Abstract
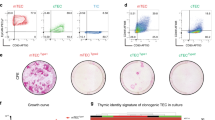
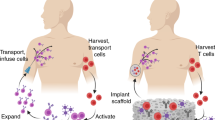
Biocompatible inorganic matrices have been used to enhance bone repair by integrating with endogenous bone architecture. Hypothesizing that a three-dimensional framework might support reconstruction of other tissues as well, we assessed the capacity of a tantalum-coated carbon matrix to support reconstitution of functioning thymic tissue. We engineered a thymic organoid by seeding matrices with murine thymic stroma. Co-culture of human bone marrow-derived hematopoietic progenitor cells within this xenogeneic environment generated mature functional T cells within 14 days. The proportionate T-cell yield from this system was highly reproducible, generating over 70% CD3+ T cells from either AC133+ or CD34+ progenitor cells. Cultured T cells expressed a high level of T-cell receptor excision circles (TREC), demonstrating de novo T lymphopoiesis, and function of fully mature T cells. This system not only facilitates analysis of the T-lymphopoietic potential of progenitor cell populations; it also permits ex vivo genesis of T cells for possible applications in treatment of immunodeficiency.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bobyn, J.D., Stackpool, G.J., Hacking, S.A., Tanzer, M. & Krygier, J.J. Characteristics of bone ingrowth and interface mechanics of a new porous tantalum biomaterial. J. Bone Joint Surg. Br. 81, 907–914 (1999).
Black, J. Biological performance of tantalum. Clin. Mater. 16, 167–173 (1994).
Boyd, R.L. et al. The thymic microenvironment. Immunol. Today 14, 445–459 (1993).
Van Vliet, E., Melis, M. & Van Ewijk, W. Monoclonal antibodies to stromal cell types of the mouse thymus. Eur. J. Immunol. 14, 524–529 (1984).
Van Vilet, E., Jenkinson, E.J., Kingston, R., Owen, J.J. & Van Ewijk, W. Stromal cell types in the developing thymus of the normal and nude mouse embryo. Eur. J. Immunol. 15, 675–681 (1985).
van Ewijk, W. Cell surface topography of thymic microenvironments. Lab. Invest. 59, 579–590 (1988).
van Ewijk, W. T-cell differentiation is influenced by thymic microenvironments. Annu. Rev. Immunol. 9, 591–615 (1991).
Freedman, A.R., Zhu, H., Levine, J.D., Kalams, S. & Scadden, D.T. Generation of human T lymphocytes from bone marrow CD34+ cells in vitro. Nat. Med. 2, 46–51 (1996).
Gardner, J.P. et al. T-lymphopoietic capacity of cord blood-derived CD34+ progenitor cells. Exp. Hematol. 26, 991–999 (1998).
Pawelec, G., Muller, R., Rehbein, A., Hahnel, K. & Ziegler, B.L. Extrathymic T cell differentiation in vitro from human CD34+ stem cells. J. Leukoc. Biol. 64, 733–739 (1998).
Galy, A., Verma, S., Barcena, A. & Spits, H. Precursors of CD3+CD4+CD8+ cells in the human thymus are defined by expression of CD34. Delineation of early events in human thymic development. J. Exp. Med. 178, 391–401 (1993).
Anderson, G., Jenkinson, E.J., Moore, N.C. & Owen, J.J. MHC class II-positive epithelium and mesenchyme cells are both required for T-cell development in the thymus. Nature 362, 70–73 (1993).
Barcena, A. et al. Phenotypic and functional analysis of T-cell precursors in the human fetal liver and thymus: CD7 expression in the early stages of T- and myeloid-cell development. Blood 82, 3401–3414 (1993).
Barcena, A. et al. Lymphoid and myeloid differentiation of fetal liver CD34+ lineage- cells in human thymic organ culture. J. Exp. Med. 180, 123–132 (1994).
Fisher, A.G. et al. Human thymocyte development in mouse organ cultures. Int. Immunol. 2, 571–578 (1990).
Yeoman, H. et al. Human bone marrow and umbilical cord blood cells generate CD4+ and CD8+ single-positive T cells in murine fetal thymus organ culture. Proc. Natl. Acad. Sci. USA 90, 10778–10782 (1993).
Res, P. et al. CD34+CD38dim cells in the human thymus can differentiate into T, natural killer, and dendritic cells but are distinct from pluripotent stem cells. Blood 87, 5196–5206 (1996).
Plum, J., De Smedt, M., Defresne, M.P., Leclercq, G. & Vandekerckhove, B. Human CD34+ fetal liver stem cells differentiate to T cells in a mouse thymic microenvironment. Blood 84, 1587–1593 (1994).
Shortman, K., Egerton, M., Spangrude, G.J. & Scollay, R. The generation and fate of thymocytes. Semin. Immunol. 2, 3–12 (1990).
van Ewijk, W. et al. Thymic microenvironments, 3-D versus 2-D? Semin. Immunol. 11, 57–64 (1999).
Fujimoto, S. & Yamagishi, H. Isolation of an excision product of T-cell receptor alpha-chain gene rearrangements. Nature 327, 242–243 (1987).
Okazaki, K., Davis, D.D. & Sakano, H. T cell receptor beta gene sequences in the circular DNA of thymocyte nuclei: direct evidence for intramolecular DNA deletion in V-D-J joining. Cell 49, 477–485 (1987).
Livak, F. & Schatz, D.G. T-cell receptor alpha locus V(D)J recombination by-products are abundant in thymocytes and mature T cells. Mol. Cell Biol. 16, 609–618 (1996).
Takeshita, S., Toda, M. & Yamagishi, H. Excision products of the T cell receptor gene support a progressive rearrangement model of the alpha/delta locus. EMBO J. 8, 3261–3270 (1989).
Douek, D.C. et al. Changes in thymic function with age and during the treatment of HIV infection. Nature 396, 690–695 (1998).
Genevee, C. et al. An experimentally validated panel of subfamily-specific oligonucleotide primers (V alpha 1-w29/V beta 1-w24) for the study of human T cell receptor variable V gene segment usage by polymerase chain reaction. Eur. J. Immunol. 22, 1261–1269 (1992).
Zhang, X.M., Cathala, G., Soua, Z., Lefranc, M.P. & Huck, S. The human T-cell receptor gamma variable pseudogene V10 is a distinctive marker of human speciation. Immunogenetics 43, 196–203 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poznansky, M., Evans, R., Foxall, R. et al. Efficient generation of human T cells from a tissue-engineered thymic organoid. Nat Biotechnol 18, 729–734 (2000). https://doi.org/10.1038/77288
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/77288
This article is cited by
-
Lateral plate mesoderm cell-based organoid system for NK cell regeneration from human pluripotent stem cells
Cell Discovery (2022)
-
Engineered hydrogels for mechanobiology
Nature Reviews Methods Primers (2022)
-
The role of the thymus in allogeneic bone marrow transplantation and the recovery of the peripheral T-cell compartment
Seminars in Immunopathology (2021)
-
Recent advances in chemically defined and tunable hydrogel platforms for organoid culture
Bio-Design and Manufacturing (2021)
-
Designing natural and synthetic immune tissues
Nature Materials (2018)