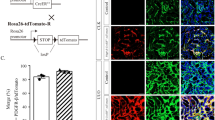
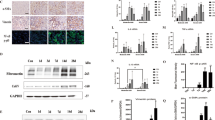
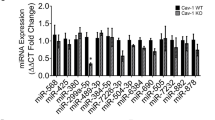
Abstract
Akt kinase is activated by transforming growth factor-β1 (TGF-β) in diabetic kidneys, and has important roles in fibrosis, hypertrophy and cell survival in glomerular mesangial cells1,2,3,4,5,6,7,8,9,10,11. However, the mechanisms of Akt activation by TGF-β are not fully understood. Here we show that TGF-β activates Akt in glomerular mesangial cells by inducing the microRNAs (miRNAs) miR-216a and miR-217, both of which target PTEN (phosphatase and tensin homologue), an inhibitor of Akt activation. These miRNAs are located within the second intron of a non-coding RNA (RP23-298H6.1-001). The RP23 promoter was activated by TGF-β and miR-192 through E-box-regulated mechanisms, as shown previously3. Akt activation by these miRs led to glomerular mesangial cell survival and hypertrophy, which were similar to the effects of activation by TGF-β. These studies reveal a mechanism of Akt activation through PTEN downregulation by two miRs, which are regulated by upstream miR-192 and TGF-β. Due to the diversity of PTEN function12,13, this miR-amplifying circuit may have key roles, not only in kidney disorders, but also in other diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yamamoto, T., Nakamura, T., Noble, N. A., Ruoslahti, E. & Border, W. A. Expression of transforming growth factor beta is elevated in human and experimental diabetic nephropathy. Proc. Natl Acad. Sci. USA 90, 1814–1818 (1993).
Sharma, K. & Ziyadeh, F. N. Hyperglycemia and diabetic kidney disease. The case for transforming growth factor-β as a key mediator. Diabetes 44, 1139–1146 (1995).
Kato, M. et al. MicroRNA-192 in diabetic kidney glomeruli and its function in TGF-beta-induced collagen expression via inhibition of E-box repressors. Proc. Natl Acad. Sci. USA 104, 3432–3437 (2007).
Kato, M. et al. Role of the Akt/FoxO3a pathway in TGF-β1-mediated mesangial cell dysfunction: a novel mechanism related to diabetic kidney disease. J. Am. Soc. Nephrol 17, 3325–3335 (2006).
Mahimainathan, L., Das, F., Venkatesan, B. & Choudhury, G. G. Mesangial cell hypertrophy by high glucose is mediated by downregulation of the tumor suppressor PTEN. Diabetes 55, 2115–2125 (2006).
Nagai, K. et al. Gas6 induces Akt/mTOR-mediated mesangial hypertrophy in diabetic nephropathy. Kidney Int. 68, 552–561 (2005).
Xin, X., Chen, S., Khan, Z. A. & Chakrabarti, S. Akt activation and augmented fibronectin production in hyperhexosemia. Am. J. Physiol. Endocrinol. Metab. 293, E1036–1044 (2007).
Yi, J. Y., Shin, I. & Arteaga, C. L. Type I transforming growth factor β receptor binds to and activates phosphatidylinositol 3-kinase. J. Biol. Chem. 280, 10870–10876 (2005).
Runyan, C. E., Schnaper, H. W. & Poncelet, A. C. The phosphatidylinositol 3-kinase/Akt pathway enhances Smad3-stimulated mesangial cell collagen I expression in response to transforming growth factor-β1. J. Biol. Chem. 279, 2632–2639 (2004).
Ghosh Choudhury, G. & Abboud, H. E. Tyrosine phosphorylation-dependent PI 3 kinase/Akt signal transduction regulates TGFβ-induced fibronectin expression in mesangial cells. Cell Signal 16, 31–41 (2004).
Wolf, G. & Ziyadeh, F. N. Molecular mechanisms of diabetic renal hypertrophy. Kidney Int. 56, 393–405 (1999).
Cully, M., You, H., Levine, A. J. & Mak, T. W. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nature Rev. Cancer 6, 184–192 (2006).
Lazar, D. F. & Saltiel, A. R. Lipid phosphatases as drug discovery targets for type 2 diabetes. Nature Rev. Drug Discov. 5, 333–342 (2006).
Stefani, G. & Slack, F. J. Small non-coding RNAs in animal development. Nature Rev. Mol. Cell Biol. 9, 219–230 (2008).
Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Sun, Y. et al. Development of a micro-array to detect human and mouse microRNAs and characterization of expression in human organs. Nucleic Acids Res. 32, e188 (2004).
Tian, Z., Greene, A. S., Pietrusz, J. L., Matus, I. R. & Liang, M. MicroRNA-target pairs in the rat kidney identified by microRNA microarray, proteomic, and bioinformatic analysis. Genome Res. 18, 404–411 (2008).
Wang, Q. et al. MicroRNA-377 is up-regulated and can lead to increased fibronectin production in diabetic nephropathy. Faseb J. 22, 4126–4135 (2008).
Landgraf, P. et al. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell 129, 1401–1414 (2007).
Hua, X., Liu, X., Ansari, D. O. & Lodish, H. F. Synergistic cooperation of TFE3 and smad proteins in TGF-β-induced transcription of the plasminogen activator inhibitor-1 gene. Genes Dev. 12, 3084–3095 (1998).
Sekido, R. et al. The delta-crystallin enhancer-binding protein delta EF1 is a repressor of E2-box-mediated gene activation. Mol. Cell Biol. 14, 5692–5700 (1994).
Comijn, J. et al. The two-handed E box binding zinc finger protein SIP1 downregulates E-cadherin and induces invasion. Mol. Cell 7, 1267–1278 (2001).
Nakagawa, Y. et al. TFE3 transcriptionally activates hepatic IRS-2, participates in insulin signaling and ameliorates diabetes. Nature Med. 12, 107–113 (2006).
Wienholds, E. et al. MicroRNA expression in zebrafish embryonic development. Science 309, 310–311 (2005).
Lim, L. P., Glasner, M. E., Yekta, S., Burge, C. B. & Bartel, D. P. Vertebrate microRNA genes. Science 299, 1540 (2003).
Xiao, C. et al. Lymphoproliferative disease and autoimmunity in mice with increased miR-17-92 expression in lymphocytes. Nature Immunol. 9, 405–414 (2008).
Elmen, J. et al. LNA-mediated microRNA silencing in non-human primates. Nature 452, 896–899 (2008).
Glass, D. J. Signalling pathways that mediate skeletal muscle hypertrophy and atrophy. Nature Cell Biol. 5, 87–90 (2003).
Salih, D. A. & Brunet, A. FoxO transcription factors in the maintenance of cellular homeostasis during aging. Curr. Opin. Cell Biol. 20, 126–136 (2008).
Braun, C. J. et al. p53-Responsive MicroRNAs 192 and 215 Are Capable of Inducing Cell Cycle Arrest. Cancer Res. 68, 10094–10104 (2008).
Gou, D., Zhang, H., Baviskar, P. S. & Liu, L. Primer extension-based method for the generation of a siRNA/miRNA expression vector. Physiol. Genomics 31, 554–562 (2007).
Bravo-Egana, V. et al. Quantitative differential expression analysis reveals miR-7 as major islet microRNA. Biochem. Biophys. Res. Commun. 366, 922–926 (2008).
Perren, A. et al. Mutation and expression analyses reveal differential subcellular compartmentalization of PTEN in endocrine pancreatic tumors compared to normal islet cells. Am. J. Pathol. 157, 1097–1103 (2000).
Omori, K. et al. Improvement of human islet cryopreservation by a p38 MAPK inhibitor. Am. J. Transplant 7, 1224–1232 (2007).
Acknowledgements
This work was supported by grants from the NIH-National Institute of Diabetes and Digestive and Kidney Diseases and the Juvenile Diabetes Research Foundation (to RN). We are grateful to M. C.-T. Hu for a generous gift of plasmids (pFRE–Luc and FoxO3a–GFP), L.Wang and J. Arizala for technical assistance and S. DaCosta for editing the manuscript.
Author information
Authors and Affiliations
Contributions
M.K. and R.N. designed research; M.K., S.P., M.W., H.Y., L.L., I.N., A.G., and I.T. performed research; M.K., M.W., Y.N, H.S. and J.J.R. contributed new reagents/analytic tools; M.K., S.P. and I.T. analysed data and M.K. and R.N. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 851 kb)
Rights and permissions
About this article
Cite this article
Kato, M., Putta, S., Wang, M. et al. TGF-β activates Akt kinase through a microRNA-dependent amplifying circuit targeting PTEN. Nat Cell Biol 11, 881–889 (2009). https://doi.org/10.1038/ncb1897
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1897