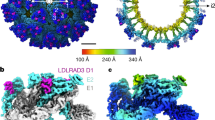
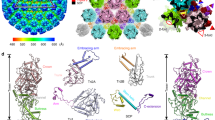
Abstract
Little is known about the three-dimensional organization of rubella virus, which causes a relatively mild measles-like disease in children but leads to serious congenital health problems when contracted in utero1. Although rubella virus belongs to the same family as the mosquito-borne alphaviruses, in many respects it is more similar to other aerosol-transmitted human viruses such as the agents of measles and mumps. Although the use of the triple MMR (measles, mumps and rubella) live vaccine has limited its incidence in western countries, congenital rubella syndrome remains an important health problem in the developing world. Here we report the 1.8 Å resolution crystal structure of envelope glycoprotein E1, the main antigen and sole target of neutralizing antibodies against rubella virus. E1 is the main player during entry into target cells owing to its receptor-binding and membrane-fusion functions. The structure reveals the epitope and the neutralization mechanism of an important category of protecting antibodies against rubella infection. It also shows that rubella virus E1 is a class II fusion protein, which had hitherto only been structurally characterized for the arthropod-borne alphaviruses and flaviviruses. In addition, rubella virus E1 has an extensive membrane-fusion surface that includes a metal site, reminiscent of the T-cell immunoglobulin and mucin family of cellular proteins that bind phosphatidylserine lipids at the plasma membrane of cells undergoing apoptosis. Such features have not been seen in any fusion protein crystallized so far. Structural comparisons show that the class II fusion proteins from alphaviruses and flaviviruses, despite belonging to different virus families, are closer to each other than they are to rubella virus E1. This suggests that the constraints on arboviruses imposed by alternating cycles between vertebrates and arthropods resulted in more conservative evolution. By contrast, in the absence of this constraint, the strictly human rubella virus seems to have drifted considerably into a unique niche as sole member of the Rubivirus genus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
The atomic coordinates and structure factors have been deposited in the Protein Data Bank under accession numbers 4ADI, 4ADG, 4ADJ and 4B3V. They correspond to the atomic models of Na+-bound E1e, Ca2+-bound E1e (from the galactose-3-sulphate headgroup soak (see Supplementary Table 2)), and to E1e crystallized in the presence of calcium acetate at 1 and 20mM, respectively.
References
Hobman, T. C. & Chantler, J. K. in Fields Virology Vol. 1 (ed. D. M. Knipe) 1069–1100 (Lippincott Williams & Wilkins, 2007)
Katow, S. & Sugiura, A. Antibody response to individual rubella virus proteins in congenital and other rubella virus infections. J. Clin. Microbiol. 21, 449–451 (1985)
Cong, H., Jiang, Y. & Tien, P. Identification of the myelin oligodendrocyte glycoprotein as a cellular receptor for rubella virus. J. Virol. 85, 11038–11047 (2011)
Katow, S. & Sugiura, A. Low pH-induced conformational change of rubella virus envelope proteins. J. Gen. Virol. 69, 2797–2807 (1988)
Kee, S. H. et al. Effects of endocytosis inhibitory drugs on rubella virus entry into VeroE6 cells. Microbiol. Immunol. 48, 823–829 (2004)
Petruzziello, R. et al. Pathway of rubella virus infectious entry into Vero cells. J. Gen. Virol. 77, 303–308 (1996)
Gibbons, D. L. et al. Visualization of the target-membrane-inserted fusion protein of Semliki Forest virus by combined electron microscopy and crystallography. Cell 114, 573–583 (2003)
Stiasny, K., Allison, S. L., Schalich, J. & Heinz, F. X. Membrane interactions of the tick-borne encephalitis virus fusion protein E at low pH. J. Virol. 76, 3784–3790 (2002)
Stiasny, K., Bressanelli, S., Lepault, J., Rey, F. A. & Heinz, F. X. Characterization of a membrane-associated trimeric low-pH-induced form of the class II viral fusion protein E from tick-borne encephalitis virus and its crystallization. J. Virol. 78, 3178–3183 (2004)
Bennett, M. J., Schlunegger, M. P. & Eisenberg, D. 3D domain swapping: a mechanism for oligomer assembly. Protein Sci. 4, 2455–2468 (1995)
Holm, L. & Rosenstrom, P. Dali server: conservation mapping in 3D. Nucleic Acids Res. 38, W545–W549 (2010)
Qiu, Z., Yao, J., Cao, H. & Gillam, S. Mutations in the E1 hydrophobic domain of rubella virus impair virus infectivity but not virus assembly. J. Virol. 74, 6637–6642 (2000)
Santiago, C. et al. Structures of T cell immunoglobulin mucin receptors 1 and 2 reveal mechanisms for regulation of immune responses by the TIM receptor family. Immunity 26, 299–310 (2007)
Goldstein, D. A. in Clinical Methods: The History, Physical, and Laboratory Examinations Ch 143 (eds H. K. Walker, W. D. Hall, & J. W. Hurst). 677–679 (1990)
Mastromarino, P., Cioe, L., Rieti, S. & Orsi, N. Role of membrane phospholipids and glycolipids in the Vero cell surface receptor for rubella virus. Med. Microbiol. Immunol. 179, 105–114 (1990)
Kobayashi, T. et al. Separation and characterization of late endosomal membrane domains. J. Biol. Chem. 277, 32157–32164 (2002)
Wolinsky, J. S. et al. An antibody- and synthetic peptide-defined rubella virus E1 glycoprotein neutralization domain. J. Virol. 67, 961–968 (1993)
Mitchell, L. A. et al. Characterization of rubella virus-specific antibody responses by using a new synthetic peptide-based enzyme-linked immunosorbent assay. J. Clin. Microbiol. 30, 1841–1847 (1992)
Battisti, A. J. et al. Cryo-electron tomography of rubella virus. J. Virol. 86, 11078–11085 (2012)
Krey, T. et al. The disulfide bonds in glycoprotein E2 of hepatitis C virus reveal the tertiary organization of the molecule. PLoS Pathog. 6, e1000762 (2010)
Roussel, A., Lescar, J., Vaney, M. C., Wengler, G. & Rey, F. A. Structure and interactions at the viral surface of the envelope protein E1 of Semliki Forest virus. Structure 14, 75–86 (2006)
Voss, J. E. et al. Glycoprotein organization of Chikungunya virus particles revealed by X-ray crystallography. Nature 468, 709–712 (2010)
Bressanelli, S. et al. Structure of a flavivirus envelope glycoprotein in its low-pH-induced membrane fusion conformation. EMBO J. 23, 728–738 (2004)
Rey, F. A., Heinz, F. X., Mandl, C., Kunz, C. & Harrison, S. C. The envelope glycoprotein from tick-borne encephalitis virus at 2 Å resolution. Nature 375, 291–298 (1995)
Modis, Y., Ogata, S., Clements, D. & Harrison, S. C. Structure of the dengue virus envelope protein after membrane fusion. Nature 427, 313–319 (2004)
Nayak, V. et al. Crystal structure of dengue virus type 1 envelope protein in the postfusion conformation and its implications for membrane fusion. J. Virol. 83, 4338–4344 (2009)
Roche, S., Bressanelli, S., Rey, F. A. & Gaudin, Y. Crystal structure of the low-pH form of the vesicular stomatitis virus glycoprotein G. Science 313, 187–191 (2006)
Heldwein, E. E. et al. Crystal structure of glycoprotein B from herpes simplex virus 1. Science 313, 217–220 (2006)
Backovic, M., Longnecker, R. & Jardetzky, T. S. Structure of a trimeric variant of the Epstein–Barr virus glycoprotein B. Proc. Natl Acad. Sci. USA 106, 2880–2885 (2009)
Kadlec, J., Loureiro, S., Abrescia, N. G., Stuart, D. I. & Jones, I. M. The postfusion structure of baculovirus gp64 supports a unified view of viral fusion machines. Nature Struct. Mol. Biol. 15, 1024–1030 (2008)
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010)
Schneider, T. R. & Sheldrick, G. M. Substructure solution with SHELXD. Acta Crystallogr. D 58, 1772–1779 (2002)
Vonrhein, C., Blanc, E., Roversi, P. & Bricogne, G. Automated structure solution with autoSHARP. Methods Mol. Biol. 364, 215–230 (2007)
Bricogne, G., Vonrhein, C., Flensburg, C., Schiltz, M. & Paciorek, W. Generation, representation and flow of phase information in structure determination: recent developments in and around SHARP 2.0. Acta Crystallogr. D 59, 2023–2030 (2003)
Abrahams, J. P. & Leslie, A. G. Methods used in the structure determination of bovine mitochondrial F1 ATPase. Acta Crystallogr. D 52, 30–42 (1996)
Cowtan, K. dm: an automated procedure for phase improvement by density modification. Joint CCP4 and ESF-EACBM Newsletter on Protein Crystallography 31, 34–38 (1994)
Langer, G., Cohen, S. X., Lamzin, V. S. & Perrakis, A. Automated macromolecular model building for X-ray crystallography using ARP/wARP version 7. Nature Protocols 3, 1171–1179 (2008)
Bricogne, G. et al. BUSTER, version 2.8.0 (Global Phasing, 2009)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Castile, J. D. & Taylor, K. M. Factors affecting the size distribution of liposomes produced by freeze–thaw extrusion. Int. J. Pharm. 188, 87–95 (1999)
Acknowledgements
We thank A. Haouz of Institut Pasteur and the PXI beam line staff at the Swiss Light Source and PROXIMA 1 beam line at SOLEIL synchrotron facilities for assistance; C. Vonrhein at Global Phasing for expertise in phase determination; G. Bricogne for input, G. Dupras and J.M. Mallet at École Normale Supérieure in Paris for the synthesis of galactose-3-sulphate used in co-crystallization trials; and J. Casasnovas, L. Chernomordik, Y. Gaudin, S. Harrison, F. Heinz, M. Kielian and J. Lepault for discussion. R.M.D. was supported by a ‘Pasteur–Howard’ fellowship. This work was mainly funded by grant ANR-05-MIIM-012-02-Dentry to F.A.R., who also acknowledges support from Merck-Serono from the French Government’s Investissements d’Avenir program: Laboratoire d’Excellence ‘Integrative Biology of Emerging Infectious Diseases’ (grant no. ANR-10-LABX-62-IBEID).
Author information
Authors and Affiliations
Contributions
R.M.D. produced and crystallized E1e. R.M.D. and M.-C.V. solved the E1e structure (with initial help from C. Vonrhein) and participated in manuscript preparation. M.A.T. performed liposome flotation assays and crystallized E1e with calcium acetate. R.A.-K. performed liposome flotation assays and electron microscopy studies. G.B.-S. characterized E1e produced in mammalian cells. T.K. constructed the modified pMT plasmid used for E1e expression. F.A.R. conceived the experiments and wrote the manuscript with R.M.D. and M.C.V.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-8, Supplementary Methods, Supplementary Tables 1-2 and Supplementary Results and Discussion. These Supplementary items provide further detail regarding the biochemical and structural features of E1e, including details about the metal-binding site and comparison to other class II viral fusion proteins. (PDF 6782 kb)
The fusion surface
Each subunit is colored according to domains (yellow, red and blue, as above, with the stem region pink/magenta). Domain II is represented in gold and pale yellow, as in figures S4 and S8. The side chains of residues at the fusion surface are drawn as sticks colored according to atom type (carbon white, nitrogen blue, oxygen red). The video initially shows a ribbon diagram of the trimer, and then switches to a full-surface representation (both solid and semitransparent). The conformation displayed is that of calcium acetate bound, as observed in crystal form 2, with the Ca2+ ion represented as a green sphere and the acetate anion as sticks colored according to atom type, with carbon atoms cyan. The sticks in the acetate ion switch to spheres later in the video, so that the acetate can still be seen in the context of the molecular surface of the trimer. The video shows that FL1 and FL2 make a ridge at the edges of a relatively flat surface, rich in aromatic residues, that is exposed to the membrane (termed the “fusion surface”). The three metal ions are on the internal side of the ridges, and the 3 stems end at the opposite side, at the base of the ridges. (MPG 16283 kb)
Conformational change in FL2 upon Ca2+ binding
Transition from Na+-bound to Ca2+-bound conformation and back, generated by morphing one structure into the other to illustrate the change in main chain and side chains in FL2. (MPG 1465 kb)
Rights and permissions
About this article
Cite this article
DuBois, R., Vaney, MC., Tortorici, M. et al. Functional and evolutionary insight from the crystal structure of rubella virus protein E1. Nature 493, 552–556 (2013). https://doi.org/10.1038/nature11741
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11741
This article is cited by
-
Evolution and activation mechanism of the flavivirus class II membrane-fusion machinery
Nature Communications (2022)
-
Relatives of rubella virus in diverse mammals
Nature (2020)
-
Structural basis of potent Zika–dengue virus antibody cross-neutralization
Nature (2016)
-
The ins and outs of hepatitis C virus entry and assembly
Nature Reviews Microbiology (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.