Abstract
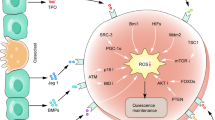
Myeloperoxidase (MPO), a pivotal lineage marker for acute myeloid leukemia (AML), has been also shown to have a prognostic value: a high percentage of MPO-positive blasts correlates to favorable prognosis. To understand the relationship between the expression of MPO in leukemia cells and the response to chemotherapeutic agents, we established MPO-expressing K562 leukemia cell lines and then treated them with cytosine arabinocide (AraC). Cells expressing wild-type MPO, but not mutant MPO that could not mature, died earlier of apoptosis than control K562 cells. Reactive oxygen species (ROS) were generated more in leukemia cells expressing MPO, and the generation was abrogated by MPO inhibitors or antioxidants. Tyrosine nitration of cellular protein also increased more in MPO-expressing K562 cells than control cells after treatment with AraC. In clinical samples, CD34-positive AML cells from high-MPO cases showed a tendency to be sensitive to AraC in the colony-formation assay, and the generation of ROS and the nitration of protein were observed only when the percentage of MPO-expressing cells was high. These data suggest that MPO enhances the chemosensitivity of AML through the generation of ROS and the nitration of proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR et al. Proposed revised criteria for the classification of acute myeloid leukemia. A report of the French–American–British Cooperative Group. Ann Intern Med 1985; 103: 620–625.
World Health Organization. Classification of Tumors. In: Jaffe ES, Harris NL, Stein H and Vardiman JW (eds). Pathology and Genetics of Tumors of Haematopoietic and Lymphoid Tissues. IARC Press: Lyon, 2001, pp. 79–80.
Hoyle CF, Gray RG, Wheatley K, Swirsky D, de Bastos M, Sherrington P et al. Prognostic importance of Sudan Black positivity: a study of bone marrow slides from 1386 patients with de novo acute myeloid leukaemia. Br J Haematol 1991; 79: 398–407.
Matsuo T, Cox C, Bennett JM . Prognostic significance of myeloperoxidase positivity of blast cells in acute myeloblastic leukemia without maturation (FAB: M1): An ECOG study. Hematol Pathol 1989; 3: 153–158.
Matsuo T, Kuriyama K, Miyazaki Y, Yoshida S, Tomonaga M, Emi N et al. The percentage of myeloperoxidase-positive blast cells is a strong independent prognostic factor in acute myeloid leukemia, even in the patients with normal karyotype. Leukemia 2003; 17: 1538–1543.
Modica-Napolitano JS, Singh KK . Mitochondria as targets for detection and treatment of cancer. Expert Rev Mol Med 2002; 4: 1–19.
Weijl NI, Cleton FJ, Osanto S . Free radicals and antioxidants in chemotherapy-induced toxicity. Cancer Treat Rev 1997; 23: 209–240.
Thannickal VJ, Fanburg BL . Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol 2000; 279: 1005–1028.
Fruehauf JP, Meyskens Jr FL . Reactive oxygen species: a breath of life or death? Clin Cancer Res 2007; 13: 789–794.
Mignotte B, Vayssiere JL . Mitochondria and apoptosis. Eur J Biochem 1998; 252: 1–15.
Winterbourn CC, Vissers MC, Kettle AJ . Myeloperoxidase. Curr Opin Hematol 2000; 7: 53–58.
Myzak MC, Carr AC . Myeloperoxidase-dependent caspase-3 activation and apoptosis in HL-60 cells: protection by the antioxidants ascorbate and (dihydro) lipoic acid. Redox Rep 7: 47–53.
Wagner BA, Buettner GR, Oberley LW, Darby CJ, Burns CP . Myeloperoxidase is involved in H2O2-induced apoptosis of HL-60 human leukemia cells. J Biol Chem 2000; 275: 22461–22469.
Morishita K, Kubota N, Asano S, Kaziro Y, Nagata S . Molecular cloning and characterization of cDNA for human myeloperoxidase. J Biol Chem 1987; 262: 3844–3851.
Nauseef WM, Brigham S, Cogley M . Hereditary myeloperoxidase deficiency due to a missense mutation of arginine 569 to tryptophan. J Biol Chem 1994; 269: 1212–1216.
Bullock O, Ray S, Krajewski S, Ibrado AM, Huang Y, Bhalla K . Intracellular metabolism of Ara-C and resulting DNA fragmentation and apoptosis of human AML HL-60 cells possessing disparate levels of Bcl-2 protein. Leukemia 1996; 10: 1731–1740.
Eiserich JP, Hristova M, Cross CE, Jones AD, Freeman BA, Halliwell B et al. Formation of nitric oxide-derived inflammatory oxidants by myeloperoxidase in neutrophils. Nature 1998; 391: 393–397.
Eiserich JP, Baldus S, Brennan ML, Ma W, Zhang C, Tousson A et al. Myeloperoxidase, a leukocyte-derived vascular NO oxidase. Science 2002; 296: 2391–2394.
Brennan ML, Wu W, Fu X, Shen Z, Song W, Frost H et al. A tale of two controversies defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J Biol Chem 2002; 227: 17415–17427.
Kuriyama K, Tomonaga M, Kobayashi T, Takeuchi J, Ohshima T, Furusawa S et al. Morphological diagnoses of the Japan adult leukemia study group acute myeloid leukemia protocols: central review. Int J Hematol 2001; 73: 93–99.
Miyazaki Y, Kuriyama K, Miyawaki S, Ohtake S, Sakamaki H, Matsuo T et al. Cytogenetic heterogeneity of acute myeloid leukaemia (AML) with trilineage dysplasia: Japan Adult Leukaemia Study Group-AML 92 study. Br J Haematol 2003; 120: 56–62.
Ambrosone CB, Ahn J, Singh KK, Rezaishiraz H, Furberg H, Sweeney C et al. Polymorphisms in genes related to oxidative stress (MPO, MnSOD, CAT) and survival after treatment for breast cancer. Cancer Res 2005; 65: 1105–1111.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Taguchi J, Miyazaki Y, Tsutsumi C, Sawayama Y, Ando K, Tsushima H et al. Expression of the myeloperoxidase gene in AC133 positive leukemia cells relates to the prognosis of acute myeloid leukemia. Leuk Res 2006; 30: 1105–1112.
Vercauteren SM, Sutherland HJ . CD133 (AC133) expression on AML cells and progenitors. Cytotherapy 2001; 3: 449–459.
Acknowledgements
This work was supported in part by grant from the Ministry of Health, Labour and Welfare of Japan. We deeply appreciate Dr T Matsuo for his thoughtful suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Rights and permissions
About this article
Cite this article
Sawayama, Y., Miyazaki, Y., Ando, K. et al. Expression of myeloperoxidase enhances the chemosensitivity of leukemia cells through the generation of reactive oxygen species and the nitration of protein. Leukemia 22, 956–964 (2008). https://doi.org/10.1038/leu.2008.8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.8
Keywords
This article is cited by
-
ROS-PIASγ cross talk channelizes ATM signaling from resistance to apoptosis during chemosensitization of resistant tumors
Cell Death & Disease (2014)
-
Expression of myeloperoxidase in acute myeloid leukemia blasts mirrors the distinct DNA methylation pattern involving the downregulation of DNA methyltransferase DNMT3B
Leukemia (2014)
-
Resveratrol ameliorates oxidative DNA damage and protects against acrylamide-induced oxidative stress in rats
Molecular Biology Reports (2012)
-
Expression of myeloperoxidase and gene mutations in AML patients with normal karyotype: double CEBPA mutations are associated with high percentage of MPO positivity in leukemic blasts
International Journal of Hematology (2011)
-
A prediction model for complete remission upon reinduction for patients with acute myeloid leukemia after failure of anthracycline and cytarabine standard chemotherapy
Annals of Hematology (2011)