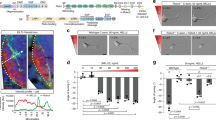
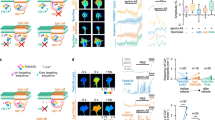
Abstract
Repulsive guidance molecule (RGM) is a recently identified protein implicated in both axonal guidance and neural tube closure1,2. The avoidance of chick RGM in the posterior optic tectum by growing temporal, but not nasal, retinal ganglion cell axons is thought to contribute to visual map formation. In contrast to ephrins, semaphorins, netrins and slits3,4, no receptor mechanism for RGM action has been defined. Here, an expression cloning strategy identified neogenin as a binding site for RGM, with a sub-nanomolar affinity. Consistent with selective axonal responsiveness to RGM, neogenin is expressed in a gradient across the chick retina. Neogenin is known to be one of several netrin-binding proteins5 but only neogenin interacts with RGM. The avoidance of RGM by temporal retinal axons is blocked by the anti-neogenin antibody and the soluble neogenin ectodomain. Dorsal root ganglion axons are unresponsive to RGM but are converted to a responsive state by neogenin expression. Thus, neogenin functions as an RGM receptor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Monnier, P.P. et al. RGM is a repulsive guidance molecule for retinal axons. Nature 419, 392–395 (2002).
Niederkofler, V., Salie, R., Sigrist, M. & Arber, S. Repulsive guidance molecule (RGM) gene function is required for neural tube closure but not retinal topography in the mouse visual system. J. Neurosci. 24, 808–818 (2004).
Tessier-Lavigne, M. & Goodman, C.S. The molecular biology of axon guidance. Science 274, 1123–1133 (1996).
Yu, T.W. & Bargmann, C.I. Dynamic regulation of axon guidance. Nature Neurosci. 4 (Suppl.), 1169–1176 (2001).
Wang, H., Copeland, N.G., Gilbert, D.J., Jenkins, N.A. & Tessier-Lavigne, M. Netrin-3, a mouse homolog of human NTN2L, is highly expressed in sensory ganglia and shows differential binding to netrin receptors. J. Neurosci. 19, 4938–4947 (1999).
Keeling, S.L., Gad, J.M. & Cooper, H.M. Mouse Neogenin, a DCC-like molecule, has four splice variants and is expressed widely in the adult mouse and during embryogenesis. Oncogene 15, 691–700 (1997).
Stein, E., Zou, Y., Poo, M. & Tessier-Lavigne, M. Binding of DCC by netrin-1 to mediate axon guidance independent of adenosine A2B receptor activation. Science 291, 1976–1982 (2001).
Keino-Masu, K. et al. Deleted in Colorectal Cancer (DCC) encodes a netrin receptor. Cell 87, 175–185 (1996).
Fazeli, A. et al. Phenotype of mice lacking functional Deleted in colorectal cancer (Dcc) gene. Nature 386, 796–804 (1997).
Srinivasan, K., Strickland, P., Valdes, A., Shin, G.C. & Hinck, L. Netrin-1/neogenin interaction stabilizes multipotent progenitor cap cells during mammary gland morphogenesis. Dev. Cell 4, 371–382 (2003).
Hong, K. et al. A ligand-gated association between cytoplasmic domains of UNC5 and DCC family receptors converts netrin-induced growth cone attraction to repulsion. Cell 97, 927–941 (1999).
Geisbrecht, B.V., Dowd, K.A., Barfield, R.W., Longo, P.A. & Leahy, D.J. Netrin binds discrete subdomains of DCC and UNC5 and mediates interactions between DCC and heparin. J. Biol. Chem. 278, 32561–32568 (2003).
Gad, J.M., Keeling, S.L., Shu, T., Richards, L.J. & Cooper, H.M. The spatial and temporal expression patterns of netrin receptors, DCC and neogenin, in the developing mouse retina. Exp. Eye Res. 70, 711–722 (2000).
Vielmetter, J., Kayyem, J.F., Roman, J.M. & Dreyer, W.J. Neogenin, an avian cell surface protein expressed during terminal neuronal differentiation, is closely related to the human tumor suppressor molecule deleted in colorectal cancer. J. Cell Biol. 127, 2009–2020 (1994).
Sheppard, A.M., Konopka, M. & Jeffrey, P.L. Thy-1 expression in the retinotectal system of the chick. Brain Res. 471, 49–60 (1988).
Sheppard, A.M., Konopka, M., Robinson, S.R., Morgan, I.G. & Jeffrey, P.L. Thy-1 antigen is specific to ganglion cells in chicks. Neurosci. Lett. 123, 87–90 (1991).
Walter, J., Henke-Fahle, S. & Bonhoeffer, F. Avoidance of posterior tectal membranes by temporal retinal axons. Development 101, 909–913 (1987).
Walter, J., Kern-Veits, B., Huf, J., Stolze, B. & Bonhoeffer, F. Recognition of position-specific properties of tectal cell membranes by retinal axons in vitro. Development 101, 685–696 (1987).
Feldheim, D.A. et al. Genetic analysis of ephrin-A2 and ephrin-A5 shows their requirement in multiple aspects of retinocollicular mapping. Neuron 25, 563–574 (2000).
Muller, B.K., Jay, D.G. & Bonhoeffer, F. Chromophore-assisted laser inactivation of a repulsive axonal guidance molecule. Curr. Biol. 6, 1497–1502 (1996).
Ming, G.L. et al. cAMP-dependent growth cone guidance by netrin-1. Neuron 19, 1225–1235 (1997).
Fournier, A.E., GrandPre, T. & Strittmatter, S.M. Identification of a receptor mediating Nogo-66 inhibition of axonal regeneration. Nature 409, 341–346 (2001).
Nakamura, F., Tanaka, M., Takahashi, T., Kalb, R.G. & Strittmatter, S.M. Neuropilin-1 extracellular domains mediate semaphorin D/III-induced growth cone collapse. Neuron 21, 1093–1100 (1998).
Liu, B.P., Fournier, A., GrandPre, T. & Strittmatter, S.M. Myelin-associated glycoprotein as a functional ligand for the Nogo-66 receptor. Science 297, 1190–1193 (2002).
Hornberger, M.R. et al. Modulation of EphA receptor function by coexpressed ephrinA ligands on retinal ganglion cell axons. Neuron 22, 731–742 (1999).
Vielmetter, J., Stolze, B., Bonhoeffer, F. & Stuermer, C.A. In vitro assay to test differential substrate affinities of growing axons and migratory cells. Exp. Brain Res. 81, 283–287 (1990).
Brinks, H. et al. The repulsive guidance molecule RGMa is involved in the formation of afferent connections in the dentate gyrus. J. Neurosci. 24, 3862–3869 (2004).
Acknowledgements
We thank R. Nordin for technical assistance, R. Salie, V. Niederkofler and S. Arber for providing the mouse RGM-A–AP expression vector and Peter L. Jeffrey for anti-Thy-1 antibody. This work is supported by a grant from the National Institutes of Health to S.M.S and by a BioChance grant from the Bundesministerium für Bildung und Forschung to Migragen. S.M.S. is an investigator of the Patrick and Catherine Weldon Donaghue Medical Research Foundation. A.C. is supported by Retina France, the Schlumberger foundation and Association pour la Recherche sur le Cancer (ARC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that two of the authors, Bernhard Mueller and Lutz Deitinghoff, were employed and supported by a commercial venture, Migragen AG, during the course of this work. Currently, both are employed by Abbott GmbH&CO.
Supplementary information
Rights and permissions
About this article
Cite this article
Rajagopalan, S., Deitinghoff, L., Davis, D. et al. Neogenin mediates the action of repulsive guidance molecule. Nat Cell Biol 6, 756–762 (2004). https://doi.org/10.1038/ncb1156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1156
This article is cited by
-
Molecular versatility during pluripotency progression
Nature Communications (2023)
-
Neogenin suppresses tumor progression and metastasis via inhibiting Merlin/YAP signaling
Cell Death Discovery (2023)
-
Transcriptomic characterization of the molecular mechanisms induced by RGMa during skeletal muscle nuclei accretion and hypertrophy
BMC Genomics (2022)
-
Increase in repulsive guidance molecule-a (RGMa) in lacunar and cortical stroke patients is related to the severity of the insult
Scientific Reports (2022)
-
Repulsive guidance molecule acts in axon branching in Caenorhabditis elegans
Scientific Reports (2021)