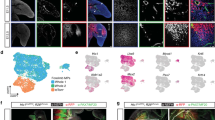
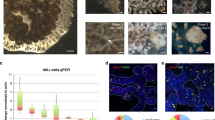
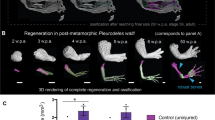
Abstract
The regrowth of amputated limbs and the distal tips of digits represent models of tissue regeneration in amphibians, fish and mice. For decades it had been assumed that limb regeneration derived from the blastema, an undifferentiated pluripotent cell population thought to be derived from mature cells via dedifferentiation. Here we show that a wide range of tissue stem/progenitor cells contribute towards the restoration of the mouse distal digit. Genetic fate mapping and clonal analysis of individual cells revealed that these stem cells are lineage restricted, mimicking digit growth during development. Transplantation of cyan-fluorescent-protein-expressing haematopoietic stem cells, and parabiosis between genetically marked mice, confirmed that the stem/progenitor cells are tissue resident, including the cells involved in angiogenesis. These results, combined with those from appendage regeneration in other vertebrate subphyla, collectively demonstrate that tissue stem cells rather than pluripotent blastema cells are an evolutionarily conserved cellular mode for limb regeneration after amputation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rinkevich, B., Shlemberg, Z. & Fishelson, L. Whole-body protochordate regeneration from totipotent blood cells. Proc. Natl Acad. Sci. USA 92, 7695–7699 (1995)
Rinkevich, Y., Paz, G., Rinkevich, B. & Reshef, R. Systemic bud induction and retinoic acid signaling underlie whole body regeneration in the urochordate Botrylloides leachi . PLoS Biol. 5, e71 (2007)
Voskoboynik, A. et al. Striving for normality: whole body regeneration through a series of abnormal generations. FASEB J. 21, 1335–1344 (2007)
Sánchez Alvarado, A. Regeneration in the metazoans: why does it happen? Bioessays 22, 578–590 (2000)
Han, M., Yang, X., Lee, J., Allan, C. H. & Muneoka, K. Development and regeneration of the neonatal digit tip in mice. Dev. Biol. 315, 125–135 (2008)
Schotte, O. E. & Smith, C. B. Wound healing processes in amputated mouse digits. Biol. Bull. 117, 546–561 (1959)
Borgens, R. B. Mice regrow the tips of their foretoes. Science 217, 747–750 (1982)
Reginelli, A. D., Wang, Y. Q., Sassoon, D. & Muneoka, K. Digit tip regeneration correlates with regions of Msx1 (Hox 7) expression in fetal and newborn mice. Development 121, 1065–1076 (1995)
Neufeld, D. A. & Zhao, W. Phalangeal regrowth in rodents: postamputational bone regrowth depends upon the level of amputation. Prog. Clin. Biol. Res. 383A, 243–252 (1993)
Illingworth, C. M. Trapped fingers and amputated fingertips in children. J. Pediatr. Surg. 9, 853–858 (1974)
Rosenthal, L. J., Reiner, M. A. & Bleicher, M. A. Nonoperative management of distal fingertip amputations in children. Pediatrics 64, 1–3 (1979)
Vidal, P. & Dickson, M. G. Regeneration of the distal phalanx. A case report. J. Hand Surg. 18, 230–233 (1993)
Morgan, T. H. Regeneration (Macmillan, 1901)
Bratincsák, A. et al. CD45-positive blood cells give rise to uterine epithelial cells in mice. Stem Cells 25, 2820–2826 (2007)
Faustman, D. L. et al. Comment on papers by Chong et al. Nishio et al., and Suri et al. on diabetes reversal in NOD mice. Science 314 1243a 10.1126/science.1129811 (2006)
Djouad, F., Bouffi, C., Ghannam, S., Noël, D. & Jorgensen, C. Mesenchymal stem cells: innovative therapeutic tools for rheumatic diseases. Nature Rev. Rheumatol 5, 392–399 (2009)
Zaidi, N. & Nixon, A. J. Stem cell therapy in bone repair and regeneration. Ann. NY Acad. Sci. 1117, 62–72 (2007)
Brockes, J. P. & Kumar, A. Appendage regeneration in adult vertebrates and implications for regenerative medicine. Science 310, 1919–1923 (2005)
Tamura, K., Ohgo, S. & Yokoyama, H. Limb blastema cell: a stem cell for morphological regeneration. Dev. Growth Differ. 52, 89–99 (2010)
Christen, B., Robles, V., Raya, M., Paramonov, I. & Belmonte, J. C. Regeneration and reprogramming compared. BMC Biol. 8, 5 (2010)
Tsonis, P. A. Stem cells and blastema cells. Curr. Stem Cell Res. Ther. 3, 53–54 (2008)
Weissman, I. L. Stem cells: units of development, units of regeneration, and units in evolution. Cell 100, 157–168 (2000)
Wagers, A. J., Sherwood, R. I., Christensen, J. L. & Weissman, I. L. Little evidence for developmental plasticity of adult hematopoietic stem cells. Science 297, 2256–2259 (2002)
Wagers, A. J. & Weissman, I. L. Plasticity of adult stem cells. Cell 116, 639–648 (2004)
Purhonen, S. J. et al. Bone marrow-derived circulating endothelial precursors do not contribute to vascular endothelium and are not needed for tumor growth. Proc. Natl Acad. Sci. USA 105, 6620–6625 (2008)
Hamrick, M. W. Development and evolution of the mammalian limb: adaptive diversification of nails, hooves, and claws. Evol. Dev. 3, 355–363 (2001)
Said, S., Parke, W. & Neufeld, D. A. Vascular supplies differ in regenerating and nonregenerating amputated rodent digits. Anat. Rec. A 278, 443–449 (2004)
Vasioukhin, V., Degenstein, L., Wise, B. & Fuchs, E. The magical touch: genome targeting in epidermal stem cells induced by tamoxifen application to mouse skin. Proc. Natl Acad. Sci. USA 96, 8551–8556 (1999)
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007)
Indra, A. K. et al. Temporally-controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ERT and Cre-ERT2 recombinases. Nucleic Acids Res. 27, 4324–4327 (1999)
Kimmel, R. A. et al. Two lineage boundaries coordinate vertebrate apical ectodermal ridge formation. Genes Dev. 14, 1377–1389 (2000)
Logan, M. et al. Expression of Cre recombinase in the developing mouse limb bud driven by a Prxl enhancer. Genesis 33, 77–80 (2002)
Akiyama, H. et al. Osteo-chondroprogenitor cells are derived from Sox9 expressing precursors. Proc. Natl Acad. Sci. USA 102, 14665–14670 (2005)
Nowak, J. A., Polak, L., Pasolli, H. A. & Fuchs, E. Hair follicle stem cells are specified and function in early skin morphogenesis. Cell Stem Cell 3, 33–43 (2008)
Pryce, B. A., Brent, A. E., Murchison, N. D., Tabin, C. J. & Schweitzer, R. Generation of transgenic tendon reporters, ScxGFP and ScxAP, using regulatory elements of the scleraxis gene. Dev. Dyn. 236, 1677–1682 (2007)
Forde, A., Constien, R., Gröne, H. J., Hämmerling, G. & Arnold, B. Temporal Cre-mediated recombination exclusively in endothelial cells using Tie2 regulatory elements. Genesis 33, 191–197 (2002)
Monvoisin, A. et al. VE-cadherin-CreERT2 transgenic mouse: a model for inducible recombination in the endothelium. Dev. Dyn. 235, 3413–3422 (2006)
Fernando, W. A. et al. Wound healing and blastema formation in regenerating digit tips of adult mice. Dev. Biol. 350, 301–310 (2011)
Forsberg, E. C., Bhattacharya, D. & Weissman, I. L. Hematopoietic stem cells: expression profiling and beyond. Stem Cell Rev. 2, 23–30 (2006)
Wright, D. E., Wagers, A. J., Gulati, A. P., Johnson, F. L. & Weissman, I. L. Physiological migration of hematopoietic stem and progenitor cells. Science 294, 1933–1936 (2001)
Liversage, R. A. in A History of Regeneration Research: Milestones in the Evolution of a Science (eds Dinsmore, C. E. ) (Cambridge Univ. Press, 1991)
Ito, M. et al. Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature 447, 316–320 (2007)
Chan, C. K. et al. Endochondral ossification is required for haematopoietic stem-cell niche formation. Nature 457, 490–494 (2009)
Gargioli, C. & Slack, J. M. Cell lineage tracing during Xenopus tail regeneration. Development 131, 2669–2679 (2004)
Kragl, M. et al. Cells keep a memory of their tissue origin during axolotl limb regeneration. Nature 460, 60–65 (2009)
Tu, S. & Johnson, S. L. Fate restriction in the growing and regenerating zebrafish fin. Dev. Cell 20, 725–732 (2011)
Acknowledgements
We thank A. Mosley for assistance with animal care and parabiosis experiments; A. Sweet-Cordero for providing Sox9Cre and Prx1Cre mice; R. Schweitzer for providing ScxCre mice; and L. Luo for providing mTmG reporter mice. We thank D. Montoro for assistance with histochemical stains and G. Paz for assistance with figure preparations. This work was supported in part by a grant from the California Institute of Regenerative Medicine (RC1 00354) and from the Smith Family Trust (to I.L.W.), the Oak Foundation and the Hagey Laboratory for Pediatric Regenerative Medicine (to M.T.L.). Y.R. is supported by a Human Frontier Science Program (HFSP) Long Term Fellowship, and the Machiah Foundation Fellowship.
Author information
Authors and Affiliations
Contributions
Y.R. and I.L.W. designed the experiments. Y.R. performed the regeneration experiments, imaged and analysed the data from all regeneration experiments. H.U. provided the Rainbow reporter mice for part of the experiments. M.T.L. provided the histochemical stains for part of the experiments. Y.R. and P.L. performed the HSC transplantations and analysed the data. Y.R. and I.L.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-7 with legends and Supplementary Table 1. (PDF 14165 kb)
Rights and permissions
About this article
Cite this article
Rinkevich, Y., Lindau, P., Ueno, H. et al. Germ-layer and lineage-restricted stem/progenitors regenerate the mouse digit tip. Nature 476, 409–413 (2011). https://doi.org/10.1038/nature10346
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10346
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.