Abstract
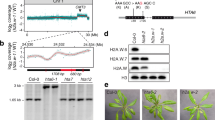
Flowering represents a crucial transition from a vegetative to a reproductive phase of the plant life cycle. Despite extensive studies, the molecular mechanisms controlling flowering remain elusive. Although the enzymes involved are unknown, methylation of histone H3 K9 and K27 correlates with repression of FLOWERING LOCUS C (FLC), an essential transcriptional repressor involved in flowering time control in Arabidopsis thaliana; in contrast, methylation of H3K4 correlates with FLC activation1,2,3,4. Here we show that loss-of-function of SET DOMAIN GROUP 8 (SDG 8), which encodes a homologue of the yeast SET2 histone methyltransferase, results in reduced dimethylation of histone H3K36, particularly in chromatin associated with the FLC promoter and the first intron, regions that contain essential cis-elements for transcription. sdg8 mutants display reduced FLC expression and flower early, establishing SDG8-mediated H3K36 methylation as a novel epigenetic memory code required for FLC expression in preventing early flowering. This is the first demonstrated role of H3K36 methylation in eukaryote development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boss, P. K., Bastow, R. M., Mylne, J. S. & Dean, C. Multiple pathways in the decision to flower: enabling, promoting, and resetting. Plant Cell 16, S18–S31 (2004).
Bastow, R. et al. Vernalization requires epigenetic silencing of FLC by histone methylation. Nature 427, 164–167 (2004).
He, Y., Doyle, M. R. & Amasino, R. M. PAF1-complex-mediated histone methylation of FLOWERING LOCUS C chromatin is required for the vernalization-responsive, winter-annual habit in Arabidopsis. Genes Dev. 18, 2774–2784 (2004).
Sung, S. & Amasino, R. M. Vernalization in Arabidopsis thaliana is mediated by the PHD finger protein VIN3. Nature 427, 159–164 (2004).
Luger, K., et al. Crystal structure of the nucleosome core particle at 2.8 A resolution. Nature 389, 251–260 (1997).
Strahl, B. D. & Allis, C. D. The language of covalent histone modifications. Nature 403, 41–45 (2000).
Sims III, R. J., Nishioka, K. & Reinberg, D. Histone lysine methylation: a signature for chromatin function. Trends Genet. 19, 629–639 (2003).
Krogan, N. J. et al. Methylation of histone H3 by Set2 in Saccharomyces cerevisiae is linked to transcriptional elongation by RNA polymerase II. Mol. Cell. Biol. 23, 4207–4218 (2003).
Xiao, T. et al. Phosphorylation of RNA polymerase II CTD regulates H3 methylation in yeast. Genes Dev. 17, 654–663 (2003).
Bannister, A. J. et al. Spatial distribution of di- and tri-methyl lysine 36 of histone H3 at active genes. J. Biol. Chem. 280, 17732–1 7736.
Strahl, B. D. et al. Set2 is a nucleosomal histone H3-selective methyltransferase that mediates transcriptional repression. Mol. Cell. Biol. 22, 1298–1306 (2002).
Rayasam, G. V. et al. NSD1 is essential for early post-implantation development and has a catalytically active SET domain. EMBO J. 22, 3153–3163 (2003).
Perry, J. & Zhao, Y. The CW domain, a structural module shared amongst vertebrates, vertebrate-infecting parasites and higher plants. Trends Biochem. Sci. 28, 576–580 (2003).
Alonso, J. M. et al. Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301, 653–657 (2003).
Michaels, S. D. & Amasino, R. M. FLOWERING LOCUS C encodes a novel MADS domain protein that acts as a repressor of flowering. Plant Cell 11, 949–956 (1999).
Sheldon, C. C. et al. The FLF MADS box gene: a repressor of flowering in Arabidopsis regulated by vernalization and methylation. Plant Cell 11, 445–458 (1999).
Lee et al. The AGAMOUS-LIKE 20 MADS domain protein integrates floral inductive pathways in Arabidopsis. Genes Dev. 14, 2366–2376 (2000).
Samach, A. et al. Distinct roles of CONSTANS target genes in reproductive development of Arabidopsis. Science 288, 1613–1616 (2000).
Fischle, W., Wang, Y. & Allis, C. D. Histone and chromatin cross-talk. Curr. Opin. Cell Biol. 15, 172–183 (2003).
He, Y., Michaels, S. D. & Amasino, R. M. Regulation of flowering time by histone acetylation in Arabidopsis. Science 302, 1751–1754 (2003).
Johnson, L. M., Cao, X. & Jacobsen, S. E. Interplay between two epigenetic marks: DNA methylation and histone H3 lysine 9 methylation. Curr. Biol. 12, 1360–1367.
Sheldon, C. C., Conn, A. B., Dennis, E. S. & Peacock, W. J. Different regulatory regions are required for the vernalization-induced repression of FLOWERING LOCUS C and for the epigenetic maintenance of repression. Plant Cell 14, 2527–2537 (2002).
Ausin, I., et al. Regulation of flowering time by FVE, a retinoblastoma-associated protein. Nature Genet. 36, 162–166 (2004).
Zhang, H. & van Nocker, S. The VERNALIZATION INDEPENDENCE 4 gene encodes a novel regulator of FLOWERING LOCUS C. Plant J. 31, 663–673 (2002).
Oh, S., Zhang, H., Ludwig, P. & van Nocker, S. A mechanism related to the yeast transcriptional regulator Paf1c is required for expression of the Arabidopsis FLC/MAF MADS box gene family. Plant Cell 16, 2940–2953 (2004).
Yu, Y., Dong, A. & Shen, W.-H. Structure-function characterization of the tobacco SET-domain protein NtSET1 reveals its role in histone methylation, chromatin binding and segregation. Plant J. 40, 699–711 (2004).
Jackson, D. in Molecular Plant Pathology: A Practical Approach (eds Bowles, D. J., Gurr, S. J. & McPherson, M.) 163–174 (Oxford University Press, Oxford, UK 1991).
Gatz, C. Novel inducible/repressible gene expression systems. Methods Cell Biol. 50, 411–424 (1995).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Acknowledgements
The authors thank the Nottingham Arabidopsis Stock Center and the Arabidopsis Biological Resource Center for seeds and BAC clone, and K. Richards and H. Huang for helpful comments on the manuscript. Z.Z. is supported by a postdoctoral fellowship from the French Ministère de la Recherche et des Nouvelles Technologies, Y.Y. by a research training fellowship from the Association Franco-Chinoise pour la Recherche Scientifique & Technique (AFCRST), and C.W. by a four-month-visiting fellowship from the United Nations Educational, Scientific and Cultural Organization (UNESCO). Research in the W.-H.S. laboratory is supported by the Centre National de la Recherche Scientifique (CNRS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1 and S2 plus Supplementary tables S1 and S2 (PDF 263 kb)
Rights and permissions
About this article
Cite this article
Zhao, Z., Yu, Y., Meyer, D. et al. Prevention of early flowering by expression of FLOWERING LOCUS C requires methylation of histone H3 K36. Nat Cell Biol 7, 1256–1260 (2005). https://doi.org/10.1038/ncb1329
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1329
This article is cited by
-
Fine mapping and analysis of candidate genes for qBT2 and qBT7.2 locus controlling bolting time in radish (Raphanus sativus L.)
Theoretical and Applied Genetics (2024)
-
SDG712, a Putative H3K9-Specific Methyltransferase Encoding Gene, Delays Flowering through Repressing the Expression of Florigen Genes in Rice
Rice (2021)
-
Genome-wide identification and expression profiling of SET DOMAIN GROUP family in Dendrobium catenatum
BMC Plant Biology (2020)
-
Bioinformatics and expression analysis of histone modification genes in grapevine predict their involvement in seed development, powdery mildew resistance, and hormonal signaling
BMC Plant Biology (2020)
-
Role of BrSDG8 on bolting in Chinese cabbage (Brassica rapa)
Theoretical and Applied Genetics (2020)