Abstract
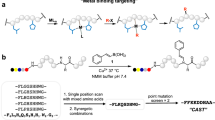
DNA–protein conjugates are important in bioanalytical chemistry, molecular diagnostics and bionanotechnology, as the DNA provides a unique handle to identify, functionalize or otherwise manipulate proteins. To maintain protein activity, conjugation of a single DNA handle to a specific location on the protein is often needed. However, preparing such high-quality site-specific conjugates often requires genetically engineered proteins, which is a laborious and technically challenging approach. Here we demonstrate a simpler method to create site-selective DNA–protein conjugates. Using a guiding DNA strand modified with a metal-binding functionality, we directed a second DNA strand to the vicinity of a metal-binding site of His6-tagged or wild-type metal-binding proteins, such as serotransferrin, where it subsequently reacted with lysine residues at that site. This method, DNA-templated protein conjugation, facilitates the production of site-selective protein conjugates, and also conjugation to IgG1 antibodies via a histidine cluster in the constant domain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Niemeyer, C. M. Semisynthetic DNA–protein conjugates for biosensing and nanofabrication. Angew. Chem. Int. Ed. 49, 1200–1216 (2010).
Stephanopoulos, N. & Francis, M. B. Choosing an effective protein bioconjugation strategy. Nature Chem. Biol. 7, 876–884 (2011).
Corey, D. R. & Schultz, P. G. Generation of a hybrid sequence-specific single-stranded deoxyribonuclease. Science 238, 1401–1403 (1987).
Saghatelian, A., Guckian, K. M., Thayer, D. A. & Ghadiri, M. R. DNA detection and signal amplification via an engineered allosteric enzyme. J. Am. Chem. Soc. 125, 344–345 (2003).
Howorka, S., Cheley, S. & Bayley, H. Sequence-specific detection of individual DNA strands using engineered nanopores. Nature Biotechnol. 19, 636–639 (2001).
Rabuka, D., Rush, J. S., deHart, G. W., Wu, P. & Bertozzi, C. R. Site-specific chemical protein conjugation using genetically encoded aldehyde tags. Nature Protoc. 7, 1052–1067 (2012).
Kazane, S. A. et al. Site-specific DNA–antibody conjugates for specific and sensitive immuno-PCR. Proc. Natl Acad. Sci. USA 109, 3731–3736 (2012).
Kazane, S. A. et al. Self-assembled antibody multimers through peptide nucleic acid conjugation. J. Am. Chem. Soc. 135, 340–346 (2013).
Netirojjanakul, C. et al. Synthetically modified Fc domains as building blocks for immunotherapy applications. Chem. Sci. 4, 266–272 (2013).
Barbuto, S. et al. Induction of innate and adaptive immunity by delivery of poly dA:dT to dendritic cells. Nature Chem. Biol. 9, 250–256 (2013).
Keppler, A. et al. A general method for the covalent labeling of fusion proteins with small molecules in vivo. Nature Biotechnol. 21, 86–89 (2003).
Los, G. V. et al. HaloTag: a novel protein labeling technology for cell imaging and protein analysis. ACS Chem. Biol. 3, 373–382 (2008).
Gautier, A. et al. An engineered protein tag for multiprotein labeling in living cells. Chem. Biol. 15, 128–136 (2008).
Niemeyer, C. M., Sano, T., Smith, C. L. & Cantor, C. R. Oligonucleotide-directed self-assembly of proteins: semisynthetic DNA–streptavidin hybrid molecules as connectors for the generation of macroscopic arrays and the construction of supramolecular bioconjugates. Nucleic Acids Res. 22, 5530–5539 (1994).
Tsukiji, S., Miyagawa, M., Takaoka, Y., Tamura, T. & Hamachi, I. Ligand-directed tosyl chemistry for protein labeling in vivo. Nature Chem. Biol. 5, 341–343 (2009).
Hughes, C. C. et al. Marinopyrrole A target elucidation by acyl dye transfer. J. Am. Chem. Soc. 131, 12094–12096 (2009).
Uchinomiya, S-H. et al. Site-specific covalent labeling of His-tag fused proteins with a reactive Ni(II)–NTA probe. Chem. Commun. 5880–5882 (2009).
Koshi, Y. et al. Target-specific chemical acylation of lectins by ligand-tethered DMAP catalysts. J. Am. Chem. Soc. 130, 245–251 (2008).
Meredith, G. D., Wu, H. Y. & Allbritton, N. L. Targeted protein functionalization using His-tags. Bioconjugate Chem. 15, 969–982 (2004).
Li, G. et al. Photoaffinity labeling of small-molecule-binding proteins by DNA-templated chemistry. Angew. Chem. Int. Ed. 52, 9544–9549 (2013).
Vinkenborg, J. L., Mayer, G. & Famulok, M. Aptamer-based affinity labeling of proteins. Angew. Chem. Int. Ed. 51, 9176–9180 (2012).
Li, X. & Liu, D. R. DNA-templated organic synthesis: nature's strategy for controlling chemical reactivity applied to synthetic molecules. Angew. Chem. Int. Ed. 43, 4848–4870 (2004).
Goodman, R. P. et al. A facile method for reversibly linking a recombinant protein to DNA. ChemBioChem 10, 1551–1557 (2009).
Snyder, T. M. & Liu, D. R. Ordered multistep synthesis in a single solution directed by DNA templates. Angew. Chem. Int. Ed. 44, 7379–7382 (2005).
Hemdan, E. S., Zhao, Y. J., Sulkowski, E. & Porath, J. Surface topography of histidine residues: a facile probe by immobilized metal ion affinity chromatography. Proc. Natl Acad. Sci. USA 86, 1811–1815 (1989).
Waldron, K. J., Rutherford, J. C., Ford, D. & Robinson, N. J. Metalloproteins and metal sensing. Nature 460, 823–830 (2009).
Aisen, P. & Listowsky, I. Iron transport and storage proteins. Annu. Rev. Biochem. 49, 357–393 (1980).
Douglas, S. M., Bachelet, I. & Church, G. M. A logic-gated nanorobot for targeted transport of molecular payloads. Science 335, 831–834 (2012).
Tran, T. N. N. et al. A universal DNA-based protein detection system. J. Am. Chem. Soc. 135, 14008–14011 (2013).
Rudchenko, M. et al. Autonomous molecular cascades for evaluation of cell surfaces. Nature Nanotech. 8, 580–586 (2013).
Jungmann, R. et al. Multiplexed 3D cellular super-resolution imaging with DNA-PAINT and Exchange-PAINT. Nature Methods 11, 313–318 (2014).
Hale, J. E. & Beidler, D. E. Purification of humanized murine and murine monoclonal antibodies using immobilized metal-affinity chromatography. Anal. Biochem. 222, 29–33 (1994).
Irving, H. & Williams, R. J. P. The stability of transition-metal complexes. J. Chem. Soc. 3192–3210 (1953).
Gatter, K. C., Brown, G., Trowbridge, I. S., Woolston, R. E. & Mason, D. Y. Transferrin receptors in human tissues: their distribution and possible clinical relevance. J. Clin. Pathol. 36, 539–545 (1983).
Qian, Z. M., Li, H., Sun, H. & Ho, K. Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacol. Rev. 54, 561–587 (2002).
Cvetkovic, A. et al. Microbial metalloproteomes are largely uncharacterized. Nature 466, 779–782 (2010).
Sliwkowski, M. X. & Mellman, I. Antibody therapeutics in cancer. Science 341, 1192–1198 (2013).
Mullard, A. Maturing antibody–drug conjugate pipeline hits 30. Nature Rev. Drug. Discov. 12, 329–332 (2013).
Kim, C. H. et al. Bispecific small molecule–antibody conjugate targeting prostate cancer. Proc. Natl Acad. Sci. USA 110, 17796–17801 (2013).
Kularatne, S. A. et al. Recruiting cytotoxic T cells to folate-receptor-positive cancer cells. Angew. Chem. Int. Ed. 52, 12101–12104 (2013).
Acknowledgements
The work was supported financially by the Danish National Research Foundation (grant number DNRF81) and Aarhus University, Faculty of Science and Technology. The authors are grateful to J. B. Knudsen for assistance in the making of the manuscript figures. The authors thank K. Stødkilde-Jørgensen for providing His6-tagged endoglycosidase H protein.
Author information
Authors and Affiliations
Contributions
C.B.R. and A.L.B.K. contributed equally to this work. C.B.R., A.L.B.K., T.T. and K.V.G. planned the research and wrote the paper. C.B.R, A.L.B.K. and T.T. developed the method, performed experiments and analysed the data. J.S.N. performed the Tf-receptor binding studies and assisted in the gel blotting. D.H.S. and A.H.O. conducted the cell studies. A.H.O. assisted in the enzymatic assay experiments. C.S. performed the MS/MS experiments. T.T., N.V.V. and K.V.G. conceived the study. J.J.E., J.K. and K.V.G. supervised the project. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
C.B.R., A.L.B.K., T.T. and K.V.G. have filed a provisional patent application regarding the current work. The other authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 29940 kb)
Rights and permissions
About this article
Cite this article
Rosen, C., Kodal, A., Nielsen, J. et al. Template-directed covalent conjugation of DNA to native antibodies, transferrin and other metal-binding proteins. Nature Chem 6, 804–809 (2014). https://doi.org/10.1038/nchem.2003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2003
This article is cited by
-
Selection of DNA-encoded chemical libraries against endogenous membrane proteins on live cells
Nature Chemistry (2021)
-
A DNA origami-based aptamer nanoarray for potent and reversible anticoagulation in hemodialysis
Nature Communications (2021)
-
Decorating bacteria with self-assembled synthetic receptors
Nature Communications (2020)
-
DNA-Scaffolded Proximity Assembly and Confinement of Multienzyme Reactions
Topics in Current Chemistry (2020)
-
Programmable Assembly of DNA-protein Hybrid Structures
Chemical Research in Chinese Universities (2020)