thin films are highly resistive and have low carrier concentrations, typically below the sensitivity of our Hall measurement system (~1014 cm−3). Therefore, the operation mechanism for our cells may be similar to that of a p-i-n cell. Figure 1b shows the possible transportation of the photogenerated carriers and the energy band diagram of cells. The Au back contact makes good Ohmic contact to the p-type spiro-OMeTAD HTL (Supplementary Fig. 1).
(a) Schematic view of the perovskite solar cell configuration: Glass substrate, FTO front contact, CH3NH3PbI3-xClx film, spiro-OMeTAD HTL and Au back contact. (b) Energy band diagram of the FTO/CH3NH3PbI3-xClx/spiro-OMeTAD HTL/Au solar cell showing the separation and collection of photogenerated carriers.
Because halide perovskite-based solar cells typically use very thin absorber layers (300–500 nm), it is critical to form perovskite thin films with full coverages and smooth surfaces. This is particularly important for the planar cell architecture because an incomplete coverage of a perovskite film results in low-resistance shunting paths and low light absorption in the solar cell. Significant efforts have been developed to improve film coverage and crystalline quality19,20,21. We found that the residual organic species or surface–OH basis group absorbed on the FTO substrates have detrimental effects on the formation of smooth perovskite thin films. These organic contaminants make the FTO substrates somewhat hydrophobic, thereby leading to a high contact angle for the polar solvents and a low wettability for the substrates. For spin-coating depositions, these effects result in rough perovskite thin films. Even after careful chemical cleaning, the surfaces of FTO substrates still contain chemisorbed small organic species. UVO or O2 plasma treatment is widely used as a cleaning process22,23,24. We found that UVO treatment can effectively remove the residual organic species and, therefore, significantly reduce the contact angle of the polar solvents (Supplementary Fig. 2). As a result, perovskite thin films with improved smoothness and coverage were obtained. Figure 2a,b shows secondary electron micrographs (SEMs) of CH3NH3PbI3-xClx perovskite thin films grown on FTO substrates with and without UVO treatment, respectively. Based on device performance, the optimal UVO treatment time was found to be ~30 min. It is seen that the CH3NH3PbI3-xClx perovskite thin films deposited on FTO substrates with UVO treatment exhibit larger grains and a lower density of pin holes than the films deposited on FTO substrates without UVO treatment. Because of the improved surface smoothness, the spiro-OMeTAD HTL can be coated smoothly, as shown in Fig. 2c. The cross-sectional SEM image of a planar perovskite-based solar cell grown directly on FTO-coated glass is shown in Fig. 2d, revealing no compact layer, mesoporous layer or porous scaffold supporting layer. For comparison, an SEM image taken from a CH3NH3PbI3-xClx perovskite thin film deposited on a FTO substrate without UVO treatment is shown in Supplementary Fig. 3. The perovskite film is less smooth as compared with the films deposited on UVO-treated FTO substrates.
Lead halide perovskites exhibit extremely high optical absorption and high defect tolerance25,26. The spin-coated CH3NH3PbI3-xClx perovskite thin films were examined by ultraviolet–visible spectroscopy, X-ray diffraction and room temperature photoluminescence (PL). The results shown in Supplementary Fig. 4 indicate that our spin-coated CH3NH3PbI3-xClx perovskite thin films exhibit similar structural and optical properties as reported in literature3,20,21,27.
Photovoltaic performance
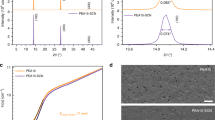
As described above, the UVO treatment of FTO substrates improves the quality of CH3NH3PbI3-xClx films, and therefore is expected to improve cell performance. Figure 3a shows the J–V curves of two typical CH3NH3PbI3-xClx-based solar cells grown on FTO substrates with (solid red curve) and without (open black curve) UVO treatment. The cell grown on FTO substrate with UVO treatment exhibited a VOC of 1.02 V, a short-circuit current (JSC) of 21.02 mA cm−2, a FF of 0.59 and a PCE of 12.72%. However, the cell fabricated with identical conditions but on a FTO substrate without UVO treatment showed a VOC of 0.96 V, a JSC of 17.93 mA cm−2, a FF of 0.45 and a PCE of 7.67%. Both cells were measured with a reverse voltage scan. Therefore, the observed differences are not caused by measurement variations. The series resistances (RS) of the cells grown on FTO substrates with and without UVO treatment are 3.90 and 7.19 Ω cm2, respectively. The shunt resistance (Rsh) of the cells grown on FTO substrates with and without UVO treatment are 900 and 360 Ω cm2, respectively. It is seen that the UVO treatment of FTO substrates improves the JSC, FF, VOC and PCE, reduces the RS and increases the Rsh. This is consistent with the observation that UVO treatment of FTO substrates improves the smoothness and coverage of CH3NH3PbI3-xClx films. The improved coverage should reduce the amount of direct contact between the FTO and the HTL, which should result in increased Rsh and carrier lifetime, and therefore VOC. Furthermore, UVO treatment has also been widely reported to increase the work functions of metal oxides28,29,30,31. It is possibly through the reduction of the concentration of oxygen vacancy and therefore a downshift of Fermi level31. It has already been confirmed that UVO treatment has an effect on electrical and photovoltaic properties of metal oxides28,29. A reduced electron concentration of the FTO surface layer is expected to lower interfacial recombination and improve VOC. These are confirmed by the VOC decay measurements, which are related to the charge carrier recombination rate and the carrier lifetime32,33. The measured VOC decay curves of two representative perovskite solar cells grown on FTO substrates with and without UVO treatment are shown in Fig. 3b. The cell grown on FTO substrate with UVO treatment exhibited a longer VOC decay time than the cell grown on FTO substrate without UVO treatment.
The thickness of the light-absorbing layer plays an important role in determining the performance of thin-film solar cells34. If the absorber layer is too thin, not enough light will be absorbed, which will lead to low current. If the absorber layer is too thick, the phtotogenerated carriers cannot be collected effectively because they must travel through the absorber layer and reach the carrier collecting layers before they recombine. We have investigated the effects of the CH3NH3PbI3-xClx film thickness on the device performance. The measured J–V curves under reverse voltage scans are shown in Fig. 4. Table 1 shows the photovoltaic parameters of perovskite solar cells with various absorber thicknesses grown on FTO substrates with UVO treatment. It is seen that both the VOC and JSC increase as the thickness of the CH3NH3PbI3-xClx film increases. However, the FF first increases then decreases as the thickness of the CH3NH3PbI3-xClx film increases. As a result, the PCE also first increases and then decreases as the thickness of the CH3NH3PbI3-xClx film increases. The optimum thickness of the perovskite CH3NH3PbI3-xClx film is ~500 nm.
It is noted that including Cl in the precursors is another key for realizing high VOC CH3NH3PbI3-xClx-based solar cells grown directly on FTO substrates without any HBLs. CH3NH3PbI3-based solar cells grown directly on FTO substrates without any HBLs have exhibited extremely poor FFs and VOC’s (Supplementary Fig. 5). The dark J–V curve indicates a poor rectify behaviour, a common feature for cells with direct contacts between absorbers and electrodes. The poor VOC is expected because holes generated in CH3NH3PbI3 will recombine with electrons from the FTO substrate. X-ray diffraction and SEM (Supplementary Fig. 6) showed that CH3NH3PbI3 thin films exhibited similar coverage and crystallinity as CH3NH3PbI3-xClx thin films. The high VOC’s seen in CH3NH3PbI3-xClx-based solar cells should, therefore, not be due to Cl-enhanced film quality. A recent theoretical study has shown that Cl may segregate to and passivate TiO2/CH3NH3PbI3-xClx interface, leading to enhanced carrier lifetime35. Passivation of interfaces at electron collecting layer and perovskite films has been found to improve the performance of perovskite solar cells36. To investigate the charge extraction process in CH3NH3PbI3-xClx and CH3NH3PbI3 films, we performed time correlated single-photon counting luminescence decays for the CH3NH3PbI3-xClx and CH3NH3PbI3 (Supplementary Fig. 7). The PL quenching measurements are related to the electron–hole diffusion lengths and lifetimes37,38. As shown in Supplementary Fig. 7, the CH3NH3PbI3-xClx film has a longer charge–carrier lifetime than the CH3NH3PbI3 film. Therefore, we speculate that the high VOC’s achieved in CH3NH3PbI3-xClx-based solar cells may be due to Cl-facilitated interface passivation. Similar effects have been observed in HCl-treated tin-doped indium oxide substrates used in organic photovoltaic cells39. Furthermore, as discussed earlier, UVO treatment to FTO substrates may also help reduce interface recombination. Cl in precursors has also been found to improve perovskite film quality such as coverage40,41. However, we did not observe significant difference on film coverage for CH3NH3PbI3 and CH3NH3PbI3-xClx films in our study. We have tried with great efforts to measure Cl concentration at FTO/CH3NH3PbI3-xClx interfaces, but no noticeable Cl has been found. We suspect that the amount of Cl present at FTO/CH3NH3PbI3-xClx interfaces could be below the detection limit of our instrument. Further investigations are needed to understand the effects of Cl.
Junction properties
The J–V curves of the best-performing CH3NH3PbI3-xClx-based solar cell grown directly on a UVO-treated FTO substrate, under illumination and in the dark, are shown in Fig. 5a. This cell has achieved a maximum PCE of 14.14% with a VOC of 1.06 V, a JSC of 19.76 mA cm−2 and a FF of 0.67 under the reverse voltage scan with a scan rate of 0.1 V s−1. It is well known that perovskite solar cells exhibit hysteresis effect, especially the cells with a planar structure36,42. We have observed similar hysteresis behaviours in our perovskite cells grown directly on FTO substrates, as shown in Supplementary Fig. 8 and Supplementary Table 1. For the same device, the VOC, JSC, FF and PCE depend on both the voltage scan direction and rate. The VOC, JSC, FF and PCE are 1.04 V, 19.78 mA cm−2, 0.65 and 13.35%, respectively, when measured under a reverse scan (from VOC to 0 V) with a scan rate of 0.1 V s−1, whereas the VOC, JSC, FF and PCE are 0.96 V, 19.97 mA cm−2, 0.52 and 10.04%, respectively, when measured under forward scan (from 0 V to VOC) with the scan rate of 0.1 V s−1. Clearly, with the same scan rate, the reverse scan resulted in higher VOC, FF and PCE than the forward scan. Furthermore, the higher scan rate resulted in a higher efficiency for the same scan direction. For example, under reverse scan, the 0.1 V s−1 scan rate showed a PCE of 13.35%, while the 0.01 V s−1 scan rate showed a PCE of 11.23%. Supplementary Fig. 9 and Supplementary Table 2 show the J–V curves of a representative HBL-free cell measured under forward and reverse voltage scans with five cycles. A stable average efficiency of 11.26% was achieved after the cell measured with five cycles. It has been demonstrated that hysteresis of perovskite solar cells can be reduced or even eliminated by special interface passivation36. Therefore, further work is needed to reduce or eliminate the hysteresis in our cells. Figure 5b shows the incident photon-to-current conversion efficiency (IPCE) spectrum of this cell. The IPCE spectrum shows efficiencies of ~80% in the 400–750-nm wavelength range, which is consistent with the ultraviolet–visible absorbance of the perovskite CH3NH3PbI3-xClx film. The dark current density–voltage curve of the solar cell based on the CH3NH3PbI3-xClx film shows good rectification characteristics (Fig. 5a).
(a) J–V curves of the best-performing cell composed of UVO-treated FTO/500 nm CH3NH3PbI3-xClx/spiro-OMeTAD/Au under AM1.5 simulated irradiation (closed red squares) and in the dark (open black squares). (b) IPCE spectrum of the best-performing cell composed of UVO-treated FTO/500 nm CH3NH3PbI3-xClx/spiro-OMeTAD/Au. (c) The linear curve of the relationship of −dV/dJ and (JSC−J)−1. (d) The plot of the relationship of ln(JSC−J) and V+RSJ. The junction properties are based on the best-performing cell. The red lines are best linear fits to the data.
Because the J–V curves show good rectification characteristics, the RS and VOC of the cell can be calculated according to the diode equation14:



where T is the absolute temperature, KB is Boltzmann constant, A is ideality factor and J0 is the reverse saturated current density14. The RS and VOC can be obtained from equations (1) and (3), respectively. Figure 5c shows a linear relationship between −dV/dJ and (JSC−J)−1. The value of RS is equal to the intercept on the y axis. The RS of the device without a conventional HBL is 4.30 Ω cm2. According to equation (2), J0 is equal to the y intercept of the line in Fig. 5d. The values of J0 and A are 1.01 × 10−4 mA cm−2 and 3.39, respectively. According to equation (3), the calculated value of the VOC is 1.068 V; this calculated value is very close to the value obtained from the J–V measurement.
Reproducibility and stability
To check the reproducibility of the performance of the simple heterojunction perovskite solar cells, we fabricated and measured 56 separate devices. Supplementary Figure 10 shows the histograms of the photovoltaic parameters. The average values of VOC and JSC are 1.00 V and 20.08 mA cm−2, respectively. Most of the devices show a PCE higher than 11%, revealing a high reproducibility. Supplementary Figure 11 shows the results of long-term stability tests of the devices without HBLs. The test was done by measuring the J–V curves of the devices after stored for a certain period of time. The tested devices were not encapsulated and were stored in atmosphere with humidity of 20% and at room temperature without light illumination. The values of FF and PCE first increased and then decreased. The values of VOC remained unchanged for a long period. The initial increase in cell performance may be attributed to oxygen-induced doping of spiro-OMeTAD, which has been reported in literature43,44. The PCE of the non-encapsulated perovskite solar cell was >70% of its initial value after 500 h (Supplementary Fig. 11).
Discussion
The highest VOC achieved with our CH3NH3PbI3-xClx-based perovskite solar cells grown directly on FTO substrates without any HBLs is 1.06 V. As the perovskite layers were deposited by the one-step spin-coat process, remnant PbI2 layers acting as HBLs can be excluded. Such a high VOC is unexpected for a solar cell grown directly on a conductive electrode. As discussed above, including Cl and applying UVO treatment to FTO substrates are the key for achieving the high VOC’s. To test if including a TiO2 HBL would further significantly improve the VOC, we have incorporated a thin layer of TiO2 on FTO substrates. Figure 6 shows the J–V curves (reverse scan) of the best-performing CH3NH3PbI3-xClx-based solar cell with a 60-nm-thick undoped TiO2 layer. The obtained VOC is ~1.06 V. It is seen that the cell efficiency has been improved. Surprisingly, however, there was no improvement on the VOC. The efficiency improvement was due to the enhancements of FF and JSC. As speculated, Cl may diffuse to and passivate FTO/CH3NH3PbI3-xClx interfaces, and lead to the observed high VOC’s for CH3NH3PbI3-xClx-based perovskite solar cells grown directly on FTO substrates without HBLs. The passivation seems to be as effective as the TiO2 HBL. Therefore, our results suggest that TiO2 may not be the ultimate HBL material for achieving high-efficiency lead halide perovskite solar cells. We anticipate that other oxide-based HBLs may also be able to produce high-efficiency lead halide perovskite solar cells and should be explored. While this paper was under review, a relevant paper was published45. It was suggested that the remnant PbI2 layer, an unavoidable residual layer for the two-step process, at FTO/CH3NH3PbI3 interface could act as a HBL.
In summary, we have demonstrated high VOC efficient planar CH3NH3PbI3-xClx-based thin-film solar cells grown directly on FTO glass substrates without any HBLs. The best-performing perovskite solar cell has achieved a PCE of 14.14% and a high VOC of 1.06 V. This VOC is as high as our best reference cell using a TiO2 HBL (PCE=16.07%, VOC=1.06 V). We have shown that UVO treatment of FTO substrates and including Cl in precursors are two key factors that ensure the high VOC for lead halide perovskite solar cells grown directly on FTO substrates without HBLs. While the actual mechanism deserves more careful study, our results strongly suggest that TiO2 may not be the ultimate HBLs for achieving high-efficiency lead halide perovskite solar cells. Our results also provide insights on how to improve the performance of lead halide perovskite solar cells using other metal oxides.
Methods
Solar cell fabrication
The perovskite CH3NH3PbI3 and CH3NH3PbI3-xClx films were synthesized by the one-step process as reported in literature6,46,47. First, 19.8 ml (0.15 mol) hydroiodic acid (Sigma-Aldrich, 57 wt.% in water, 99.99%) and 18.7 ml (0.15 mol) methylamine (Sigma-Aldrich, 33 wt.% in absolute ethanol) at a 1:1 equimolar ratio were stirred in an ice bath for 2 h. The precipitate was collected by evaporating at 50 °C for 2 h. Finally, a white powder was received by washing with diethyl ether and ethanol three times and then drying at 100 °C in a vacuum oven for 24 h. A precursor solution of CH3NH3PbI3-xClx was composed of CH3NH3I and PbCl2 (Aladdin reagent, 99.99%) with a molar ratio of 3:1. The precursor solution of CH3NH3PbI3 was composed of CH3NH3I and PbI2 (Aladdin reagent, 99.99%) with a molar ratio of 1:1. The solvent of the two precursor solutions was anhydrous dimethylformamide (Sinopharm Chemical Reagent Co., Ltd). The solutions were stirred at room temperature for 24 h. TiO2 film coated on FTO substrate was prepared by a spin-coating method and was sintered at 500 °C for 30 min (refs 48, 49). The perovskite CH3NH3PbI3 and CH3NH3PbI3-xClx films coated on FTO glasses (Asahi Glass, 14 Ohm per square) were prepared by a spin-coating method. The thicknesses of the CH3NH3PbI3-xClx films were controlled by the spin rate with a same solution. The FTO glasses were treated with UVO for 30 min. Before spin coating, the precursor solutions and the substrates were preheated at 60 °C for 15 min. The perovskite CH3NH3PbI3-xClx and CH3NH3PbI3 films were heated at 100 °C in a vacuum oven for 45 and 15 min, respectively. The hole-transporting material was composed of 68 mM spiro-OMeTAD (Shenzhen Feiming Science and Technology Co., Ltd, 99.0%), 26 mM Li-TFSI (Aladdin reagent) and 55 mM TBP (Aladdin reagent). The solvent was a mixed solution of acetonitrile and chlorobenzene with a volume ratio of 1:10. It was stirred at room temperature for 24 h and then was used to coat the perovskite films by a spin-coating method at a speed of 2,000 r.p.m. for 30 s. Device fabrication was completed by deposition of a thin gold electrode on the HTL. The active areas of the devices were controlled by a shadow mask in the thermal evaporator.
Characterization
The morphologies of perovskite solar cells were observed by a high-resolution field emission SEM (JSM 6700F). The absorption and transmission spectra of the perovskite CH3NH3PbI3-xClx film on FTO was measured by an ultraviolet–visible spectrophotometer (CARY5000, Varian, Australia) in the 400–800-nm wavelength range at room temperature. Hall measurements were conducted via a LakeShore 7704 Hall Measurement System. PL spectrum was obtained with a 532-nm laser, as the excitation source, pulsed at a frequency of 9.743 MHz. The perovskite solar cells were stored in ambient air for a short period of time and then measured under a 100 mW cm−2 (AM1.5 simulated irradiation) illumination with a standard ABET Sun 2000 Solar Simulator. The light intensity was calibrated by a standard silicon solar cell. The cell size was 2 cm × 2 cm. The active area was 0.09 cm2, which was defined by the area of the Au electrode. The J–V characteristics were measured by a CHI660D electrochemical workstation (ShangHai, China). The IPCE spectrum was measured by a Quantum Efficiency/IPCE system (PV Measurements Inc.) in the 320–800-nm wavelength range at room temperature. All the efficiencies were taken from the initial measurement, except for the test of the stability.
Additional information
How to cite this article: Ke W. et al. Efficient hole-blocking layer-free planar halide perovskite thin-film solar cells. Nat. Commun. 6:6700 doi: 10.1038/ncomms7700 (2015).
References
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009) .
Kim, H.-S. et al. Lead iodide perovskite sensitized all-solid-state submicron thin film mesoscopic solar cell with efficiency exceeding 9%. Sci. Rep. 2, 591 (2012) .
Lee, M. M., Teuscher, J., Miyasaka, T., Murakami, T. N. & Snaith, H. J. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012) .
Liu, M. Z., Johnston, M. B. & Snaith, H. J. Efficient planar heterojunction perovskite solar cells by vapour deposition. Nature 501, 395–398 (2013) .
Burschka, J. et al. Sequential deposition as a route to high-performance perovskite-sensitized solar cells. Nature 499, 316–319 (2013) .
Zhou, H. P. et al. Interface engineering of highly efficient perovskite solar cells. Science 345, 542–546 (2014) .
Jeon, N. J. et al. Compositional engineering of perovskite materials for high-performance solar cells. Nature 517, 476–480 (2015) .
National Renewable Energy Laboratory. www.nrel.gov/ncpv/images/efficiency_chart.jpg (2015) .
Shao, Y. C., Xiao, Z. G., Bi, C., Yuan, Y. B. & Huang, J. S. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nat. Commun. 5, 5784 (2014) .
Bi, D. Q., Yang, L., Boschloo, G., Hagfeldt, A. & Johansson, E. M. J. Effect of different hole transport materials on recombination in CH3NH3PbI3 perovskite-sensitized mesoscopic solar cells. J. Phys. Chem. Lett. 4, 1532–1536 (2013) .
Liu, D. Y. & Kelly, T. L. Perovskite solar cells with a planar heterojunction structure prepared using room-temperature solution processing techniques. Nat. Photon. 8, 133–138 (2013) .
Mei, A. et al. A hole-conductor–free, fully printable mesoscopic perovskite solar cell with high stability. Science 345, 295–298 (2014) .
Ku, Z. L., Rong, Y. G., Xu, M., Liu, T. F. & Han, H. W. Full printable processed mesoscopic CH3NH3PbI3/TiO2 heterojunction solar cells with carbon counter electrode. Sci. Rep. 3, 3132 (2013) .
Shi, J. J. et al. Hole-conductor-free perovskite organic lead iodide heterojunction thin-film solar cells: high efficiency and junction property. Appl. Phys. Lett. 104, 063901 (2014) .
Cao, D. H. et al. Remnant PbI2, An unforeseen necessity in high-efficiency hybrid perovskite-based solar cells. APL Mater. 2, 091101 (2014) .
Seo, J. et al. Benefits of very thin PCBM and LiF layers for solution-processed p-i-n perovskite solar cells. Energy Environ. Sci. 7, 2642–2646 (2014) .
Edri, E. et al. Why lead methylammonium tri-iodide perovskite-based solar cells require a mesoporous electron transporting scaffold (but not necessarily a hole conductor). Nano Lett. 14, 1000–1004 (2014) .
Edri, E. et al. Elucidating the charge carrier separation and working mechanism of CH3NH3PbI3-xClx perovskite solar cells. Nat. Commun. 5, 3461 (2014) .
Eperon, G. E., Burlakov, V. M., Docampo, P., Goriely, A. & Snaith, H. J. Morphological control for high performance, solution-processed planar heterojunction perovskite solar cells. Adv. Funct. Mater. 24, 151–157 (2014) .
Dualeh, A. et al. Effect of annealing temperature on film morphology of organic-inorganic hybrid pervoskite solid-state solar cells. Adv. Funct. Mater. 24, 3250–3258 (2014) .
Conings, B. et al. Perovskite-based hybrid solar cells exceeding 10% efficiency with high reproducibility using a thin film sandwich approach. Adv. Mater. 26, 2041–2046 (2014) .
Small, C. E. et al. High-efficiency inverted dithienogermole–thienopyrrolodione-based polymer solar cells. Nat. Photon. 6, 115–120 (2011) .
Hu, T. et al. Effect of UV–ozone treatment on ITO and post-annealing on the performance of organic solar cells. Synth. Met. 159, 754–756 (2009) .
Cheng, F. et al. Enhancing the performance of P3HT:ICBA based polymer solar cells using LiF as electron collecting buffer layer and UV–ozone treated MoO3 as hole collecting buffer layer. Sol. Energy Mater. Sol. Cells 110, 63–68 (2013) .
Yin, W. J., Shi, T. T. & Yan, Y. F. Unique properties of halide perovskites as possible origins of the superior solar cell performance. Adv. Mater. 26, 4653–4658 (2014) .
Yin, W. J., Shi, T. T. & Yan, Y. F. Unusual defect physics in CH3NH3PbI3 perovskite solar cell absorber. Appl. Phys. Lett. 104, 063903 (2014) .
Liang, P. W. et al. Additive enhanced crystallization of solution-processed perovskite for highly efficient planar-heterojunction solar cells. Adv. Mater. 26, 3748–3754 (2014) .
Kim, S. Y., Lee, J.-L., Kim, K.-B. & Tak, Y.-H. Effect of ultraviolet–ozone treatment of indium–tin–oxide on electrical properties of organic light emitting diodes. J. Appl. Phys. 95, 2560 (2004) .
Hains, A. W., Liu, J., Martinson, A. B. F., Irwin, M. D. & Marks, T. J. Anode interfacial tuning via electron-blocking/hole-transport layers and indium tin oxide surface treatment in bulk-heterojunction organic photovoltaic cells. Adv. Funct. Mater. 20, 595–606 (2010) .
Sugiyama, K., Ishii, H., Ouchi, Y. & Seki, K. Dependence of indium–tin–oxide work function on surface cleaning method as studied by ultraviolet and x-ray photoemission spectroscopies. J. Appl. Phys. 87, 295 (2000) .
Helander, M. G., Greiner, M. T., Wang, Z. B., Tang, W. M. & Lu, Z. H. Work function of fluorine doped tin oxide. J. Vac. Sci. Technol. A 29, 011019 (2011) .
Krishnamoorthy, T. et al. A swivel-cruciform thiophene based hole-transporting material for efficient perovskite solar cells. J. Mater. Chem. A 2, 6305–6309 (2014) .
Tao, H., Fang, G. J., Ke, W. J., Zeng, W. & Wang, J. In-situ synthesis of TiO2 network nanoporous structure on Ti wire substrate and its application in fiber dye sensitized solar cells. J. Power Sources 245, 59–65 (2014) .
Liu, D. Y., Gangishetty, M. K. & Kelly, T. L. Effect of CH3NH3PbI3 thickness on device efficiency in planar heterojunction perovskite solar cells. J. Mater. Chem. A 2, 19873–19881 (2014) .
Mosconi, E., Ronca, E. & De Angelis, F. First-principles investigation of the TiO2/organohalide perovskites interface: the role of interfacial chlorine. J. Phys. Chem. Lett. 5, 2619–2625 (2014) .
Wojciechowski, K. et al. Heterojunction modification for highly efficient organic-inorganic perovskite solar cells. ACS Nano 8, 12701–12709 (2014) .
Stranks, S. D. et al. Electron-hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science 342, 341–344 (2013) .
Roiati, V. et al. Investigating charge dynamics in halide perovskite-sensitized mesostructured solar cells. Energy Environ. Sci. 7, 1889–1894 (2014) .
Irwin, M. D. et al. Consequences of anode interfacial layer deletion. HCl-treated ITO in P3HT:PCBM-based bulk-heterojunction organic photovoltaic devices. Langmuir 26, 2584–2591 (2010) .
Tidhar, Y. et al. Crystallization of methyl ammonium lead halide perovskites: implications for photovoltaic applications. J. Am. Chem. Soc. 136, 13249–13256 (2014) .
Williams, S. T. et al. Role of chloride in the morphological evolution of organo-lead halide perovskite thin films. ACS Nano 8, 10640–10654 (2014) .
Snaith, H. J. et al. Anomalous hysteresis in perovskite solar cells. J. Phys. Chem. Lett. 5, 1511–1515 (2014) .
Cappel, U. B., Daeneke, T. & Bach, U. Oxygen-induced doping of spiro-MeOTAD in solid-state dye-sensitized solar cells and its impact on device performance. Nano Lett. 12, 4925–4931 (2012) .
Docampo, P. & Snaith, H. J. Obviating the requirement for oxygen in SnO2-based solid-state dye-sensitized solar cells. Nanotechnology 22, 225403 (2011) .
Liu, D. L., Yang, J. L. & Kelly, T. L. Compact layer free perovskite solar cells with 13.5% efficiency. J. Am. Chem. Soc. 136, 17116–17122 (2014) .
Docampo, P., Ball, J. M., Darwich, M., Eperon, G. E. & Snaith, H. J. Efficient organometal trihalide perovskite planar-heterojunction solar cells on flexible polymer substrates. Nat. Commun. 4, 2761 (2013) .
Sun, S. Y. et al. The origin of high efficiency in low-temperature solution-processable bilayer organometal halide hybrid solar cells. Energy Environ. Sci. 7, 399–407 (2014) .
Ke, W. J. et al. Perovskite solar cell with an efficient TiO2 compact film. ACS Appl. Mater. Interfaces 6, 15959–15965 (2014) .
Ke, W. J. et al. In situ synthesis of NiS nanowall networks on Ni foam as a TCO-free counter electrode for dye-sensitized solar cells. ACS Appl. Mater. Interfaces 6, 5525–5530 (2014) .
Acknowledgements
This work was supported by the National High Technology Research and Development Program (2015AA050601), the National Basic Research Program (no. 2011CB933300) of China, the National Natural Science Foundation of China (61376013, 91433203, J1210061), the Research Program of Wuhan Science and Technology Bureau (2013010501010141) and the Fundamental Research Funds for the Central Universities (2014202020207).
Author information
Authors and Affiliations
Contributions
W.K. prepared the samples and carried out the initial experiments. G.F. designed the experiments and gave supervision. Q.L., H.T. and J.W. characterized the perovskite films. J.W.W., P.Q. and H.L. analysed the data. L.X., G.Y. and M.Q. carried out the studies on device hysteresis. X.Z and Y.Y gave guidance. All authors discussed the results and contributed to the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-11 and Supplementary Tables 1-2 (PDF 1135 kb)
Rights and permissions
About this article
Cite this article
Ke, W., Fang, G., Wan, J. et al. Efficient hole-blocking layer-free planar halide perovskite thin-film solar cells. Nat Commun 6, 6700 (2015). https://doi.org/10.1038/ncomms7700
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms7700
This article is cited by
-
Development of MAPbI3.H2O and MAPbI3 perovskite solar cells using TiO2 and P3HT as charge transport layers
Journal of Materials Science: Materials in Electronics (2024)
-
Enhancement of optical, morphological and electronic properties of MoS2 thin film by annealing to improve the performance of silicon solar cells
Journal of Materials Science: Materials in Electronics (2023)
-
Improving the performance of organic lead–tin laminated perovskite solar cells from the perspective of device simulation
Optical and Quantum Electronics (2022)
-
Recent progress in electron transport bilayer for efficient and low-cost perovskite solar cells: a review
Journal of Solid State Electrochemistry (2022)
-
Perovskite Solar Cells Based on Compact, Smooth FA0.1MA0.9PbI3 Film with Efficiency Exceeding 22%
Nanoscale Research Letters (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.