Abstract
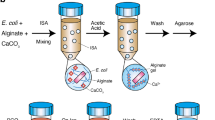
Isolating hyperproducing cells is important in biotechnology, but these cells usually grow slowly and can be overgrown by poorly producing cells. We describe a new method of isolating slowly growing cells from among rapidly growing cells, which has the potential for automation and high throughput (e.g., 100,000 cells/h). A model system is presented consisting of a mixed population of slowly growing mutant and rapidly growing wild-type yeast, which were encapsulated in double agarose gel microdrops (dGMDs); with most dGMDs initially containing single cells. Double encapsulation locates parent cells near dGMD centers, making microcolony measurement more accurate. After a 15-h incubation, fluorescent activated cell sorting was used to analyze and sort dGMDs with small microcolonies (slow growers) from dGMDs with large microcolonies (rapid growers). Successful isolation of slow growers from a mixed population of predominantly rapidly growing Saccharomyces cerevisiae cells was achieved.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ozturk, S.S. and Palsson, B.O. 1990. Loss of antibody productivity during long-term cultivation of a hybridoma cell line in low serum and serum free media. Hybridoma 9: 167–175.
McKinney, K.L., Dilwith, R., and Belfort, G. 1991. Manipulation of heterogeneous hybridoma cultures for overproduction of monoclonal antibodies. Biotech. Prog. 7: 445–454.
Backus, M.P. and Stauffer, J.F. 1955. The production and selection of a family of strains in Penicillim chrysogenum. Mycologia 47: 429–563.
AI-Rubeai, M. and Emery, A.N. 1993. Flow cytometry in animal cell culture. Bio/Technology 11: 572–579.
Shapiro, H.M. 1994. Practical flow cytometry ed. 3, A.R. Liss, New York.
Weaver, J.C., Bliss, J.G., Harrison, G.I., Powell, K.T., and Williams, G.B. 1991. Microdrop technology: a general method for separating cells by function and composition. Methods 2: 234–247.
Weaver, J.C., Bliss, J.G., Powell, K.T., Harrison, G.I., and Williams, G.B. 1991. Rapid clonal growth measurements at the single-cell level: gel microdroplets and flow cytometry. Bio/Technology 9: 873–877.
Sahar, E., Nir, R. and Lamed, R. 1994. Flow cytometric analysis of entire micro-colonies. Cytometry 15: 213–221.
Ryan, C., Nguyen, B-T., and Sullivan, S.J. 1995. A rapid assay for mycobacterial growth and antibiotic sensitivity using gel microdrop encapsulation. J. Clin. Microbiol. 33: 1720–1726.
Weaver, J.C., Seissler, P.E., Threefoot, S.A., Lorenz, J.W., Huie, T., Rodrigues, R., et al. 1984. Microbiological measurements by immobilization of cells within small volume elements. Ann. NY Acad. Sci. 434: 363–372.
Weaver, J.C., Williams, G.B., Klibanov, A.M., and Demain, A.L. 1988. Gel microdroplets: rapid detection and enumeration of individual microorganisms by their metabolic activity. Bio/Technology 6: 1084–1089.
Powell, K.T. and Weaver, J.C. 1990. Gel microdroplets and flow cytometry: rapid determination of antibody secretionby individual cells within a cell population. Bio/Technology 8: 333–337.
Gray, F., Kenney, J.S., and Dunne, J.F. 1995. Secretion capture and report web: use of affinity derived agarose microdroplets for the selection of hybridoma cells. J. Imm. Meth. 182: 155–163.
Kenney, J.S., Gray, F., Ancel, M-H., and Dunne, J.F. 1995. Production of monoclonal antibodies using a secretion capture and report web. Bio/Technology 13: 787–790.
Weaver, J.C., Dunne, J.F., Gray, F., Lazzari, K.G., and Lin, J.B. 1995. Gel microdrop assays for antibody secretion by hybridoma cells, pp. 39–62 in Flow cytometry applications in cell culture. AI-Rubeai, M. and Emery, A.N. (eds.), Marcel Dekker, New York.
Lazzari, K., Nguyen, A., Abebe, J., Mac, I., Chang, A., Lin, J.B., et al. 1995. Encapsulation and stabilization of chromsomes in gel microdrops. Cytometry 21: 111–119.
Melamed, E.R., Lindmo, T., and Mendelsohn, M.L. (eds.). 1990. Flow cytometry and sorting, ed. 2, Wiley-Liss, New York.
Givan, A.L. 1992. Flow cytometry: first principles, Wiley-Liss, New York.
Haugland, R.P. 1992. Handbook of fluorescent probes and research chemicals, molecular probes, Eugene,OR.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gift, E., Park, H., Paradis, G. et al. FACS-based isolation of slowly growing cells: Double encapsulation of yeast in gel microdrops. Nat Biotechnol 14, 884–887 (1996). https://doi.org/10.1038/nbt0796-884
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0796-884
This article is cited by
-
FACS-Based Functional Protein Screening via Microfluidic Co-encapsulation of Yeast Secretor and Mammalian Reporter Cells
Scientific Reports (2020)
-
Applications of cell sorting in biotechnology
Microbial Cell Factories (2006)
-
A non-invasive crop ideotype to reduce invasive potential
Euphytica (2006)
-
Gel microdrop technology for rapid isolation of rare and high producer cells
Nature Medicine (1997)