Abstract
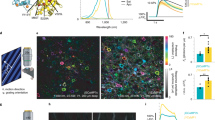
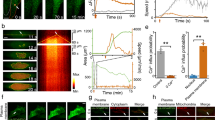
Studies of protein function would be facilitated by a general method to inactivate selected proteins in living cells noninvasively with high spatial and temporal precision. Chromophore-assisted light inactivation (CALI)1 uses photochemically generated, reactive oxygen species to inactivate proteins acutely, but its use has been limited by the need to microinject dye-labeled nonfunction-blocking antibodies. We now demonstrate CALI of connexin43 (Cx43) and α1C L-type calcium channels, each tagged with one or two small tetracysteine (TC) motifs2 that specifically bind the membrane-permeant, red biarsenical dye, ReAsH3,4. ReAsH-based CALI is genetically targeted, requires no antibodies or microinjection, and inactivates each protein by ∼90% in <30 s of widefield illumination. Similar light doses applied to Cx43 or α1C tagged with green fluorescent protein (GFP) had negligible to slight effects with or without ReAsH exposure, showing the expected molecular specificity. ReAsH-mediated CALI acts largely via singlet oxygen because quenchers or enhancers of singlet oxygen respectively inhibit or enhance CALI.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Jay, D.G. Selective destruction of protein function by chromophore-assisted laser inactivation. Proc. Natl. Acad. Sci. USA 85, 5454–5458 (1988).
Griffin, B.A., Adams, S.R. & Tsien, R.Y. Specific covalent labeling of recombinant protein molecules inside live cells. Science 281, 269–272 (1998).
Adams, S.R. et al. New biarsenical ligands and tetracysteine motifs for protein labeling in vitro and in vivo: synthesis and biological applications. J. Am. Chem. Soc. 124, 6063–6076 (2002).
Gaietta, G. et al. Multicolor and electron microscopic imaging of connexin trafficking. Science 296, 503–507 (2002).
Gu, Z. et al. Role of duplicate genes in genetic robustness against null mutations. Nature 421, 63–66 (2003).
Jay, D.G. & Sakurai, T. Chromophore-assisted laser inactivation (CALI) to elucidate cellular mechanisms of cancer. Biochim. Biophys. Acta. 1424, M39–M48 (1999).
Liao, J.C., Roider, J. & Jay, D.G. Chromophore-assisted laser inactivation of proteins is mediated by the photogeneration of free radicals. Proc. Natl. Acad. Sci. USA 91, 2659–2663 (1994).
Surrey, T. et al. Chromophore-assisted light inactivation and self-organization of microtubules and motors. Proc. Natl. Acad. Sci. USA 95, 4293–4298 (1998).
Beck, S. et al. Fluorophore-assisted light inactivation: a high-throughput tool for direct target validation of proteins. Proteomics 2, 247–255 (2002).
Marek, K.W. & Davis, G.W. Transgenically encoded protein photoinactivation (FlAsH-FALI): acute inactivation of synaptotagmin I. Neuron 36, 805–813 (2002).
Yeager, M. & Nicholson, B.J. Structure of gap junction intercellular channels. Curr. Opin. Struct. Biol. 6, 183–192 (1996).
Falk, M.M. Connexin-specific distribution within gap junctions revealed in living cells. J. Cell Sci. 113(Pt 22), 4109–4120 (2000).
Campbell, R.E. et al. A monomeric red fluorescent protein. Proc. Natl. Acad. Sci. USA 99, 7877–7882 (2002).
Foote, C.S. & Clennan, E.L. Properties and reactions of singlet dioxygen. Struct. Energ. React. Chem. Ser. 2, 105–140 (1995).
Rajfur, Z., Roy, P., Otey, C., Romer, L. & Jacobson, K. Dissecting the link between stress fibres and focal adhesions by CALI with EGFP fusion proteins. Nat. Cell Biol. 4, 286–293 (2002).
Parrinello, S. et al. Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat. Cell Biol. 5, 741–747 (2003).
Moan, J. On the diffusion length of singlet oxygen in cells and tissues. J. Photochem. Photobiol. B. Biol. 6, 343–347 (1990).
Baker, A. & Kanofsky, J.R. Quenching of singlet oxygen by biomolecules from L1210 leukemia cells. Photochem. Photobiol. 55, 523–528 (1992).
Piedras-Renteria, E.S. et al. Increased expression of alpha(1A) Ca2+ channel currents arising from expanded trinucleotide repeats in spinocerebellar ataxia type 6. J. Neurosci. 21, 9185–9193 (2001).
Acknowledgements
We wish to thank M.M. Falk for providing the Cx43-GFP construct, M.H. Ellisman for providing the Cx43-TC construct and Richard W. Tsien for critical reading of the manuscript. This work was supported by the Howard Hughes Medical Institute, Department of Energy contract DE-AC03-76SF00098 and National Institutes of Health grant NS27177 to R.Y.T. S.R.A. was supported in part by the National Institutes of Health grant PO1 DK54441 (to S.S. Taylor).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Tour, O., Meijer, R., Zacharias, D. et al. Genetically targeted chromophore-assisted light inactivation. Nat Biotechnol 21, 1505–1508 (2003). https://doi.org/10.1038/nbt914
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt914
This article is cited by
-
Photodynamic treatment of pathogens
La Rivista del Nuovo Cimento (2022)
-
Optical control of neuronal ion channels and receptors
Nature Reviews Neuroscience (2019)
-
Pericyte Plasticity in the Brain
Neuroscience Bulletin (2019)
-
Chromophore-Assisted Light Inactivation of the V-ATPase V0c Subunit Inhibits Neurotransmitter Release Downstream of Synaptic Vesicle Acidification
Molecular Neurobiology (2019)
-
Green monomeric photosensitizing fluorescent protein for photo-inducible protein inactivation and cell ablation
BMC Biology (2018)