Abstract
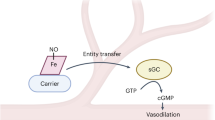
Nitric oxide (NO) is a ubiquitous, cell-permeable intercellular messenger1. The current concept assumes that NO diffuses freely through the plasma membrane2 into the cytoplasm of a target cell, where it activates its cytosolic receptor enzyme3, soluble guanylyl cyclase (sGC). Recent evidence, however, suggests that cellular membranes are not only the predominant site of calcium-dependent NO synthesis4, but also the site of its distribution and binding5. Here we extend this concept to NO signalling to show that active sGC is partially associated with the plasma membrane in a state of enhanced NO sensitivity. After cellular activation, sGC further translocates to the membrane fraction in human platelets and associates with the NO-synthase-containing caveolar fraction in rat lung endothelial cells, in a manner that is dependent on the concentration of intracellular calcium. Our data suggest that the entire NO signalling pathway is more spatially confined than previously assumed and that sGC dynamically translocates to the plasma membrane, where it is sensitized to NO.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Schmidt, H. H. H. W. & Walter, U. Cell 78, 919–925 (1994).
Lancaster, J. R. Jr Nitric Oxide 1, 18–30 (1997).
Arnold, W. P., Mittal, C. K., Katsuki, S. & Murad, F. Proc. Natl Acad. Sci. USA 74, 3203–3207 (1977).
Sessa, W. C., Barber, C. M. & Lynch, K. R. Circ. Res. 72, 921–924 (1993).
Pawloski, J. R., Hess, D. T. & Stamler, J. S. Nature 409, 622–626 (2001).
Stasch, J. P. et al. Nature 410, 212–215 (2001).
Garbers, D. L. J. Biol. Chem. 254, 240–243 (1979).
Shaw, A. W. & Vosper, A. J. J. Chem. Soc. Faraday Trans. 8, 1239 (1977).
Malinski, T. et al. Biochem. Biophys. Res. Com. 193, 1076–1082 (1993).
Pollock, J. S. et al. Proc. Natl Acad. Sci. USA 88, 10480–10484 (1991).
Garcia-Cardena, G., Oh, P., Liu, J., Schnitzer, J. E. & Sessa, W. C. Proc. Natl Acad. Sci. USA 93, 6448–6453 (1996).
Zabel, U., Weeger, M., La, M. & Schmidt, H. H. H. W. Biochem. J. 335, 51–57 (1998).
Mundel, P., Gambaryan, S., Bachmann, S., Koesling, D. & Kriz, W. Histochemistry 103, 75–79 (1995).
Markert, T. et al. J. Clin. Invest. 96, 822–830 (1995).
Schnitzer, J. E., Oh, P., Jacobson, B. S. & Dvorak, A. M. Proc. Natl Acad. Sci. USA 92, 1759–1763 (1995).
Büchler, W. A., Nakane, M. & Murad, F. Biochem. Biophys. Res. Com. 174, 351–357 (1991).
Rizzo, V., Sung, A., Oh, P. & Schnitzer, J. E. J. Biol. Chem. 273, 26323–26329 (1998).
Brouet, A., Sonveaux, P., Dessy, C., Balligand, J. L. & Feron, O. J. Biol. Chem. 276, 32663–32669 (2001).
Feussner, M., Richter, H., Baum, O. & Gossrau, R. Acta Histochem. 103, 265–277 (2001).
Russwurm, M., Wittau, N. & Koesling, D. J. Biol. Chem. 276, 44647–44652 (2001).
Whitmarsh, A. J., Cavanagh, J., Tournier, C., Yasuda, J. & Davis, R. J. Science 281, 1671–1674 (1998).
Liu, X. et al. J. Biol. Chem. 273, 18709–18713 (1998).
Verma, A., Hirsch, D. A., Glatt, C. E., Ronnett, G. V. & Snyder, S. H. Science 259, 381–384 (1993).
Pitts, B. J. R. J. Biol. Chem. 254, 6232–6235 (1979).
Zabel, U., Häusler, C., Weeger, M. & Schmidt, H. H. H. W. J. Biol. Chem. 274, 18149–18152 (1999).
McNicol, A. in Platelets: A practical approach 1–26 (eds Rickwood, D. & Hames, B. D., Oxford University Press, 1996).
Acknowledgements
We wish to thank E. Martinson for editing the manuscript, and H. Fella and M. Hoch for technical assistance. R. Venema is gratefully acknowledged for providing us with caveolin expression vectors. We thank J. Lancaster, H. Repp and K. Wingler for very helpful discussions, L. Neyses for advice with the preparation of rat heart membranes, and J. Geiger for help in determining free calcium concentrations. This work was supported by grants from the Deutsche Forschungsgemeinschaft (SFB547/C7), the Unibund Würzburg, and the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zabel, U., Kleinschnitz, C., Oh, P. et al. Calcium-dependent membrane association sensitizes soluble guanylyl cyclase to nitric oxide. Nat Cell Biol 4, 307–311 (2002). https://doi.org/10.1038/ncb775
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb775
This article is cited by
-
Pretreatment with cilnidipine attenuates hypoxia/reoxygenation injury in HL-1 cardiomyocytes through enhanced NO production and action potential shortening
Hypertension Research (2020)
-
A shear-dependent NO-cGMP-cGKI cascade in platelets acts as an auto-regulatory brake of thrombosis
Nature Communications (2018)
-
Hydrogen sulphide donors selectively potentiate a green tea polyphenol EGCG-induced apoptosis of multiple myeloma cells
Scientific Reports (2017)
-
17α-ethinyl estradiol attenuates depressive-like behavior through GABAA receptor activation/nitrergic pathway blockade in ovariectomized mice
Psychopharmacology (2016)
-
Compartmentalization of cyclic nucleotide signaling: a question of when, where, and why?
Pflügers Archiv - European Journal of Physiology (2013)