Abstract
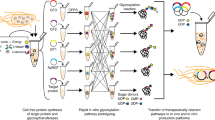
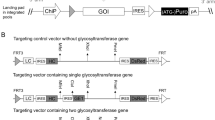
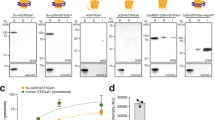
Heterogeneity in the N-glycans on therapeutic proteins causes difficulties for protein purification and process reproducibility and can lead to variable therapeutic efficacy. This heterogeneity arises from the multistep process of mammalian complex-type N-glycan synthesis. Here we report a glycoengineering strategy—which we call GlycoDelete—that shortens the Golgi N-glycosylation pathway in mammalian cells. This shortening results in the expression of proteins with small, sialylated trisaccharide N-glycans and reduced complexity compared to native mammalian cell glycoproteins. GlycoDelete engineering does not interfere with the functioning of N-glycans in protein folding, and the physiology of cells modified by GlycoDelete is similar to that of wild-type cells. A therapeutic human IgG expressed in GlycoDelete cells had properties, such as reduced initial clearance, that might be beneficial when the therapeutic goal is antigen neutralization. This strategy for reducing N-glycan heterogeneity on mammalian proteins could lead to more consistent performance of therapeutic proteins and modulation of biopharmaceutical functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ferrara, C. et al. Modulation of therapeutic antibody effector functions by glycosylation engineering: influence of Golgi enzyme localization domain and co-expression of heterologous β1, 4-N-acetylglucosaminyltransferase III and Golgi α-mannosidase II. Biotechnol. Bioeng. 93, 851–861 (2006).
Elliott, S. et al. Control of rHuEPO biological activity: the role of carbohydrate. Exp. Hematol. 32, 1146–1155 (2004).
Jacobs, P.P., Geysens, S., Vervecken, W., Contreras, R. & Callewaert, N. Engineering complex-type N-glycosylation in Pichia pastoris using GlycoSwitch technology. Nat. Protoc. 4, 58–70 (2008).
Hamilton, S.R. et al. Humanization of yeast to produce complex terminally sialylated glycoproteins. Science 313, 1441–1443 (2006).
Herter, S. et al. Preclinical activity of the type II CD20 antibody GA101 (obinutuzumab) compared with rituximab and ofatumumab in vitro and in xenograft models. Mol. Cancer Ther. 12, 2031–2042 (2013).
Kanda, Y. et al. Comparison of cell lines for stable production of fucose-negative antibodies with enhanced ADCC. Biotechnol. Bioeng. 94, 680–688 (2006).
Reeves, P.J., Callewaert, N., Contreras, R. & Khorana, H.G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl. Acad. Sci. USA 99, 13419–13424 (2002).
Stals, I. et al. Identification of a gene coding for a deglycosylating enzyme in Hypocrea jecorina. FEMS Microbiol. Lett. 303, 9–17 (2010).
Grundmann, U., Nerlich, C., Rein, T. & Zettlmeissl, G. Complete cDNA sequence encoding human β-galactoside α-2,6-sialyltransferase. Nucleic Acids Res. 18, 667 (1990).
Verstraete, K. et al. Structural insights into the extracellular assembly of the hematopoietic Flt3 signaling complex. Blood 118, 60–68 (2011).
Bernales, S., Papa, F.R. & Walter, P. Intracellular signaling by the unfolded protein response. Annu. Rev. Cell Dev. Biol. 22, 487–508 (2006).
Lee, F. et al. Isolation of cDNA for a human granulocyte-macrophage colony-stimulating factor by functional expression in mammalian cells. Proc. Natl. Acad. Sci. USA 82, 4360–4364 (1985).
Forno, G. et al. N- and O-linked carbohydrates and glycosylation site occupancy in recombinant human granulocyte-macrophage colony-stimulating factor secreted by a Chinese hamster ovary cell line. Eur. J. Biochem. 271, 907–919 (2004).
Crispin, M. et al. Inhibition of hybrid- and complex-type glycosylation reveals the presence of the GlcNAc transferase I-independent fucosylation pathway. Glycobiology 16, 748–756 (2006).
Ericsson, U.B., Hallberg, B.M., DeTitta, G.T., Dekker, N. & Nordlund, P. Thermofluor-based high-throughput stability optimization of proteins for structural studies. Anal. Biochem. 357, 289–298 (2006).
Kitamura, T. et al. Establishment and characterization of a unique human cell line that proliferates dependently on GM-CSF, IL-3, or erythropoietin. J. Cell. Physiol. 140, 323–334 (1989).
Mössner, E. et al. Increasing the efficacy of CD20 antibody therapy through the engineering of a new type II anti-CD20 antibody with enhanced direct and immune effector cell–mediated B-cell cytotoxicity. Blood 115, 4393–4402 (2010).
Nallet, S. et al. Glycan variability on a recombinant IgG antibody transiently produced in HEK-293E cells. N. Biotechnol. 29, 471–476 (2012).
Jefferis, R. Glycosylation as a strategy to improve antibody-based therapeutics. Nat. Rev. Drug Discov. 8, 226–234 (2009).
Roopenian, D.C. & Akilesh, S. FcRn: the neonatal Fc receptor comes of age. Nat. Rev. Immunol. 7, 715–725 (2007).
Lux, A., Yu, X., Scanlan, C.N. & Nimmerjahn, F. Impact of immune complex size and glycosylation on IgG binding to human FcγRs. J. Immunol. 190, 4315–4323 (2013).
Hamblin, M.W. & Metcalf, M.A. Primary structure and functional characterization of a human 5–HT1D-type serotonin receptor. Mol. Pharmacol. 40, 143–148 (1991).
Tradtrantip, L., Ratelade, J., Zhang, H. & Verkman, A.S. Enzymatic deglycosylation converts pathogenic neuromyelitis optica anti–aquaporin-4 immunoglobulin G into therapeutic antibody. Ann. Neurol. 73, 77–85 (2013).
Nandakumar, K.S. et al. Dominant suppression of inflammation by glycan-hydrolyzed IgG. Proc. Natl. Acad. Sci. USA 110, 10252–10257 (2013).
Allhorn, M. & Collin, M. Sugar-free antibodies—the bacterial solution to autoimmunity? Ann. NY Acad. Sci. 1173, 664–669 (2009).
Anonymous. Biosimilar, biobetter and next generation therapeutic antibodies. MAbs 3, 107–110 (2011).
MacLean, B. et al. Skyline: an open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 26, 966–968 (2010).
Tada, H., Shiho, O., Kuroshima, K., Koyama, M. & Tsukamoto, K. An improved colorimetric assay for interleukin 2. J. Immunol. Methods 93, 157–165 (1986).
Ramsland, P.A. et al. Structural basis for Fc γRIIa recognition of human IgG and formation of inflammatory signaling complexes. J. Immunol. 187, 3208–3217 (2011).
Magistrelli, G. et al. Robust recombinant FcRn production in mammalian cells enabling oriented immobilization for IgG binding studies. J. Immunol. Methods 375, 20–29 (2012).
Acknowledgements
We thank G. Pontini, Y. Poitevin, L. Bernasconi, D. Schrag and S. Raimondi (NovImmune) for their help with anti-CD20 generation and characterization; S. Savvides (Ghent University) for providing the 293SGnTI(−) clone expressing the Flt3 receptor extracellular domain; E. Van Damme (Ghent University) for providing ConA; and E. Dirksen and K. Nooijen (Merck) for the LC-MS analysis of anti-CD20. L.M. and M.B. are supported by predoctoral fellowships, and N.F. by a postdoctoral fellowship, of the Fund for Scientific Research-Flanders (FWO). F.S. and S.D. are supported by predoctoral fellowships of agentschap voor Innovatie door Wetenschap en Technologie (IWT) Flanders (Strategic Basic Research fellowship nos. 101456 and 111252). This research was supported by VIB, Ghent University Industrial Research Fund (UGent-IOF) Advanced Grant no. 041 (N.C.), FWO research project grant no. G.0.541.08.N.10 (N.C.) and Hercules Foundation grant no. AUGE019 (B.D.).
Author information
Authors and Affiliations
Contributions
L.M.: took the GlycoDelete technology from concept to reality: cell engineering, cell characterization, GM-CSF analytics, and thermofluor assays and co-wrote the manuscript. F.S.: made key contributions in the full exploration of the GlycoDelete technology: GM-CSF analytics, anti-CD20 analytics, and pharmacokinetics experiment, and co-wrote the manuscript. G.E.: made key contributions to the exploration of GlycoDelete technology for modulating antibody properties: experimental design, and supervision and interpretation of anti-CD20 bioanalytics experiments, and manuscript correction. N.F.: bioactivity, immunogenicity ELISA and pharmacokinetics experiments, contributed to the manuscript. M.B.: gene expression experiments and data processing. A.D.S.: ADCC, ELISA FcγR binding and CD20-binding assays. S.D.: LC-MS/MS experiments and analysis. F.R.: anti-CD20 expression construct. (This plasmid is not explicitly mentioned in the text and is an unpublished resource that was made available to us through the work of F.R.) E.P.: assisted with immunogenicity ELISA experiments. E.H.: pharmacokinetics experiment. P.M. and G.M.: SPR and BLI experiments. L. Cons and L. Chatel: pharmacokinetics experiment. B.D.: supervision of LC-MS/MS experiments. N.C.: conceived the GlycoDelete technology, initiated the project, assisted in experimental design and interpretation, and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
L.M. and N.C. are inventors on patent applications covering the GlycoDelete technology (WO/2010/015722).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15, Supplementary Tables 1–8 and Supplementary Notes 1–6 (PDF 4124 kb)
Rights and permissions
About this article
Cite this article
Meuris, L., Santens, F., Elson, G. et al. GlycoDelete engineering of mammalian cells simplifies N-glycosylation of recombinant proteins. Nat Biotechnol 32, 485–489 (2014). https://doi.org/10.1038/nbt.2885
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2885
This article is cited by
-
Immobilized enzyme cascade for targeted glycosylation
Nature Chemical Biology (2024)
-
Antibody-dependent cellular cytotoxicity-null effector developed using mammalian and plant GlycoDelete platform
Scientific Reports (2022)
-
Homogeneous production and characterization of recombinant N-GlcNAc-protein in Pichia pastoris
Microbial Cell Factories (2020)
-
Engineering orthogonal human O-linked glycoprotein biosynthesis in bacteria
Nature Chemical Biology (2020)
-
Structural basis for potent neutralization of SARS-CoV-2 and role of antibody affinity maturation
Nature Communications (2020)