Abstract
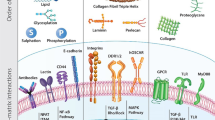
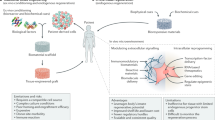
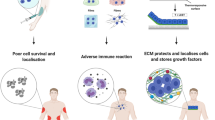
Biomaterials play a pivotal role in field of tissue engineering. Biomimetic synthetic polymers have been created to elicit specific cellular functions and to direct cell-cell interactions both in implants that are initially cell-free, which may serve as matrices to conduct tissue regeneration, and in implants to support cell transplantation. Biomimetic approaches have been based on polymers endowed with bioadhesive receptor-binding peptides and mono- and oligosaccharides. These materials have been patterned in two-and three-dimensions to generate model multicellular tissue architectures, and this approach may be useful in future efforts to generate complex organizations of multiple cell types. Natural polymers have also played an important role in these efforts, and recombinant polymers that combine the beneficial aspects of natural polymers with many of the desirable features of synthetic polymers have been designed and produced. Biomaterials have been employed to conduct and accelerate otherwise naturally occurring phenomena, such as tissue regeneration in wound healing in the otherwise healthy subject; to induce cellular responses that might not be normally present, such as healing in a diseased subject or the generation of a new vascular bed to receive a subsequent cell transplant; and to block natural phenomena, such as the immune rejection of cell transplants from other species or the transmission of growth factor signals that stimulate scar formation. This review introduces the biomaterials and describes their application in the engineering of new tissues and the manipulation of tissue responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Langer, R., and Vacanti, J.P. 1993. Tissue engineering. Science 260: 920–926.
Peppas, N.A., and Langer, R. 1994. Challenges in biomaterials. Science 263: 1715–1720.
Andrade, J.D., and Hlady, V. 1986. Protein adsorption and materials biocom-patibility: a tutorial review and suggested hypotheses. Adv. Polym. Sci. 79: 1–63.
Norde, W. 1994. Protein adsorption at solid surfaces: a thermodynamic approach. Pure Appl. Chem. 66: 491–496.
Buck, C., and Horwitz, A.F. 1987. Cell surface receptors for extracellular matrix molecules. Annu. Rev. Cell Biol. 3: 179–205.
Singer, I.I., Kawka, D.W., Scott, S., Mumford, R.A., and Lark, M.W. 1987. The fibomectin cell attachment sequence Arg-Gly-Asp-Ser promotes folcal contact formation during early fibroblast attachment and spreading. J. Cell Biol. 104: 573–584.
Juliano, R.L., and Haskill, S. 1993. Signal transduction from the extracellular matrix. J. Cell Biol. 120: 577–585.
Singer, I.I., Scott, S., Kawka, D.W., Kazazis, D.M., Gailit, J., and Ruoslahti, E. 1988. Cell surface distribution of fibronectin and vitronectin receptors depends on substrate composition and extracellular matrix accumulation. J. Cell Biol. 106: 2171–2182.
Mosher, D.F. 1993. Assembly of fibronectin into extracellular matrix. Curr. Opin. Struct. Biol. 3: 214–222.
Hynes, R.O. 1992. Integrins: versatility, modulation and signaling in cell adhesion. Cell 69: 11–25.
Yamada, K.M. 1991. Adhesive recognition sequences. J. Biol. Chem. 266: 12809–12812.
Humphries, M.J. 1990. The molecular basis and specificity of integrin-ligand interactions. J. Cell Sci. 97: 585–592.
Graf, J., Ogle, R.C., Robey, F.A., Sasaki, M., Martin, G.R., Yamada, Y., and Kleinman, H.K. 1987. A pentapeptide from the laminin Bl chain that mediates cell adhesion and binds the 67000 laminin receptor. Biochemistry 26: 6896–6900.
Mecham, R.P. 1991. Laminin receptors. Annu. Rev. Cell Biol. 7: 71–91.
Ruoslahti, E., and Pierschbacher, M. 1986. Tetrapeptides useful in surgery and therapeutic reconstruction and treatment of injuries. U.S. Patent 4,578,079.
Massia, S.P., and Hubbell, J.A. 1990. Covalently grafted ROD- and YIGSR-containing synthetic peptides support receptor-mediated adhesion of cultured fibroblasts. Anal. Biochem. 187: 292–301.
Massia, S.P., and Hubbell, J.A. 1991. An RGD spacing of 440 nm is sufficient for integrin αvβ3-mediated fibroblast spreading and 140 nm for focal contact and stress fiber formation. J. Cell Biol. 114: 1089–1100.
Massia, S.P., Rao, S.S., and Hubbell, J.A. 1993. Covalently immobilized laminin peptide Tyr-Ile-Gly-Ser-Arg (YIGSR) supports cell spreading and co-localization of the 67-kilodalton laminin receptor with ot-actinin and vinculin. J. Biol. Chem. 268: 8053–8059.
DiMilla, P.A., Stone, J.A., Quinn, J.A., Albelda, S.M., and Lauffenberger, D.A. 1993. Maximal migration of human smooth muscle cells on fibronectin and type IV collagen occurs at an intermediate attachment strength. J. Cell Biol. 122: 729–737.
Kygourakis, K., Bizioz, R., and Markenscoff, P. 1991. Proliferation of anchorage-dependent contact-inhibited cells, 1: development of theoretical models based on cellular automata. Biotechnol. Bioeng. 38: 459–470.
Zygourakis, K., Markenscoff, P., and Bizios, R. 1991. Proliferation of anchorage-dependent contact-inhibited cells, 2: experimental results and validation of the theoretical models. Biotechnol. Bioeng. 38: 471–479.
Ward, M.D., and Hammer, D.A. 1993. A theoretical analysis for the effect of focal contact formation on cell-substrate attachment strength. Biophys. J. 64: 936–959.
Lin, H.-B., Zhao, Z.C., Garcia-Echeverria, C., Rich, D.H., and Cooper, S.L. 1992. Synthesis of a novel polyurethane copolymer containing covalently attached RGD peptide. J. Biomater. Sci., Polym. Ed. 3: 217–227.
Clapper, D.L., Kirkham, S.M., and Guire, P.E. 1994. ECM proteins coupled to device surfaces improve in vivo tissue integration. J. Cell. Biochem. Supplement 18C: 283.
Guire, P.E. 1993. Biocompatible device with calently bonded biocompati-ble agent. U.S. Patent 5: 263–992.
Hubbell, J.A., Massia, S.P., Desai, N.P., and Drumheller, P.D. 1991. Endothelial cell-selective materials for tissue engineering in the vascular graft via a new receptor. Bio/Technology 9: 568–572.
Massia, S.P., and Hubbell, J.A. 1992. Vascular endothelial cell adhesion and spreading promoted by the peptide REDV of the IIICS region of plasma fibronectin is mediated by integrin α4β1. J Biol. Chem. 267: 14019–14026.
Chaikof, E.L., Merrill, E.W., Coleman, J.E., Ramberg, K., Connoll, R.J., and Callow, A.D. 1990. Platelet interactions with poly(ethylene oxide) networks. AIChE J. 36: 994–1001.
Drumheller, P.D., and Hubbell, J.A. 1994. Polymer networks with grafted cell adhesive peptides for highly biospecific cell adhesive substrates. Anal. Biochem. 222: 380–388.
Jackson, R.L., Busch, S.J., and Cardin, A.D. 1991. Glycosaminoglycans: molecular properties, protein interactions, and role in physiological processes. Physiol. Rev. 71: 481–539.
LeBaron, R.G., Esko, J.D., Woods, A., Johansson, S., and Hook, M. 1988. Adhesion of glycosaminoglycan-deficient Chinese hamster ovary cell mutants to fibronectin substrata. J. Cell Biol. 106: 945–952.
Massia, S.P., and Hubbell, J.A. 1992. Immobilized amines and basic amino acids as mimetic heparin-binding domains for cell surface proteoglycan-medi-ated adhesion. J. Biol. Chem. 267: 10133–10141.
Geffen, I., and Spiess, M. 1992. Asialoglycoprotein receptor. Int. Rev. Cytol. 1378: 181–219.
Gutsche, A.T., Parsons-Wingerter, P., Chand, D., Saltzman, W.M., and Leong, K.W. 1994. N-acetylglucosamine and adenosine derivatized surfaces for cell culture: 3T3 fibroblast and chicken hepatocyte response. Biotechnol. Bioeng. 43: 801–809.
Kobayashi, K., Kobayashi, A., and Akaike, T. 1994. Culturing hepatocytes on lactose-carrying polystyrene layer via asialoglycoprotein receptor-mediated interactions. Meth. Enzymol. 247: 409–418.
Kobayashi, A., Kobayashi, K., and Akaike, T. 1992. Control of adhesion and detachment of parenchymal liver cells using lactose-carrying polystyrene as substratum. J. Biomat. Sci, Polym. Ed. 3: 499–508.
Lasky, L.A. 1992. Selecting: interpreters of cell-specific carbohydrate infor-mation during inflammation. Science 258: 964–969.
Varki, A. 1994. Selectin ligands. Proc. Nat. Acad. Sci. USA 91: 7390–7397.
Hatanaka, K., Ito, Y., Maruyama, A., Akaike, T., Ishio, K., and Uryo, T. 1993. Synthesis of a new polymer containing a blood-group antigenic oligosaccharide chain. Macromolecules 26: 1483–1486.
Vanderrest, M., and Garrone, R. 1991. Collagen family of proteins. FASEB J. 5: 2814–2823.
Parkhurst, M.R. and Saltzman, W.M. 1992. Quantification of human neu-trophil motility in 3-dimensional collagen gels: effect of collagen concentration. Biophys. J. 61: 306–315.
Krewson, C.E., Chung, S.W., Dai, W.G. and Saltzman, W.M. 1994. Cell aggregation and neurite growth in gels of extracellular matrix molecules. Biotechnol. Bioeng. 43: 555–562.
Naeme, P.J. and Barry, F.P. 1993. The link proteins. Experimentia 49: 393–402.
Urman, B., Gomel, V. and Jetha, N. 1991. Effect of hyaluronic acid on postoperative intraperitoneal adhesion prevention in the rat model. Fertil. Steril. 56: 563–567.
Sung, K.C. and Topp, E.M. 1994. Swelling properties of hyaluronic acid ester membranes. J. Membrane Sci. 92: 157–167.
Kvam, B.J., Atzori, M., Toffanin, R., Paoktti, S. and Biviano, F. 1992. 1H-NMR and 13C-NMR studies of solutions of hyaluronic acid esters and salts in methyl sulfoxide: comparison of hydrogen bond patterns and conformational behavior. Carbohyd. Res. 230: 1–13.
Krejchi, M.T., Atkins, E.D.T., Waddon, A.J., Fournier, M.J., Mason, T.L. and Tirrell, D.A. 1994. Chemical sequence control of beta-sheet assembly in macromolecular crystals of periodic polypeptides. Science 265: 1427–1432.
Yoshikawa, E., Fournier, M.J., Mason, T.L. and Tirrell, D.A. 1994. Genetically engineered fluoropolymers: synthesis of repetitive polypeptides containing p-fluorophenylalanine residues. Macromolecules 27: 5471–5475.
Dessipri, E. and Tirrell, D.A. 1994. Trifluoroalanine N-carboxyanyhdride: a reactive intermediate for the synthesis of low surface energy polypeptides. Macromolecules 27: 5463–5470.
Cappello, J. 1992. Genetic production of synthetic protein polymers. MRS Bulletin 17: 48–53.
Putnam, D. and Cappello, J. 1993. Improving the growth of anchorage-dependent cells upon abrupt passage to serum-free media. Am. Biotechnol. Lab. 11: 14.
Anderson, J.P., Cappello, J. and Martin, D.C. 1994. Morphology and primary crystal structure of a silk-like protein polymer synthesized by genetically engineered escherichia coli bacteria. Biopolymers 34: 1049–1058.
Rosenbloom, J., Abrams, W.R. and Mecham, R. 1993. Extracellular matrix, 4: the elastic fiber. FASEB J. 7: 1208–1218.
McPherson, D.T., Morrow, C., Minehan, D.S., Wu, J.G., Hunter, E. and Urry, D.W. 1992. Production and purification of a recorribinant elastomeric polypeptide G(VPGVG)19VPGV from Escherichia coli. Biotechnol. Progress 8: 347–352.
Urry, D.W. 1993. Molecular machines: how motion and other functions of living organisms can result from reversible chemical changes. Angew. Chem. Int. Ed. Engl. 32: 819–841.
Nicol, A., Gowda, D.C. and Urry, D.W. 1992. Cell adhesion and growth on synthetic elastomeric matrices containing Arg-Gly-Asp-Ser3 . J. Biomed. Mater. Res. 26: 393–413.
Vert, M. and Li, S.M. 1992. Bioresorbability and biocompatibility of aliphatic polyesters. J. Mater. Sci. Mater. Med. 3: 432–446.
Hoffman, K.R. and Casey, D.J. 1985. Effect of carboxyl end groups on hydrolysis of polyglycolic acid. J. Polym. Sci, Polym. Chem. Ed. 23: 1939–1954.
Miller, R.A., Brady, J.M. and Cutright, D.E. 1977. Degradation rates of oral resorbable implants (polylactates and polyglycolates): rate modification with changes in PLA/PGA copolymer ratios. J. Biomed. Mater. Res. 11: 711–719.
Pitt, C.G., Gratzl, M.M., Kimmel, G.L., Surles, J. and Schindler, A. 1981. Aliphatic polyesters, II: the degradation of poly(dl-lactide), poly(ε-caprolac-tone) and their copolymers in vivo. Biomaterials 2: 215–220.
Dunn, R.L., Yewey, G.L., Duysen, E.D., Poison, E.M. and Southard, G.L. 1994. In situ forming of biodegradable polymeric implants for tissue regeneration. Abst. Am. Chem. Soc. 208: 247–POLY
Slepian, M.J., Campell, P.K., Berrigan, K., Roth, L., Massia, S.P., Weselcouch, E., Ron, E., Mathiowitz, E., Jacob, J., Chickering, D. and Philbrook, M. 1994. Biodegradable endoluminal polymer layers provide sustained transmural heparin delivery to the arterial wall in vivo. Circulation 90: 20.
Gombotz, W.R., Guanghui, W. and Hoffman, A.S. 1990. Protein adsorption to poly(ethylene oxide) surfaces. J. Appl. Polym. Sci. 37: 91–107.
Sawhney, A.S., Pathak, C.P. and Hubbell, J.A. 1993. Bioerodible hydrogels based on photopolymerized poly(ethylene glycol)-co-poly(α-hydroxy acid) diacrylate macromers. Macromolecules 26: 581–587.
West, J.L. and Hubbell, J.A. 1995. Photopolymerized hydrogel materials for drug delivery applications. Reactive Polym. In press.
Leach, R.E. and Henry, R.L. 1990. Reduction of postoperative adhesion in the rat uterine hom model with Poloxamer 407. Am. J. Obstet. Gynecol. 162: 1317–1319.
Intveld, P.J.A., Shen, Z.R., Takens, G.A.J., Dijkstra, P.J. and Feijen, J. 1994. Glycine glycolic acid based copolymers. J. Polym. Sci. Polym. Chem. 32: 1063–1069.
Barrera, D.A., Zylstra, E., Lansbury, P.T. and Langer, R. 1995. Copolymerization and degradation of poly(lactic acid-colysine). Macromolecules 28: 425–432.
Barrera, D.A., Zylstra, E., Lansbury, P.T. and Langer, R. 1993. Synthesis and ROD peptide modification of a new biodegradable copolymer polyflactic acid-co-lysine). J. Am. Chem. Soc. 115: 11010–11011.
Kleinfeld, D., Kahler, K.H. and Hockberger, P.E. 1988. Controlled outgrowth of dissociated neurons on patterned substrates. J. Neurosci. 8: 4098–4120.
Stenger, D.A., Gerger, J.H., Dulcey, C.S., Hickman, J.J., Rudolph, A.S., Nielsen, T.B., McCort, S.M. and Calvert, J.M. 1992. Coplanar molecular assemblies of aminoalkylsilane and perfluorinated alkylsilane: characterization and geometric definition of mammalian cell adhesion and growth. J. Am. Chem. Soc. 114: 8435–8442.
Bain, C.D. and Whitesides, G.M. 1989. Modeling organic surfaces with self-assembled monolayers. Angew. Chem. Int. Edn. Engl. 28: 506–512.
Folkers, J.P., Laibinis, P.E. and Whitesides, G.M. 1992. Self-assembled monolayers of alkanethiols on gold: comparisons of monolayers containing mixtures of short-chain and long-chain constituents with CH3 and CH2OH terminal groups. Langmuir 8: 1330–1341.
Prime, K.L. and Whitesides, G.M. 1993. Adsorption of proteins onto surfaces containing end-attached oligoiethylene oxide): a model system using self-assembled monolayers. J. Am. Chem. Soc. 115: 10714–10721.
Kamar, A., Biebuyck, H.A. and Whitesides, G.M. 1994. Patterning self-assembled monolayers: applications in materials science. Langmuir 10: 1498–1511.
Yannas, I.V. 1994. Applications of ECM analogs in surgery. J. Cell. Biochem. 56: 188–191.
Yannas, I.V. 1990. Biologically active analogs of the extracellular matrix: artificial skin and nerves. Angew. Chem. Int. Ed. Engl. 29: 20–35.
Grinnell, F. 1994. Fibroblasts, myofibroblasts, and wound contraction. J. Cell Biol. 124: 401–404.
Pettit, D.K., Hoffman, A.S. and Horbett, T.A. 1994. Correlation between corneal epithelial cell outgrowth and monoclonal antibody binding to the cell-binding domain of adsorbed fibronectin. J. Biomed. Mater. Res. 28: 685–691.
Norde, W. and Favier, J.P. 1992. Structure of adsorbed and desorbed proteins. Coll. Surf. 64: 87–93.
Humphries, M.J., Akiyama, S.K., Komoriya, A., Olden, K. and Yamada, K.M. 1986. Identification of an alternatively spliced site in human plasma fibronectin that mediates cell type specific adhesion. J. Cell Biol. 103: 2637–2647.
Srnghvi, R., Kumar, A., Lopez, G.P., Stephanopoulos, G.N., Wang, D.I.C., Whitesides, G.M. and Ingber, D.E. 1994. Engineering cell shape and function. Science 264: 696–698.
Healy, K.E., Lorn, B. and Hockberger, P.E. 1994. Spatial distribution of mammalian cells dictated by material surface chemistry. Biotechnol. Bioeng. 43: 792–800.
Ranieri, J.P., Bellamkonda, R., Jacob, J., Vargo, T.G., Gardella, J.A. and Aebischer, P. 1993. Selective neuronal cell attachment to a covalently patterned monoamine on fluorinated ethylene-propylene films. J. Biomed. Mater. Res. 27: 917–925.
Ranieri, J.P., Bellamkonda, R., Bekos, E.J., Gardella, J.A., Mathieu, H.J., Ruiz, L. and Aebischer, P. 1994. Spatial control of neuronal cell attachment and differentiation on covalently patterned laminin oligopeptide substrates. Int. J. Develop. Neurosci. 12: 725–735.
Moghaddam, M.J. and Matsuda, T. 1993. Molecular design of 3-dimensional artificial extracellular matrix: photosensitive polymers containing cell adhesive peptide. J. Polym. Sci. Polym. Chem. 31: 1589–1597.
Bell, E., Rosenberg, M., Kemp, P., Gay, R., Green, G.D., Muthukumaran, N. and Note, C. 1991. Recipies for reconstituting skin. J. Biomech. Eng. Trans. ASME 113: 113–119.
Mikos, A.G., Bao, Y., Cima, L.G., Ingber, D.E., Vacant!, J.P. and Langer, R. 1993. Preparation of poly(glycoltc acid) bonded fiber structures for cell attachment and transplantation. J. Biomed. Mater. Res. 27: 183–189.
Freed, L.E., Grande, D.A., Lingbin, Z., Emmanual, J., Marquis, J.C. and Langer, R. 1994. Joint resurfacing using allograft chondrocytes and synthetic biodegradable polymer scaffolds. J. Biomed. Mater. Res. 28: 891–899.
Freed, L.E., Vunjaknovakovic, G., Biron, R.J., Eagles, D.B., Lesnoy, D.C., Barlow, S.K. and Langer, R. 1994. Biodegradable polymer scaffolds for tissue engineering. Bio/Technology 12: 689–693.
Freed, L.E., Vunjaknovakovic, G., Emmanual, J. and Langer, R. 1994. Composition of cell-polymer cartilage implants. Biotechnol. Bioeng. 43: 605–614.
Mooney, D.J., Organ, G., Vacanti, J.P. and Langer, R. 1994. Design and fabrication of biodegradable polymer devices to engineer tubular tissues. Cell Transplant. 3: 203–210.
Wake, M.C., Patrick, C.W. and Mikos, A.G. 1994. Pore morphology effects on the fibrovascular tissue growth in porous polymer substrates. Cell Transplant. 3: 339–343.
Johnson, L.B., Aiken, J., Mooney, D.J., Schloo, B.L., Griffith-Cima, L., Langer, R. and Vacanti, J.P. 1994. The mesentery as a lamininated vascular bed for hepatocyte transplantation. Cell Transplant. 3: 273–281.
Mooney, D., Hansen, L., Vacanti, J., Langer, R., Farmer, S. and Ingber, D. 1992. Switching from differentiation to growth in hepatocytes: control by extracellular matrix. J. Cell. Physiol. 151: 497–505.
Hansen, L.K., Mooney, D.J., Vacanti, J.P. and Ingber, D.E. 1994. Integral binding and cell spreading on extracellular matrix act at different points in the cell cycle to promote hepatocyte growth. Molec. Biol. Cell 5: 967–975.
Dunn, J.C.Y., Thomkins, R.G. and Yarmush, M.L. 1992. Hepatocytes in collagen sandwich: evidence, for transcriptional and translational regulation. J. Cell Biol. 116: 1043–1053.
Parsons- Wingerter, P.A. and Salteman, W.M. 1993. Growth versus function in the 3-dimensional culture of single and aggregated hepatocytes within collagen gels. Biotechol. Progr. 9: 600–607.
Dia, W.G., Belt, J. and Saltzman, W.M. 1994. Cell-binding peptides conjugated to poly(ethylene glycol) promote neural cell aggregation. Bio/Technology 12: 797–801.
Nyberg, S.L., Shirabe, K., Peshwa, M.V., Sielaff, T.D., Crotty, P.L., Mann, H.J., Remmel, R.P., Payne, W.D., Hu, W.S. and Cerra, F.B. 1993. Extracorporeal application of a gel entrapment, bioartificial liver: demonstration of drug metabolism and other biochemical functions. Cell Transplant. 2: 441–452.
Spargo, B.J., Testoff, M.A., Nielsen, T.B., Stenger, D.A., Hickman, J.J. and Rudolph, A. 1994. Spatially controlled adhesion, spreading, and differentiation of endothelial cells on self-assembled molecular monolayers. Proc. Nat. Acad. Sci. USA 91: 11070–11074.
Edelman, E.R., Mathiowitz, E., Langer, R. and Klagsbrun, M. 1991. Controlled and modulated release of basic fibroblast growth factor. Biomaterials 12: 619–626.
Lee, S., Shea, M., Battle, M.A., Kazitza, K., Ron, E., Turek, T., Scarab, R.G. and Hayes, W.C. 1994. Healing of large segmental defects in rat femurs is aided by RHBMP-2 in PLGA. J. Biomed. Mater. Res. 28: 1149–1156.
Guenard, V., Kleitman, N., Morrissey, T.K., Bunge, R.P. and Aebischer, P. 1992. Syngeneic Schwann cells derived from adult nerves seeded in semipermeable guidance channels enhance peripheral nerve regeneration. J. Neurosci. 12: 3310–3320.
Williams, S.K., Rose, D.G. and Jarrell, B.E. 1994. Microvascalar endothelial cell sodding 6f ePTFB vascular grafts: improved patency and stability of the cellular lining. J. Biomed. Mater. Res. 28: 203–212.
Lum, Z.P., Tai, I.T., Krestow, M., Norton, J., Vacek, I. and Sun, A.M. 1991. Prolonged reversal of diabetic state in NOD mice by xenografts of microencapsulated rat islets. Diabetes 40: 1511–1516.
Soon-Shiong, P., Feldman, E., Nelson, R., Heintz, R., Yao, Q., Yao, Z.W., Zheng, T.L., Merideth, N., Skjakbraek, G., Espevik, T., Smidsrod, O. and Sandford, P. 1993. Long-term reversal of diabetes by the injection of immunoprotected islets. Proc. Nat. Acad. Sci. USA 98: 5843–5847.
Joseph, J.M., Goddard, M.B., Mills, J., Padrun, V., Zum, A., Zielinski, B., Favre, J., Gardaz, J.P., Mosimann, F., Sagen, J., Christenson, L. and Aebischer, P. 1994. Transplantation of encapsulated bovine chromaffin cells in the sheep subarachnoid space: a preclinical study for the treatment of cancer pain. Cell Transplant. 3: 355–364.
Uludag, H., Horvath, V., Black, J.P. and Sefton, M.V. 1994. Viability and protein secretion from human hepatoma (HEPG2) cells encapsulated in 400 um polyacrylate microcapsules by submerged nozzle liquid extrusion. Biotechnol. Bioeng. 44: 1199–1204.
Dionne, K.E., Colton, C.K. and Yarmush, M.L. 1993. Effect of hypoxia on insulin secretion by isolated rat and canine islets of Langerhans. Diabetes 42: 12–21.
Sawhney, A.S., Pathak, C.P. and Hubbell, J.A. 1994. Modification of Islet of Langerhans surfaces with immunoprotective poly(ethylene glycol) coatings via interfacial photopolymerization. Biotechnol. Bioeng. 44: 383–386.
Ferns, G.A.A., Raines, E.W., Sprugel, K.H., Motani, A.S., Reidy, M.A. and Ross, R. 1991. Inhibition of neointimal smooth muscle accumulation after angioplasty by an antibody to PDGF. Science 253: 1129–1132.
Hill-West, J.L., Chowdhury, S.M., Stopian, M.J. and Hubbell, J.A. 1994. Inhibition of thrombosis and intimal thickening by in situ photopolymerization of thin hydrogel barriers. Proc. Nat. Acad. Sci. USA 91: 5967–5971.
Hill-West, J.L., Chowdhury, S.M., Sawhney, A.S., Pathak, C.P., Dunn, R.C. and Hubbell, J.A. 1994. Prevention of postoperative adhesions in the rat by in situ photopolymerization of bioresorbable hydrogel barriers. Obstet. Gynecol. 83: 59–64.
Radzilowski, L.H. and Stupp, S.I. 1994. Nanophase separation in monodisperse rodcoil diblock polymers. Macromolecules 27: 7747–7753.
Messersmith, P.B. and Stupp, S.I. 1992. Synthesis of nanocomposites: organoceramics. J. Mater. Res. 7: 2599–2611.
Chen, G.H. and Hoffinan, A.S. 1995. Graft copolymers that exhibit temperature-induced phase transitions over a wide range of pH. Nature 393: 49–52.
Brauker, J., Martinson, L.A., Hill, R.S., Young, S.K., GirrbMiKW, V.E. and Johnson, R.C. 1992. Neovascularizatioo of immunoisolation membranes: the effect of architecture and encapsulation of tissue. Transplant. Proc. 24: 2924.
Schmidt, J.A. and von Recum, A.F. 1992. Macrophage response to microtextured silicone. Biomaterials 13: 1059–1069.
Green, A.M., Jansen, J.A., van der Waerden, J.P.C.M. and von Recum, A.F. 1994. Fibroblast response to microtextured silicone surfaces: texture orientation into or out of the Surface. J. Biomed. Mater. Res. 28: 647–653.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hubbell, J. Biomaterials in Tissue Engineering. Nat Biotechnol 13, 565–576 (1995). https://doi.org/10.1038/nbt0695-565
Issue Date:
DOI: https://doi.org/10.1038/nbt0695-565
This article is cited by
-
Recent advances in strategies of nanocellulose surface and/or interface engineering for potential biomedical applications as well as its ongoing challenges: a review
Cellulose (2023)
-
Co‐electrospinning of lignocellulosic nanoparticles synthesized from walnut shells with poly(caprolactone) and gelatin for tissue engineering applications
Cellulose (2021)
-
Additively Manufactured Magnesium-Based Bio-Implants and their Challenges
Transactions of the Indian National Academy of Engineering (2021)
-
The silk of gorse spider mite Tetranychus lintearius represents a novel natural source of nanoparticles and biomaterials
Scientific Reports (2020)