Abstract
The presence of calcification is the most significant ultrasonographic finding in evaluating thyroid nodules. Calcifications are more frequently detected in papillary thyroid carcinoma than in other thyroid lesions. However, the clinical significance of calcification, including clinical correlations and impact on survival, and the molecular mechanism responsible for calcification in papillary thyroid carcinoma remain uncertain. We performed a retrospective study of patients with primary common-type papillary thyroid carcinoma to determine the clinical correlations of calcification and its impact on survival. Histologically, calcification was classified as either psammoma bodies, stromal calcification, or bone formation. They were identified in 25, 47, and 13% of all 229 cases of papillary thyroid carcinoma, respectively. The presence of psammoma bodies was significantly correlated with gross lymph node metastasis and stage grouping. Both stromal calcification and bone formation were significantly correlated with patient age. In addition, stromal calcification was associated with pT classification and gross lymph node metastasis. Papillary thyroid carcinoma with, compared to that without, psammoma bodies was associated with poorer disease-free survival. We examined the quantitative expression of BMP-1, a metalloproteinase that is reported to be involved in bone and extracellular matrix formations, and found that its expression was significantly higher in tumors with psammoma bodies or with stromal calcification (P=0.0464 and 0.0272, respectively). These results suggest that the presence of psammoma bodies is a useful predictor of outcome for patients suffering from papillary thyroid carcinoma.
Similar content being viewed by others
Main
The ability to predict patient outcome using clinical and pathological parameters is of great interest to both clinicians and patients. Earlier investigations have shown that patient age, sex, tumor size, histological grouping, extrathyroid invasion, and lymph node status were significant markers in predicting prognosis in patients suffering from papillary thyroid carcinoma.1, 2, 3 However, calcification, which is a frequent histological characteristic of papillary thyroid carcinoma, is neglected by most of the pathologists. Thyroid ultrasonography is widely used to detect calcification in the preoperative evaluation of thyroid nodules.4, 5 Although calcification can be present in both benign and malignant thyroid lesions, a marked association between cancer status and calcification has been consistently noted in several reports.4, 5, 6, 7 Papillary thyroid carcinoma represents the largest proportion of thyroid malignancies; however, the clinical significance of calcification in papillary thyroid carcinomas remains unclear. On the basis of histological features, calcification was classified as psammoma bodies, stromal calcification, or bone formation. Therefore, we investigated a group of patients with papillary thyroid carcinoma to determine the clinical significance of each type of calcification and its impact on survival.
Little is known about the molecular interactions that result from psammoma bodies, stromal calcification, or bone formation in thyroid, except that members of the bone morphogenetic protein (BMP) family are capable of inducing ectopic bone formation.8 In fact, one thyroid carcinoma cell line, 8505C, was reported to produce BMP-1 and BMP-2.9 BMP-1, also known as procollagen C-peptidase, is a member of the BMP-1 family, but differs from other BMPs in the sense that it is a metalloproteinase; other BMP family members, BMPs 2–7, are TGF-β-like proteins.8 BMP-1 may convert precursor proteins, including laminin-5, procollagen, growth and differentiation factors 8 and 11, and TGF-β1, into their active forms, which play manifold roles in cell adhesion and in regulating mineralization in the extracellular matrix of hard tissue.8, 10, 11, 12, 13, 14, 15, 16, 17 BMP-1 may also activate two other BMP family members, BMP-2 and BMP-4, by cleaving their antagonist, chordin.11 Therefore, BMP-1 is believed to be an intermediate factor between the TGF-β and BMP signaling pathways. Knowledge of the diverse functions of BMP-1 provided the impetus to examine its quantitative expression in papillary thyroid carcinoma and its association with the clinicopathological parameters of papillary thyroid carcinoma, particularly with respect to the presence of psammoma bodies, stromal calcification, and bone formation.
Materials and methods
Patients
We reviewed the records of patients with a diagnosis of primary papillary thyroid carcinoma who underwent surgical treatment in 1990–1991 at the Kuma Hospital, Kobe, Japan. Patients with certain variants of papillary thyroid carcinoma and those with poorly differentiated carcinoma of the thyroid, as defined by the WHO criteria,1 were excluded. In total, 181 patients with common-type (also called classic type or usual type) papillary thyroid carcinoma, and with complete clinical information and follow-up data were enrolled. All patients had a primary tumor measuring at least 10 mm in diameter, with no evidence of distant metastasis. Patients with microcarcinoma were excluded because they have a much better prognosis than do patients with other types of papillary thyroid carcinoma, as noted in our earlier study.18 The average follow-up period was 131±47 (mean±s.d.) months, and their age at surgery was 46±14 (mean±s.d.) years. No postoperative chemotherapy, radiation, or I131 treatment was administrated to these patients (In Japan, I131 treatment is not an option for patients with papillary thyroid carcinoma, unless distant metastasis is identified.). During the follow-up period, 28 (15%) patients developed cancer recurrence, and 8 (4%) patients died: 4 because of cancer and 4 because of other causes.
Forty-eight patients who underwent primary surgery for primary papillary thyroid carcinoma, and from whom fresh carcinoma tissue and non-neoplastic thyroid tissue were available, were also enrolled. Total RNA from both the carcinoma tissue and the non-neoplastic tissue was extracted for molecular analysis.
This study was approved by the ethics committees of both the Wakayama Medical University and the Kuma Hospital, Japan.
Definition of Psammoma Bodies, Stromal Calcification, and Bone Formation
All sections stained with hematoxylin and eosin from a total of 229 cases of papillary thyroid carcinoma, including 181 with the follow-up data and 48 from whom fresh tissues were available, were screened by two pathologists, YB and KK, for the presence of psammoma bodies, stromal calcification, and bone formation within the tumor mass. The presence of psammoma bodies is a diagnostic characteristic of papillary thyroid carcinoma. It is defined as spherical calcified foci with concentric laminations,1, 19, 20 and is usually located within stromal stalks of tumor papillae, and is distinct from intrafollicular inspissated colloid.19 In addition, in our experience, it is occasionally observed inside stromal calcifications. Bone formation was regarded as positive only when both bone matrix and osteocytes were identifiable. All calcific masses that did not meet the criteria of psammoma bodies or bone formation were categorized as stromal calcifications, including psammomatoid calcifications, which are characterized by a spherical shape lacking laminations or by an irregular shape with laminations.20
RNA Extraction and Synthesis of First-strand cDNA
Fresh carcinoma tissue and paired non-neoplastic tissue were snap-frozen immediately and stored at −80°C until RNA extraction. Total RNA was isolated using the ULTRASPEC™ RNA isolation system (Biotecx, TX), according to the manufacturer's instructions. First-strand cDNA was synthesized using 3 μg of total RNA as a template and oligo(dT)12−18 as a primer in a 20-μl reaction unit, according to the instructions provided with the SuperScript First-Strand Synthesis System for RT-PCR (Invitrogen, CA).
Quantitative Real-Time PCR
First-strand cDNA from 48 paired papillary thyroid carcinomas and non-neoplastic thyroid tissues were used as templates to amplify BMP-1 using the ABI PRISM 7000 Sequence Detection System (ABI, Japan). β-Actin served as an endogenous control in this study, because it was reported to harbor a prior stable expression in thyroid tissues compared with other endogenous controls, such as ribosomal protein, P0, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH).21 PCR primers and TaqMan probes for β-actin (ABI; Catalog no. 4310881E) and BMP-1 (ABI; Assay ID Hs00241807_ml) were purchased from Applied Biosystems.
The PCR reactions were performed in 96-well optical reaction plates according to the instructions of TaqMan Gene Expression Assays. Triplicates were run for each cDNA sample on the same plate. Quantification of the target transcript was defined by the relative cycle threshold values (ΔCt), using the average triplicate Ct value for each target minus the average triplicate Ct value for β-actin. Differences between the matched tumor and the non-neoplastic thyroid tissue were calculated using the formula 2exp (ΔCt tumor–ΔCt non), and expressed as a fold change in expression, as reported earlier.22, 23
Statistical Analysis
Data analysis was performed using StatView-J version 5.0. statistical software. Time-independent, categorical, and continuous data were evaluated using the χ2-, Fisher's exact, or the Mann–Whitney U-tests as appropriate. Disease-free survival and univariate analysis were calculated using the Kaplan–Meier method and the log-rank test. Multivariate analysis was performed using the Cox proportional hazards regression model. Statistical significance was defined as P<0.05.
Results
Clinical Significance of Psammoma Bodies, Stromal Calcification, and Bone Formation
Psammoma bodies were present in 25% (58 out of 229) of papillary thyroid carcinoma cases. The presence of psammoma bodies was positively correlated with gross lymph node metastasis (P=0.0347) and stage grouping (TNM classification of UICC, P=0.0255). The presence of psammoma bodies tended to be associated with extrathyroid invasion (P=0.0618), but there was no correlation between the presence of psammoma bodies and age, sex, tumor size, pT (TNM classification of UICC), and histological lymph node metastasis (Table 1).
Approximately 47% (107 out of 229) of papillary thyroid carcinoma cases showed evidence of stromal calcification. An association was noted between stromal calcification and advanced age (≥60 years, P=0.0435), high pT stage (P=0.0078), and gross lymph node metastasis (P=0.0291), but not with the other clinicopathological parameters (Table 2).
The presence of bone formation was lower than that of psammoma bodies and stromal calcification. Bone formation was observed in 13% of our 229 cases. The presence of bone formation was significantly more frequent in older patients (≥60 years, P=0.0127; Table 3).
To determine the relationship between psammoma bodies, stromal calcification, and bone formation pairs, the χ2-test was performed. There was a close correlation between psammoma bodies and stromal calcification (P=0.0161). Both were present in 15% (35 out of 229) and absent in 43% (99 out of 229) of the papillary thyroid carcinoma samples in our series. A strong correlation was noted between the presence of bone formation and stromal calcification (P<0.0001), and the coexistence of stromal calcification was noted in 96% (27 out of 28) of cases with bone formation. There was no correlation between the presence of bone formation and psammoma bodies (P=0.1264).
Correlation Between Disease-Free Survival and Presence of Psammoma Bodies, Stromal Calcification, or Bone Formation
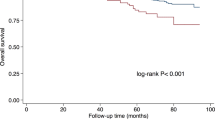
Disease-free survival was analyzed in 181 cases of papillary thyroid carcinomas by using the Kaplan–Meier method, together with the log-rank test. Survival was worse for patients with psammoma bodies. There was no correlation between survival and the presence of stromal calcification or bone formation (Figure 1). The 5- and 10-year disease-free survival rates are shown in Table 4.
Analyses of Risk Factors for Recurrence
We evaluated histological features, including psammoma bodies, stromal calcification, bone formation, as well as age, sex, tumor diameter, extrathyroid invasion, pT, gross lymph node metastasis, histological lymph node metastasis, stage grouping, and surgical margin as risk factors for recurrence (Table 5). Among these, psammoma bodies, age, extrathyroid invasion, pT, gross lymph node metastasis, histological lymph node metastasis, stage grouping, and surgical margin were found to be significant risk factors in univariate analysis. Subsequent multivariate analysis revealed that advanced age (≥60 years) and the presence of gross lymph node metastasis were independent predictors of recurrence; there was no such association for psammoma bodies (data not shown).
BMP Expression and Psammoma Bodies, Stromal Calcification, and Bone Formation
Quantitative expression of BMP-1 cDNA was analyzed in 48 cases of papillary thyroid carcinoma, and was expressed as the ratio between its expressions in tumor tissue and paired non-neoplastic tissue (T/N ratio). BMP-1 was upregulated in carcinoma tissue (T/N ratio>1) in 41 of 48 cases (85%). Psammoma bodies, stromal calcification, and bone formation were observed in 23, 25, and in 2 of the 48 cases, respectively. The correlations between BMP-1 expression and both psammoma bodies and stromal calcification are shown in Figure 2. Significantly higher expression of BMP-1 was observed in carcinoma tissues with, compared to those without, psammoma bodies or stromal calcification. A high expression of BMP-1 was also noted in two bone formation-positive cases (T/N ratio, 10.12 and 40.90), but more cases are needed before a definite conclusion can be reached.
There was no significant association between BMP-1 expression and any other clinicopathological parameters, including patient age, sex, tumor size, extrathyroid invasion, pT, lymph node metastasis (either gross or histological), and stage grouping evaluated using the Mann–Whitney U-test (data not shown).
Discussion
Calcification observed on thyroid ultrasonography can be classified as macro- or microcalcifications. Histological examination reveals that psammoma bodies mostly represent microcalcification.6 The presence of microcalcification is strongly suggestive of malignancy in preoperative diagnosis. Psammoma bodies are a diagnostic characteristic for papillary thyroid carcinoma, although they are occasionally observed in benign thyroid lesions.4, 6, 19, 20 In this study, we examined the clinical significance of psammoma bodies in papillary thyroid carcinoma, and found that patients with psammoma bodies are more likely to have gross lymph node metastasis and high-stage cancer (stage IVa). Moreover, gross lymph node metastasis was associated with an unfavorable prognosis, which is consistent with other reports.3, 24 Furthermore, papillary thyroid carcinoma patients with, compared to those without, psammoma bodies showed a significantly lower disease-free survival rate, which appears to be a novel finding. In summary, these findings suggest that the presence of psammoma bodies is a useful predictor of prognosis, although the association was not statistically significant in multivariate analysis. In 1985, Carcangiu et al25 reported that patients whose papillary thyroid carcinoma contained psammoma bodies were found to be affected more often by persistent disease on follow-up examination, and to have a higher incidence of nodal and pulmonary metastasis, but the differences were not statistically significant. Their results do not contradict our findings, as the papillary thyroid carcinomas found in our study represent a more homogenous entity, excluding all specific variants and poorly differentiated carcinomas, most concepts of which were developed only in the recent decades.
In our analyses, stromal calcification was significantly correlated with advanced age (≥60 years), high pT stage, and gross lymph node metastasis; bone formation was associated with older age. Unlike psammoma bodies, neither of them had an impact on disease-free survival. This is not surprising because psammoma bodies are a marker suggesting malignancy, whereas stromal calcification and bone formation frequently arise in benign lesions. Moreover, in our cases, advanced age (≥60 years) was significantly correlated with high pT (P<0.001). Stromal calcification was also correlated with advanced age; therefore, the significant correlation between stromal calcification and pT in our series was most likely because of the influence of age.
Little is known of the mechanisms responsible for the formation of psammoma bodies, stromal calcification, and bone formation. Morphologically, psammoma bodies were initially regarded as being formed by necrosis and calcification of intravascular or intralymphatic tumor thrombi;19 however, other opinions appeared, which claimed that psammoma bodies were formed by the intracellular calcifications in viable cells as the nidus. The most widely accepted opinion is that psammoma bodies in papillary thyroid carcinoma are found in association with tumor cells, within lymphatic spaces or within the tumor stroma.1 Stromal calcification arises because of calcium phosphate deposition in the fibrous stroma. In this study, 27 of 28 cases with bone formation also had adjacent or coexisting stromal calcification, which suggests that the bone arises from a preexisting calcification that underwent osseous metaplasia. At the molecular level, the BMP signal pathway was reported to be critical for calcification and bone formation.8, 11, 12, 13, 26 The BMP family has at least 30 members, among which BMPs- 1–7, which were initially isolated from demineralized bone matrix, are capable of inducing ectopic bone formation.8 BMP-1 is a kind of metalloproteinase with conserved domains, and can convert a variety of precursor proteins into mature or active forms that are involved in extracellular matrix formation.12, 13, 27, 28 It is BMP-1 that converts procollagen types I–III and VII into their mature forms and mediates C-terminal processing of procollagen V homotrimer.13, 27, 28 We noted an increased expression of BMP-1 in carcinoma tissues with psammoma bodies or with stromal calcification, which confirms the hypothesized role of BMP-1 in calcification and reveals a possible new role for BMP-1 in the formation of psammoma bodies.
We examined the frequency of PBS in 229 cases of papillary thyroid carcinoma and investigated its clinical significance and survival impact. The presence of psammoma bodies, but not of stromal calcification or bone formation, was a predictor of patient outcome. We also provided new information on the molecular processes responsible for the formation of psammoma bodies and stromal calcification in papillary thyroid carcinoma, namely, that BMP-1 is upregulated in carcinoma tissues with psammoma bodies or with stromal calcification.
References
LiVolsi VA, Mazzaferri EL, Schneider AB, et al. Papillary carcinoma In: Delellis RA, Lloyd RV, Heitz PU, Eng C, (eds). World Health Organization Classification of Tumors, Pathology and Genetics of Tumors of Endocrine Organs. International Agency for Research on Cancer: Lyon, France, 2004, pp 57–66.
Kakudo K, Tang W, Ito Y, et al. Papillary carcinoma of the thyroid in Japan: subclassification of common type and identification of low risk group. J Clin Pathol 2004;57:1041–1046.
Bai Y, Kakudo K, Li Y, et al. Subclassification of non-solid-type papillary thyroid carcinoma identification of high-risk group in common type. Cancer Sci 2008;99:1908–1915.
Consorti F, Anello A, Benvenuti C, et al. Clinical value of calcifications in thyroid carcinoma and multinodular goiter. Anticancer Res 2003;23:3089–3092.
Seiberling KA, Dutra JC, Grant T, et al. Role of intrathyroidal calcifications detected on ultrasound as a marker of malignancy. Laryngoscope 2004;114:1753–1757.
Triggiani V, Guastamacchia E, Licchelli B, et al. Microcalcifications and psammoma bodies in thyroid tumors. Thyroid 2008;18:1017–1018.
Wang N, Xu Y, Ge C, et al. Association of sonographically detected calcification with thyroid carcinoma. Head Neck 2006;28:1077–1083.
Hopkins DR, Keles S, Greenspan DS . The bone morphogenetic protein 1/Tolloid-like metalloproteinases. Matrix Biol 2007;26:508–523.
Hatakeyama S, Gao YH, Ohara-Nemoto Y, et al. Expression of bone morphogenetic proteins of human neoplastic epithelial cells. Biochem Mol Biol Int 1997;42:497–505.
Wolfman NM, McPherron AC, Pappano WN, et al. Activation of latent myostatin by the BMP-1/tolloid family of metalloproteinases. Proc Natl Acad Sci USA 2003;100:15842–15846.
Ge G, Greenspan DS . BMP1 controls TGFbeta1 activation via cleavage of latent TGFbeta-binding protein. J Cell Biol 2006;175:111–120.
Amano S, Scott IC, Takahara K, et al. Bone morphogenetic protein 1 is an extracellular processing enzyme of the laminin 5 gamma 2 chain. J Biol Chem 2000;275:22728–22735.
Lee S, Solow-Cordero DE, Kessler E, et al. Transforming growth factor-beta regulation of bone morphogenetic protein-1/procollagen C-proteinase and related proteins in fibrogenic cells and keratinocytes. J Biol Chem 1997;272:19059–19066.
Giannelli G, Falk-Marzillier J, Schiraldi O, et al. Induction of cell migration by matrix metalloprotease-2 cleavage of laminin-5. Science 1997;277:225–228.
Tsubota Y, Yasuda C, Kariya Y, et al. Regulation of biological activity and matrix assembly of laminin-5 by COOH-terminal, LG4-5 domain of alpha3 chain. J Biol Chem 2005;280:14370–14377.
Veitch DP, Nokelainen P, McGowan KA, et al. Mammalian tolloid metalloproteinase, and not matrix metalloprotease 2 or membrane type 1 metalloprotease, processes laminin-5 in keratinocytes and skin. J Biol Chem 2003;278:15661–15668.
Ge G, Hopkins DR, Ho WB, et al. GDF11 forms a bone morphogenetic protein 1-activated latent complex that can modulate nerve growth factor-induced differentiation of PC12 cells. Mol Cell Biol 2005;25:5846–5858.
Zuo H, Tang W, Yasuoka H, et al. A review of 227 cases of small papillary thyroid carcinoma. Eur J Surg Oncol 2007;33:370–375.
Johannessen JV, Sobrinho-Simoes M . The origin and significance of thyroid psammoma bodies. Lab Invest 1980;43:287–296.
Ellison E, Lapuerta P, Martin SE . Psammoma bodies in fine-needle aspirates of the thyroid: predictive value for papillary carcinoma. Cancer 1998;84:169–175.
Fluge O, Bruland O, Akslen LA, et al. Gene expression in poorly differentiated papillary thyroid carcinomas. Thyroid 2006;16:161–175.
Wang X, Nakamura M, Mori I, et al. Calcitonin receptor gene and breast cancer: quantitative analysis with laser capture microdissection. Breast Cancer Res Treat 2004;83:109–117.
Xu LL, Stackhouse BG, Florence K, et al. PSGR, a novel prostate-specific gene with homology to a G protein-coupled receptor, is overexpressed in prostate cancer. Cancer Res 2000;60:6568–6572.
Noguchi S, Murakami N, Kawamoto H . Classification of papillary cancer of the thyroid based on prognosis. World J Surg 1994;18:552–557.
Carcangiu ML, Zampi G, Pupi A, et al. Papillary carcinoma of the thyroid. A clinicopathologic study of 241 cases treated at the University of Florence, Italy. Cancer 1985;55:805–828.
Jasuja R, Ge G, Voss NG, et al. Bone morphogenetic protein 1 prodomain specifically binds and regulates signaling by bone morphogenetic proteins 2 and 4. J Biol Chem 2007;282:9053–9062.
Rattenholl A, Pappano WN, Koch M, et al. Proteinases of the bone morphogenetic protein-1 family convert procollagen VII to mature anchoring fibril collagen. J Biol Chem 2002;277:26372–26378.
Kessler E, Fichard A, Chanut-Delalande H, et al. Bone morphogenetic protein-1 (BMP-1) mediates C-terminal processing of procollagen V homotrimer. J Biol Chem 2001;276:27051–27057.
Acknowledgements
Part of the research fund was covered by a scholarship to Yanhua Bai from the Rotary Yoneyama Memorial Foundation, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Bai, Y., Zhou, G., Nakamura, M. et al. Survival impact of psammoma body, stromal calcification, and bone formation in papillary thyroid carcinoma. Mod Pathol 22, 887–894 (2009). https://doi.org/10.1038/modpathol.2009.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2009.38
Keywords
This article is cited by
-
Predictive Risk-scoring Model For Central Lymph Node Metastasis and Predictors of Recurrence in Papillary Thyroid Carcinoma
Scientific Reports (2020)
-
Papillary thyroid carcinoma: an ultrasound-based nomogram improves the prediction of lymph node metastases in the central compartment
European Radiology (2020)
-
In papillary thyroid carcinoma, expression by immunohistochemistry of BRAF V600E, PD-L1, and PD-1 is closely related
Virchows Archiv (2018)
-
PD-L1 and PD-1 expression are correlated with distinctive clinicopathological features in papillary thyroid carcinoma
Diagnostic Pathology (2017)
-
Correlation between Ultrasound Elastography and Histologic Characteristics of Papillary Thyroid Carcinoma
Scientific Reports (2017)