Abstract
Archaeopteryx has been regarded as an icon of evolution ever since its discovery from the Late Jurassic limestone deposits of Solnhofen, Germany in 1861. Here we report the first evidence of colour from Archaeopteryx based on fossilized colour-imparting melanosomes discovered in this isolated feather specimen. Using a phylogenetically diverse database of extant bird feathers, statistical analysis of melanosome morphology predicts that the original colour of this Archaeopteryx feather was black, with 95% probability. Furthermore, reexamination of the feather's morphology leads us to interpret it as an upper major primary covert, contrary to previous interpretations. Additional findings reveal that the specimen is preserved as an organosulphur residue, and that barbule microstructure identical to that of modern bird feathers had evolved as early as the Jurassic. As in extant birds, the extensive melanization would have provided structural advantages to the Archaeopteryx wing feather during this early evolutionary stage of dinosaur flight.
Similar content being viewed by others
Introduction
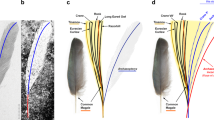
In 1861, Archaeopteryx lithographica was unearthed from the 150-million-year-old Solnhofen Lagerstätte1. The mosaic of reptilian traits (for example, teeth, long vertebral tail and manual unguals) and avian plumage in these fossils (Fig. 1a,b) has since inspired a rich scientific literature on Archaeopteryx and the origin of birds1,3, yet the animal's colour, a diverse and multifunctional trait in modern birds4,5,6, has remained only speculative. Additionally, unresolved questions have persisted regarding the anatomical identity and composition of the isolated feather specimen, which was the first described Archaeopteryx fossil7,8,9 and the only one preserved as a dark trace (Fig. 1a)1,10,11.
(a) Optical photograph of the isolated fossil feather (MB.Av.100). Scale bar: 5 mm. (b) Berlin skeletal specimen (HMN 1880), showing the ventral surface of the wings1,2. Scale bar: 5 cm. (c) Dorsal surface of left wing showing approximate location of isolated feather (in black), interpreted as a distal member of the major tract of upper primary coverts. Wing redrawn after Wellnhofer1, based on the Berlin skeleton and showing phalanges (ph I,II), primaries (I–XII), secondaries (1–14) and hypothetical reconstructions of major, median and minor tracts of upper coverts (dashed lines)1. Scale bar: 5 cm.
Similar fossilized feather traces have been found to preserve colour-imparting organelles called melanosomes12,13,14,15,16, which consist of melanins, the most widespread and abundant pigments in animals and found in the feathers of all birds (excluding albinos)4. Melanins primarily fall into two categories, eumelanins and phaeomelanins4, and the morphological properties of the associated melanosomes have been used to reconstruct various plumage colours of extinct dinosaurs12,13,14,15,16. Elucidating the phylogenetic and anatomical distributions of feather colouration can further our understanding of dinosaur evolution, palaeobiology and aesthetics.
Here we provide the first evidence of colour from Archaeopteryx, along with new information regarding the identity, preservation and microstructure of the isolated feather.
Results
Identity
The specimen is considered to be a wing feather with aerodynamic function, based on its vane asymmetry9,10,17,18, lateral curvature17 and obtusely angled distal tip10. In particular, the feather has previously been identified as a distal secondary1,10 or distal primary19. However, compared with the secondary and primary feathers of the skeletal specimens, the isolated feather is much wider relative to its length10. Furthermore, either interpretation would require that this small, moulted1,2,10 feather originated from an individual much smaller than the other known Archaeopteryx specimens2,10,20, which all represent immature individuals21,22. However, previous interpretations did not consider the relatively long calamus, which was originally visible on the specimen as a faint trace9 but has since worn away20. While the mode of feather attachment in Archaeopteryx is unknown20, comparison of the relative calamus length (22% of total feather length, 1.5 cm/6.9 cm)9 to that of modern bird feathers23 suggests that the specimen may represent a distal member of the upper major primary coverts (Fig. 1c). With respect to other wing coverts, this particular interpretation of the feather is further supported by its angled distal tip10 as well as the asymmetric and almost entirely closed pennaceous nature of its vanes23. Our interpretation is also more consistent with the relative width and small size of the feather, and would imply that the specimen was from the wing of an individual within the size range of the skeletal specimens of Archaeopteryx (presuming that the feather is conspecific1).
Preservation
We interpret the feather's dark trace to be a melanic organosulphur residue, based on the following. First, we detected no manganese among nine point analyses throughout the feather, indicating that preservation was not due to precipitation of the inorganic mineral, manganese dioxide (MnO2), as has been suggested by some workers1,2,10,24. Second, a potential organocopper biomarker for melanin was previously detected in this specimen; this biomarker has also been correlated with the presence of melanosomes in three fossil bird taxa25. We hypothesize that melanosome structures fossilize simply by virtue of being solid aggregations of melanins26,27,28, which themselves have high preservation potential29 as large, insoluble polymers resistant to degradation4. Third, the dark trace is associated with sulphur (Fig. 2), which may have derived from the sulphur-rich feather keratin23,25 and crosslinked with the melanin4; this is consistent with the sulphurization mechanism responsible for high-fidelity organic preservation in the fossil record30. Indeed, the dark trace preserves very fine details of the feather barbs and barbules (Fig. 2b).
(a) Optical photograph indicating sample locations (circles; not to scale) examined via SEM. Red-filled circles (n=2) represent locations where data were collected for statistical analysis (Supplementary Fig. S1). White-filled circles (n=4) represent locations where melanosomes were observed but not measured due to poor preservation; unfilled circles (n=17) represent locations where no melanosomes were observed. Scale bar: 5 mm. (b) Detail of boxed area (a), backscattered electron image showing barb and barbule (arrow) microstructure preserved as melanic organosulphur residue. Scale bar: 500 μm. (c) Detail of boxed area (a), spectral X-ray map for sulphur Kα; blue and black correspond to higher and lower intensities (concentrations), respectively. Scale bar: 500 μm. (d,e) SEM images taken from location of proximal red-filled circle (a), showing melanosomes (arrows) preserved three-dimensionally and as moulded imprints, respectively. Scale bars: 1 μm.
Microstructure
Barbules are also preserved as topographic impressions within the limestone matrix, and are indistinguishable from those of modern pennaceous feathers with respect to morphology23 and barbule angles31 (Fig. 3). These impressions include a parallel array of distal barbules (Fig. 3b) that contain moulded imprints of melanosomes. Other melanosome imprints are aligned along the longitudinal axis of a proximal barbule (Fig. 3c,d), an arrangement previously reported in the barbules of extant and more recent fossil bird feathers14,15.
(a) Optical photograph of boxed area within the Archaeopteryx feather (inset). Scale bar: 1 mm. (b) SEM image from (a, red arrow) showing topographic impressions of a parallel array of distal barbules and a proximal barbule (boxed area). Scale bar: 50 μm. (c) Detail of boxed area (b). Dashed black lines indicate ridges of proximal barbule; dashed red line indicates possible ridge of corresponding barb ramus. Scale bar: 10 μm. (d) Detail of boxed area (c), showing rod-shaped melanosome imprints aligned along longitudinal axis of barbule. Scale bar: 1 μm. (e,f) SEM images of barbs from a primary covert (ventral and dorsal surfaces, respectively) of American Woodcock (Scolopax minor). Arrow (f) indicates barbicel of overlapping distal barbule from adjacent barb; neighbouring distal barbules to the right have been removed to expose underlying proximal barbules. Scale bars: 100 μm (e) and 50 μm (f). (g) SEM image of longitudinal cross-section from barbule of White-breasted Nuthatch (Sitta carolinensis), showing alignment of melanosomes along longitudinal axis of barbule. Scale bar: 1 μm. Abbreviations: b, barb; db, distal barbule; pb, proximal barbule; r, rachis; rm, ramus.
Colour
Only rod-shaped eumelanosomes were observed in the Archaeopteryx feather (including melanosomes that were not measured), and are preserved both as imprints (~0.53–1.7 μm long, 180–360 nm wide, n=86) and as three-dimensional (3D) structures (~0.55–1.2 μm long, 140–280 nm wide, n=22) (Fig. 2d,e; Supplementary Table S1). Long and short axes are significantly greater in imprints than in 3D melanosomes (both P-values <0.001), as previously reported12, and the aspect ratio of imprints is significantly smaller (P-value=0.034).
Based on statistical analysis of five properties of melanosome morphology, we predict the original colour of the Archaeopteryx feather to be black, with 95% probability (Fig. 4). Separate analyses of imprints and 3D melanosomes also predict black colouration, with 91 and 75% probability, respectively.
Quadratic discriminant analysis of brown, black, grey and penguin type12 from extant birds (solid points, n=115; Supplementary Table S2) and Archaeopteryx (red-filled black circle; two unfilled black circles denote each melanosome preservation type analysed separately). Data points and 95% confidence bounds (ellipses) are shown in the colour of their respective classes; penguin type is shown in blue. Morphology of reconstructed feather is based on von Meyer (Plate VIII, Fig. 3)9; note original calamus length relative to that in Figure 1a.
Discussion
Herein we have provided the first evidence of colour from Archaeopteryx, and have demonstrated that the isolated fossil feather is most likely an upper major primary covert, exhibits modern barbule microstructure, and is preserved as an organosulphur residue. It is improbable that either of the other major classes of feather pigments, carotenoids and porphyrins, were also present in the Archaeopteryx feather, as they are relatively uncommon4 and generally do not occur in flight feathers, presumably for structural reasons23,32. Additionally, even if there were codeposition of another pigment, the black melanin would likely mask its presence4. Indeed, the melanic residue is ubiquitous throughout the feather vanes, and no banding patterns15 are visible. We do not consider minor variation in local residue abundance to be indicative of pigmentation patterns24, as light and dark areas often correspond to dark and light areas in the specimen's counterpart (BSP 1869 VIII 1), respectively (see Fig. 5.7 in Wellnhofer1); missing barbs are often preserved on the counterpart as well24. However, the residue does appear darkest at the distal tip of both this specimen (Fig. 1a) and its counterpart9, which may reflect a greater degree of melanization in this region.
The black pigmentation observed in areas of the feather not generally visible during the animal's life, such as the proximal region obscured by overlapping covert feathers (Fig. 1c) and proximal barbules obscured by overlapping distal barbules (Fig. 3c,e,f), suggests at least a partially non-visual role for this melanization23. Although black pigmentation in the wing may have had a role in the crypsis, communication and/or thermoregulation4 of Archaeopteryx, such inferences cannot be made with any certainty. Similarly, in modern birds, melanins confer protection against feather-degrading bacteria that flourish in humid habitats4,33,34, but it is uncertain whether the dry climate of the Jurassic Solnhofen1,35 would have provided sufficient evolutionary pressure to select for such an adaptation.
Nonetheless, melanins are large polymers that strongly crosslink with proteins4, and their presence substantially increases feather keratin hardness (by ~39% (ref. 36)), thickness and resistance to fracture from abrasion and cyclical loading during flight4,5,6,23,36,37. This accounts for the fact that wing feathers (and especially their distal tips, where aerodynamic forces are high) display the greatest tendency towards melanization4,5,6,38. The melanization observed throughout the Archaeopteryx wing feather would have provided similar structural advantages during this early evolutionary stage of dinosaur flight.
Methods
Fossil specimen
The isolated Archaeopteryx feather is specimen MB.Av.100, housed at the Humboldt Museum für Naturkunde in Berlin, Germany. The main slab (not analysed in this study) is BSP 1869 VIII 1, housed at the Bayerische Staatssammlung für Paläontologie und Geologie in Munich, Germany. MB.Av.100 has always been the darker of the two feather traces, although it is considered to be the counter slab owing to its original position in the rock (Schwarz-Wings, personal communication). The isolated feather was the original holotype of the nominal species, Archaeopteryx lithographica von Meyer1,8; the London specimen (BMNH 37001, housed at the Natural History Museum in London, England) has recently been designated as the neotype39.
Scanning electron microscopy
The feather specimen was examined uncoated using a Zeiss Supra 55VP field emission gun scanning electron microscope (FE-SEM) equipped with an Oxford INCA Energy 300 SEM-energy dispersive X-ray spectroscopy system, at the Carl Zeiss Nanotechnology Center in Oberkochen, Germany. Melanosome imprints were imaged using an angle-selected backscattered detector at 20.00 kV and variable vacuum pressure (Supplementary Fig. S1a–e). Melanosomes preserved three-dimensionally were imaged using a secondary electron detector at 1.80 kV and high vacuum pressure (Supplementary Fig. S1g–l); one additional image used in the analysis was acquired using a Zeiss Ultra Plus FE-SEM at 3.00 kV (Supplementary Fig. S1f). ImageJ software (http://rsbweb.nih.gov/ij/) was used to measure long and short axes of melanosomes (Supplementary Table S1).
Statistical analysis
Measurements of imprints and 3D melanosomes from the Archaeopteryx feather (Supplementary Table S1) were compared with a two-tailed Student's t-test for long and short axes (homoscedastic) and a two-tailed Welch's t-test for aspect ratios (heteroscedastic) using Microsoft Excel. Melanosome data from 115 extant bird feathers (Supplementary Table S2) were collected and analysed using quadratic discriminant analysis as previously described12,13. These samples represent a phylogenetically diverse range of 87 taxa from 27 orders, and included 56 additions that approximately doubled the sample size of the previous database. Extant bird samples were predicted to four classes (brown, black, grey and penguin type) based on five properties of melanosome morphology and with 80% cross-validation accuracy. Significant (α=0.10) discriminating variables of melanosome morphology included long axis (P-value <0.001), short axis (P-value <0.001), short axis variation (P-value <0.001), aspect ratio skew (P-value=0.008) and long axis skew (P-value=0.060). The discriminant function was highly significant (Pillai's trace, F15,324=11.24, P-value <0.001). Canonical axis 1 explained 71.32% of the variation and was most strongly associated with long axis; canonical axis 2 explained 17.23% of the variation and was most strongly associated with short axis.
Additional information
How to cite this article: Carney, R. M. et al. New evidence on the colour and nature of the isolated Archaeopteryx feather. Nat. Commun. 3:637 doi: 10.1038/ncomms1642 (2012).
References
Wellnhofer, P. Archaeopteryx, The Icon of Evolution (Dr F. Pfeil-Verlag, 2009).
Wellnhofer, P. in Feathered Dragons (eds Currie, P. J., Koppelhus, E. B., Shugar, M. A. & Wright, J. L.) 282–300 (Indiana Univ. Press, 2004).
Wellnhofer, P. A short history of research on Archaeopteryx and its relationship with dinosaurs. Geo. Soc. London 343, 237–250 (2010).
Hill, G. E. & McGraw, K. J. Bird Coloration Vol. 1 (Harvard Univ. Press, 2006).
Burtt, E. H., Jr. An analysis of physical, physiological, and optical aspects of avian coloration with emphasis on wood-warblers. Ornithol. Monogr. 38, 1–126 (1986).
Burtt, E. H. Jr. in The Behavioral Significance of Color (ed. Burtt, E. H., Jr.) 75–110 (Garland STMP Press, 1979).
von Meyer, H. Vogel-Feder und Palpipes priscus von Solenhofen. N. Jb. Geol. Paläont. Mh. Jahr. 1861, 561 (1861).
von Meyer, H. Archaeopterix lithographica (Vogel-Feder) und Pterodactylus von Solenhofen. N. Jb. Geol. Paläont. Mh. Jahr. 1861, 678–679 (1861).
von Meyer, H. Archaeopteryx lithographica aus dem lithographischen Schiefer von Solenhofen. Palaeontographica 10, 53–56 (1862).
Griffiths, P. J. The isolated Archaeopteryx feather. Archaeopteryx 14, 1–26 (1996).
Davis, P. G. & Briggs, D. E. G. Fossilization of feathers. Geology 23, 783–786 (1995).
Clarke, J. A. et al. Fossil evidence for evolution of the shape and color of penguin feathers. Science 330, 954–957 (2010).
Li, Q. et al. Plumage color patterns of an extinct dinosaur. Science 327, 1369–1372 (2010).
Vinther, J., Briggs, D. E. G., Clarke, J., Mayr, G. & Prum, R. O. Structural coloration in a fossil feather. Biol. Lett. 6, 128–131 (2010).
Vinther, J., Briggs, D. E. G., Prum, R. O. & Saranathan, V. The colour of fossil feathers. Biol. Lett. 4, 522–525 (2008).
Zhang, F. et al. Fossilized melanosomes and the colour of Cretaceous dinosaurs and birds. Nature 463, 1075–1078 (2010).
Norberg, R. A. in The Beginnings of Birds: Proceedings of the International Archaeopteryx Conference, Eichstätt 1984 (eds Hecht, M. K., Ostrom, J. H., Viohl, G. & Wellnhofer, P.) 303–318 (Freunde des Jura-Museums, 1985).
Feduccia, A. & Tardoff, H. B. Feathers of Archaeopteryx: asymmetric vanes indicate aerodynamic function. Science 203, 1021–1022 (1979).
Savile, D. B. O. The primaries of Archaeopteryx. Auk 74, 99–101 (1957).
Elzanowski, A. in Mesozoic Birds, Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 129–159 (Univ. of California Press, 2002).
Houck, M. A., Gauthier, J. A. & Strauss, R. E. Allometric scaling in the earliest fossil bird, Archaeopteryx lithographica. Science 247, 195–198 (1990).
Erickson, G. M. et al. Was dinosaurian physiology inherited by birds? Reconciling slow growth in Archaeopteryx. PLoS ONE 4, e7390 (2009).
Lucas, A. M. & Stettenheim, P. R. Avian Anatomy: Integument (US Department of Agriculture, 1972).
Tischlinger, H. & Unwin, D. UV-untersuchungen des Berliner exemplars von Archaeopteryx lithographica H. v. Meyer 1861 und der isolierten Archaeopteryx-Feder. Archaeopteryx 22, 17–50 (2004).
Wogelius, R. A. et al. Trace metals as biomarkers for eumelanin pigment in the fossil record. Science 333, 1622–1626 (2011).
Raposo, G. & Marks, M. S. The dark side of lysosome-related organelles: specialization of the endocytic pathway for melanosome biogenesis. Traffic 3, 237–248 (2002).
Delevoye, D., Giordano, F., Marks, M. S. & Raposo, G. in Melanins and Melanosomes: Biosynthesis, Biogenesis, Physiological, and Pathological Functions (eds Borovanský, J., & Riley, P. A.) 247–294 (Wiley-Blackwell, 2011).
Seiji, M., Fitzpatrick, T. B., Simpson, R. T. & Birbeck, M. S. C. Chemical composition and terminology of specialized organelles (melanosomes and melanin granules) in mammalian melanocytes. Nature 197, 1082–1084 (1963).
Hollingworth, N. T. J. & Barker, M. J. in The Processes of Fossilization (ed Donovan, S. K.) 105–119 (Columbia Univ. Press, 1991).
McNamara, M. E. et al. High-fidelity organic preservation of bone marrow in ca. 10 Ma amphibians. Geology 34, 641–644 (2006).
Carlisle, G. C. Some observations on the base: pennulum ratio and angular ratio of the barbules of the primaries in various groups of birds. Ibis 67, 908–919 (1925).
Olson, S. L. Specializations of some carotenoid-bearing feathers. Condor 72, 424–430 (1970).
Gunderson, A. R., Frame, A. M., Swaddle, J. P. & Forsyth, M. H. Resistance of melanized feathers to bacterial degradation: is it really so black and white? J. Avian Biol. 39, 539–545 (2008).
Goldstein, G. et al. Bacterial degradation of black and white feathers. Auk 121, 656–659 (2004).
de Buisonjé, P. H. in The Beginnings of Birds: Proceedings of the International Archaeopteryx Conference, Eichstätt 1984 (eds Hecht, M. K., Ostrom, J. H., Viohl, G. & Wellnhofer, P.) 45–65 (Freunde des Jura-Museums, 1985).
Bonser, R. H. C. Melanin and the abrasion resistance of feathers. Condor 97, 590–591 (1995).
Voitkevich, A. A. The Feathers and Plumage of Birds (October House, 1966).
Averill, C. K. Black wing tips. Condor 25, 57–59 (1923).
International Commission on Zoological Nomenclature. Archaeopteryx lithographica von Meyer, 1861 (Aves): conservation of usage by designation of a neotype. Bull. Zoo. Nom. 68, 230–233 (2011).
Acknowledgements
We thank D. Schwarz-Wings and the Humboldt Museum für Naturkunde Berlin for generous access to the Archaeopteryx specimen and for supplying the optical photographs used in the figures, S. Gatesy and N. Konow (Brown University) for helpful comments on the manuscript, and S. Moritz (Brown University) for translating the German references. This work was supported by a National Geographic Society Waitt grant (R.M.C.), a National Geographic Society Expeditions Council grant (R.M.C.) and United States Air Force Office of Scientific Research grant FA9550-09-1-0159 (M.D.S.).
Author information
Authors and Affiliations
Contributions
R.M.C., J.V. and M.D.S. designed the project. J.A., R.M.C., J.V., M.D.S. and L.D. collected the data. M.D.S. performed the statistical analysis. R.M.C. prepared the manuscript, and J.V., M.D.S., L.D. and J.A. edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figure S1 and Supplementary Tables S1-S2. (PDF 8490 kb)
Rights and permissions
About this article
Cite this article
Carney, R., Vinther, J., Shawkey, M. et al. New evidence on the colour and nature of the isolated Archaeopteryx feather. Nat Commun 3, 637 (2012). https://doi.org/10.1038/ncomms1642
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms1642
This article is cited by
-
Ancient skin sported intricately patterned scales
Nature (2024)
-
Evidence corroborates identity of isolated fossil feather as a wing covert of Archaeopteryx
Scientific Reports (2020)
-
Pheomelanin pigment remnants mapped in fossils of an extinct mammal
Nature Communications (2019)
-
Crossed tracks: Mesolimulus, Archaeopteryx, and the nature of fossils
Biology & Philosophy (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.