Abstract
The interactions between fibroblasts and the extracellular matrix in wound contraction are mainly mediated via integrin signaling. Integrin-linked kinase (ILK) is a key mediator in integrin signal transduction. We investigated the role of ILK in cutaneous wound contraction. We found that ILK was involved in cutaneous wound healing in rats, and ILK and PI3K/AKT inhibitors inhibited wound contraction and re-epithelialization, consequently delaying wound healing in vivo. Further, using in vitro studies, we demonstrated that ILK and PI3K/AKT inhibitors suppressed the contraction of fibroblast-populated collagen lattices, inhibited fibroblast migration, and interrupted the effect of TGF-β1 on promoting alpha smooth muscle actin (α-SMA) expression in fibroblasts. When ILK expression was directly blocked by ILK small interfering RNA transfection, the migration and α-SMA expression of normal dermal fibroblasts were significantly suppressed as well. The data suggest that the ILK–PI3K/AKT signaling pathway mediates cutaneous wound contraction by regulating fibroblast migration and differentiation to myofibroblasts.
Similar content being viewed by others
Main
Wound contraction has an important role in full-thickness wound healing. Accurate wound contraction effectively reduces the wound area and shortens the healing time.1 However, excessive wound contraction contributes to hypertrophic scar formation.2 Two main concepts explain the mechanisms underlying wound contraction: (1) the force of wound contraction is generated from the crawling of fibroblasts, which migrate from the periphery to the center; (2) wound contraction is related to contractile forces in myofibroblasts.3 Fibroblast migration is required for the early phase of wound healing. During later stages, wound contraction is mainly attributed to myofibroblast contractile forces.1 The interaction between fibroblasts (or myofibroblasts) and the extracellular matrix (ECM) is necessary for wound contraction. Fibroblast-populated collagen lattice (FPCL) is a model for the study of wound contraction as well as cell–ECM interaction in vitro. In FPCL, fibroblasts are suspended in a collagen lattice to form a similar three-dimensional structure, with visible contractile function in vitro.4
The integrin signaling pathway has a crucial role in mediating the interaction between cells and the ECM. The biological signals are transmitted in cells via the transmembrane integrin receptors and activation of the signaling pathway.5 Integrin-linked kinase (ILK) was first identified and cloned by Hannigan et al in 1996 based on its interaction with the β1-integrin cytoplasmic domain. ILK is a serine/threonine kinase with three different and highly conserved domains.6 The ILK N-terminal ankyrin domain contains four ankyrin repeats, which mediate the crosstalk between ILK and other signaling pathways, such as the receptor tyrosine kinase signaling pathway.7, 8, 9 ILK’s central structure is a pleckstrin homology (PH)-like domain that binds with phosphatidylinositol-3-phosphate and regulates the catalytic activity of the enzyme.10 The ILK C terminal is a catalytic domain that interacts with the cytoplasmic domains of the integrin beta subunits. 11 The ILK C terminal phosphorylates AKT on Ser473 in a phosphatidylinositol-3 kinase (PI3K)-dependent manner12, 13, 14, 15 to regulate proliferation, migration, and other cell functions.10
Transforming growth factor-β1 (TGF-β1) is one of the most important cytokines related to wound healing and scar formation.16 It is involved in multiple aspects of wound healing, including ECM synthesis, fibroblast migration and proliferation regulation, and induction of alpha smooth muscle actin (α-SMA) expression.17 α-SMA is characteristically expressed by myofibroblasts, which are differentiated from fibroblasts. It contributes to wound contraction.18 Recent studies suggest that ILK is a key intracellular mediator in the ‘crosstalk’ between an integrin and the TGF-β1 pathway. ILK overexpression promotes α-SMA expression as well as epithelial–mesenchymal transition (EMT) in renal tubular epithelial cells.19 In renal tubular epithelial cells, TGF-β1 markedly upregulates ILK expression in a time- and dose-dependent manner, and TGF-β1 promotes the expression of fibronectin, snail 1, and MMP2, which is blocked by ILK inhibitor QLT0267.20
Previous studies reported that ILK may be involved in the regeneration of various tissues after injury. Overexpression of ILK in isolated stellate cells leads to enhanced motility and adhesion as well as increases in α-SMA and type-I collagen expression. ILK is involved in stellate cell activation and fibrogenesis during liver injury.21 Further, the ILK–PI3K/AKT pathway has an important role in nerve growth factor-mediated neurite outgrowth and thymosin β4-mediated cardiomyocyte migration, survival and repair after damage.22, 23 Thus, we hypothesize that ILK–PI3K/AKT pathway has a critical role in the course of cutaneous wound healing as well. In the current study, we detected ILK expression at different time points of dermal wound healing in rats using immunohistochemistry. We observed the effects of ILK inhibitor QLT0267 on rat wound contraction. Furthermore, we demonstrated the effect of ILK inhibitor QLT0267 and PI3K/AKT inhibitor LY294002 on FPCL contractility, cell proliferation, and migration, as well as TGF-β1-induced α-SMA expression. We directly targeted the ILK gene by ILK small interfering RNA (siRNA) transfection to observe the foregoing effects. We found that the ILK–PI3K/AKT pathway is involved in wound contraction by regulating fibroblast proliferation and migration as well as myofibroblast differentiation.
MATERIALS AND METHODS
Rat Wound Surgery and Treatment
Adult, female Sprague-Dawley rats were anesthetized by intraperitoneal injection of 1% pentobarbital sodium 3 ml/kg, and the dorsal skin was shaved. After sterilizing with 70% ethanol, two 2.5 × 2.5-cm full-thickness excisional wounds were created on both sides of the spine. Wound tissue specimens were collected on days 1, 5, 10, 15, and 20 after injury. We detected ILK expression in the normal skin and wound tissue specimens using immunohistochemical staining.
We divided the injured rats into three groups. We injected the treated rats with 5 ml of QLT0267 (at a concentration of 5 μM) around the wound tissue once daily after injury. In the control rats, we injected the area surrounding the wound tissue with 5 ml of diluted dimethyl sulfoxide (DMSO) or physiological saline. We photographed each wound daily using a digital camera and analyzed the wound area using Image-Pro Plus Software. We calculated the wound closure percentage at each time point using the following formula: (1−(current wound area/initial wound area)) × 100.
Histology and Immunohistochemical Staining
We immediately fixed the wound tissue specimens with paraformaldehyde and embedded them in paraffin after dehydration. Then, we collected 4-μm-thick sections and incubated them at 60 °C for 2 h before staining. Sections were stained with H&E for histological analysis. Average epidermal thickness of each section was calculated by measuring five random microscopic fields at 100 times. For immunohistochemical staining, sections were blocked with 3% hydrogen peroxidase for 10 min and normal goat serum for 20 min. Sections were then incubated with rabbit anti-mouse-ILK monoclonal antibody (Abcam, UK) or rabbit anti-mouse-p-AKT monoclonal antibody (Abcam), overnight at 4 ºC, followed by incubation with a biotin-labeled anti-rabbit secondary antibody (Maixin-Bio, China) for 1 h at room temperature. The antibody location was visualized with peroxidase-labeled streptavidin (Maixin-Bio) and diaminobenzidine (Maixin-Bio). The slides were then counterstained with hematoxylin.
Cell Culture
Normal human skin fibroblasts were obtained from cell outgrowths in ablated adult normal skin explants, as previously described.24 We cut the dermal specimens into ~1 mm3 explants and transferred them into culture flasks with 10 ml of Dulbecco-modified Eagle’s medium (DMEM; Gibco, USA) supplemented with 10% fetal calf serum (Gibco). The dermal specimens were cultured in a humidified incubator at 37 °C with a 5% carbon dioxide atmosphere. The dermal specimens adhered to the culture flasks, and after 24 h we changed the culture media. We changed the culture media every 2 days until the normal human skin fibroblasts migrated out of the tissue explants and reached confluency. The primary cells were passaged 1:1 by trypsinization and then successively passaged at confluency. We passaged cells four to six times in our study, which was approved by the local Ethics Committee, following informed consent from all donors.
siRNA Transfection
Normal human skin fibroblasts were transfected with two series of siRNA (Abnova, Taiwan, target sequence of ILK siRNA1: ggcacggatcaatgtaatg, target sequence of ILK siRNA2: ttctcgcatccaaatgtgc specifically targets the ILK gene, at 50–60% confluency, using 10 μl Lipofectamine 2000 (Invitrogen, USA) in Opti-MEM (Invitrogen) medium for 6 h according to the manufacturer’s instructions. Meanwhile, the fibroblasts were transfected with control siRNA, which carried non-sense RNA sequences as control. The transfected cells were collected at 24 h after incubation in DMEM for determination.
QLT0267 and LY294002 Treatment
Highly specific small-molecule inhibitors QLT0267 (Dermira, CA) and LY294002 (Sigma-Aldrich, USA) were dissolved in DMSO (Sigma, USA) at 10 mM concentration, and reconstituted to 1.60 mg of QLT0267 or 1.72 mg of LY294002 in 500 μl DMSO, respectively. The solution was filtered through a 0.22-μM pore and stored at –80 °C. Before usage, the frigorific solution was thawed and diluted to the needed concentrations. A 500-μl solution of DMSO was diluted into equal volumes as control.
Cell Proliferation Assay
We determined the cell proliferation and cytotoxicity using a 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)carbonyl]-2H-tetrazolium hydroxide (XTT, Sigma-Aldrich) assay, as previously described.25 We plated normal human skin fibroblasts at 7500 cells per well in 96-well plates and incubated overnight for adherence. We added a XTT/PMS (phenazine methosulfate, Sigma-Aldrich) mixture to the medium at different time points after treatment and determined the water-soluble XTT–formazan product absorbance (450 nm) in a microplate reader (Bio-Rad, USA) after incubation at 37 °C for 3 h. The cell proliferation was calculated as mean absorbance of cells treated divided by the mean absorbance of untreated cells. All assays were performed in triplicate.
Fibroblast-Populated Collagen Lattices
FPCLs were prepared as follows. We suspended 1 × 105 cells in 1 ml of serum-free DMEM culture medium. The cell suspension was combined with 0.954 ml of Cultrex 3-D Culture Matrix Rat Collagen I (R&D Systems, USA). Immediately, 0.046 ml of sterile sodium hydroxide (1 M) was added to the mixture to achieve neutral pH. The solution was mixed gently to avoid gas bubble formation. All the above procedures were performed on ice. The 2 ml mixture was delivered to a 35-mm-diameter well within a six-well culture plate and polymerized to form an FPCL by incubating at 37 °C for 10 min. After casting, 2 ml of DMEM culture media with 10% fetal calf serum was added to the wells. Subsequently, the FPCLs were detached from the wells gently with a glass rod and cultured in a 37 °C incubator in a 5% carbon dioxide atmosphere for 6 h. After cell extension, the FPCLs were cultured in DMEM (with 10% fetal calf serum) containing LY294002 (at a final concentration of 50 μM, according to the instructions) or QLT0267 (at a final concentration of 5 μM). Untreated FPCLs and DMSO-treated FPCLs were used as controls. The culture medium was changed every 2 days. We photographed FPCLs at 0, 12, 24, 48, and 72 h, respectively. Image pro plus software was used to calculate the FPCL area and contraction rate. The contraction rate at each time point was calculated by the following formula: (1−(current wound area/initial wound area)) × 100.
Scratched Cell Migration Assay and Transwell Cell Migration Assay
In the scratched cell migration assay, 5 × 105 cells were plated in a six-well culture plate and grown to 80% confluency. The cell monolayer centers were scraped with a sterile micropipette tip to create a denuded gap after removing the medium. The cell debris was washed out with PBS solution and the cells cultured in serum-free DMEM culture media for 8 h at 37 °C and 5% carbon dioxide. Subsequently, the culture medium was replaced with DMEM (with 10% fetal calf serum) containing LY294002 (at a final concentration of 50 μM) or QLT0267 (at a final concentration of 5 μM). Untrtated cells and DMSO-treated cells were used as controls. We photographed them at 0, 8, and 24 h after scratching with an inverted phase contrast microscope (Nikon, JP). The mobility was calculated using the following formula: (1−(current denuded zone area/initial denuded zone area)) × 100.
Further, transwell inserts (Corning, USA; 8.0-μM pore size, polycarbonate membrane) were adopted to determine mobility. The bottom chamber in the transwell chamber was filled with DMEM culture media containing 10% fetal calf serum. We suspended cells 24 h after treatment or transfection at a density of 1.0 × 106 cells per ml in 100 nl DMEM (Gibco) without serum and placed them in the upper chamber. The cells were incubated for 10 h at 37 °C in a 5% carbon dioxide atmosphere. We removed the medium, and cells that failed to migrate through the polycarbonate membrane in the upper chamber were scraped with a cotton tip. Subsequently, we removed the polycarbonate membrane and stained it with hematoxylin for 10 min after fixing it in 4% paraformaldehyde. We detected the average cell number in five randomly chosen fields using a light microscope.
Immunofluorescence Staining
Cells were fixed with 4% paraformaldehyde, permeabilized with 0.3% Triton X-100, and washed. They were incubated with rabbit anti-human-ILK monoclonal antibody (Abcam) overnight at 4 °C followed by CY3-labeled anti-rat secondary antibodies (Abcam). Nuclei were stained with 4′,6-diamidino-2-phenylindole (Sigma-Aldrich) and image acquisition was performed by laser confocal microscopy (Carl Zeiss, Jena, Germany).
Western Blot
Western blot assays were performed as previously described. Cells were lysed (Bio-Rad), and the membranes were probed with rabbit anti-human-ILK monoclonal antibody (Abcam), rabbit anti-α-SMA monoclonal antibody (Abcam), rabbit anti-AKT monoclonal antibody (Abcam), rabbit anti-p-AKT monoclonal antibody (Abcam), and rabbit anti-GAPDH monoclonal antibody (Abcam) as an internal reference overnight at 4 °C. Membranes were washed in RIPA lysis buffer (Beyotime, CHN) and centrifuged at 12 000 g and 4 °C for 5 min to extract the total protein. The total protein concentration was measured using the bicinchoninic acid assay kit (Beyotime, Cnina) according to the manufacturer’s instructions. We separated 20 nl of total protein in each sample hole using 10% SDS-PAGE and transferred to a nitrocellulose membrane. The proteins were then washed with TBST and incubated with horseradish peroxidase-labeled goat anti-rabbit secondary antibody (Beyotime, CHN) for 1 h at 37 °C, and incubated with enhanced chemiluminescence (Thermo, USA) for 5 min in the dark at room temperature. The photographs were collected using a gel imaging system (Bio-Rad) and analyzed using Image Lab software (Bio-Rad).
Statistical Analysis
Data are expressed as means±s.e.m. Statistical analysis was performed using an independent Student’s t-test or one-way ANOVA. P-values <0.05 were considered statistically significant.
RESULTS
ILK Mediates Rat Wound Healing
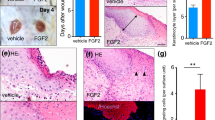
To investigate ILK expression in the course of wound healing and its effects in wound contraction in vivo, we first determined the ILK expression at different time points by immunohistochemistry. We found that ILK expression was mainly located in the basal layer of the epidermis, hair follicles, and fibroblasts. In normal skin, the intensity of expression was low. After injury, the ILK expression was significantly upregulated from day 5 to day 15, reaching the maximum on day 5 when the greatest degree of wound contraction was observed. It then rapidly declined by day 20 following complete recovery (Figures 1a and b). The percentage of wound closure was measured after ILK inhibitor QLT0267 treatment. DMSO or physiological saline treatment was used as a control. We found that after QLT0267 treatment, the p-AKT expression remarkably declined (Figures 1e and f). Further QLT0267 treatment significantly inhibited wound contraction. The percentage of wound closure after QLT0267 treatment was markedly lower than after DMSO or physiological saline treatment at each time point from day 5 to day 20. Then, average epidermal thickness was determined by H&E histological analysis. The epidermis of QLT0267-treated wound was significantly thinner than that of the DMSO- or physiologically treated wound. In QLT0267-treated rats, the wounds finally healed at 27.4±3.7 days. However, DMSO-treated wounds healed at 20.6±2.3 days, and the physiological saline-treated wounds healed at 19.5±3.8 days. Thus, wound healing was significantly delayed following QLT0267 treatment (Figures 1c).
ILK mediates rat wound healing in vivo. We created full-thickness excisional wounds on rats dorsally. (a and b) We collected wound tissue specimens on days 1, 5, 10, 15, and 20 after injury and detected ILK expression in normal skin and wound tissue specimens using immunohistochemical staining (*P<0.05, compared with normal skin, #P<0.05 compared with day 20). (c and d) We injected QLT0267 into tissues surrounding the wound once daily after injury and injected them with DMSO or physiological saline as a control. We calculated the percentage of wound closure using the following formula: (1−(current wound area/initial wound area)) × 100. (e and f) The expression of p-AKT in wound at different time points were determined by immunohistochemical staining (*P<0.05, compared with DMSO treated wounds, #P<0.05 compared with physiological saline treated wounds). (g and h) The wound tissue specimens were stained with H&E for histological analysis. The average epidermal thickness of each section was calculated by measuring five random microscopic fields at 100 times. (*P<0.05, compared with DMSO-treated wounds, #P<0.05 compared with physiological saline-treated wounds). DMSO, dimethyl sulfoxide; ILK, integrin-linked kinase.
Human Skin Fibroblast Proliferation and AKT Phosphorylation After Treatment with Different QLT0267 Concentrations
After 48 h of treatment with DMSO and QLT0267 at concentrations of 5, 10, 20, and 30 μM, respectively, we found that the DMSO and 5 μM QLT0267 treatment had no effect on cell morphology and numbers. However, when the concentrations exceeded 5 μM and increased gradually, the cell number and filopodia decreased in a concentration-dependent manner (Figure 2a). We used an XTT assay to determine the cell proliferation ability within 48 h. The results suggested that the proliferation of cells treated with 10, 20, and 30 μM QLT0267 was significantly suppressed compared with cells treated with 5 μM QLT0267. However, DMSO and 5 μM QLT0267 treatments failed to alter cell proliferation compared with non-treated cells (Figure 2b). Furthermore, we detected the ILK downstream AKT and p-AKT by western blot assays after treatment with different QLT0267 concentrations. We found that any concentration of QLT0267 effectively inhibited AKT phosphorylation. The p-AKT expression gradually decreased with increasing QLT0267 concentration (Figure 2c). The 5 μM QLT0267 treatment significantly inhibited ILK catalytic activity without visible cytotoxicity. Therefore, 5 μM was selected as the optimal concentration for our study.
Human skin fibroblast proliferation and AKT phosphorylation were inhibited by QLT0267 in a dose-dependent manner. We isolated normal human skin fibroblasts from cell outgrowths in ablated normal skin explants of adults and treated with different QLT0267 concentrations. (a and b) After 48 h of treatment, we photographed the cell morphology (a) and detected cell proliferation using an XTT assay (b). (c) The cells were lysed to detect AKT and p-AKT expression using the western blot 48 h after exposure to different QLT0267 concentrations (*P<0.05 compared with untreated cells). DMSO, dimethyl sulfoxide; XTT, 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)carbonyl]-2H-tetrazolium hydroxide.
QLT0267 and LY294002 Inhibit FPCL Contraction
To investigate the effects of ILK and downstream PI3K/AKT on wound contraction in vitro, FPCLs were cultured in the medium supplemented with ILK inhibitor QLT0267 (at a final concentration of 5 μM) or PI3K inhibitor LY294002 (at a final concentration of 50 μM) for 72 h, using the solvent DMSO-treated and -untreated FPCLs as controls. We measured FPCL contraction rates using Image-Pro Plus at different time points. We found that collagen lattices without fibroblasts had no contractibility (data not shown). FPCLs contracted starting from 12 h after formation. QLT0267 and LY294002 remarkably suppressed FPCL contraction from 24 to 72 h when compared with control. No significant difference was found between DMSO-treated and -untreated FPCLs at each time point. QLT0267- or LY294002-treated FPCL contraction curves were always lower than those of the control (Figures 3a and b). Due to stronger contraction, the cell densities of DMSO-treated and -untreated FPCLs were significantly higher than QLT0267- or LY294002-treated FPCLs, and additional stretch marks were found in DMSO-treated and -untreated FPCLs (Figure 3c).
FPCL contractility was suppressed by QLT0267 or LY294002. We constructed FPCLs with normal human skin fibroblasts and cultured them in DMEM (supplemented with 10% fetal calf serum) containing 5 μM QLT0267 or 50 mM LY294002. Untreated FPCLs and DMSO-treated FPCLs were used as controls. (a) We photographed FPCLs at 0, 12, 24, 48, and 72 h, respectively. (b) We calculated the contraction rates using the following formula: (1−(current wound area/initial wound area)) × 100. (c) We obtained microscopic FPCL images using an inverted phase contrast microscope at 72 h. DMEM, Dulbecco-modified Eagle’s medium; DMSO, dimethyl sulfoxide; FPCL, fibroblast-populated collagen lattices.
Effect of ILK–PI3K/AKT Pathway on Human Skin Fibroblast Migration In vitro
To investigate the inhibitory effects of ILK–PI3K/AKT pathway on human skin fibroblast migration, a scratch wound-healing assay and a transwell assay were employed in vitro. In the scratch wound-healing assay, the cells that migrated to the denuded zone were counted after incubation with QLT0267 (at a final concentration of 5 μM) and LY294002 (at a final concentration of 50 μM) for 24 h. The outcomes demonstrated that the QLT0267 or LY294002 treatment equally suppressed cell migration to the denuded zone (Figures 4a and b). In the transwell assay, we also detected that either QLT0267 or LY294002 treatment significantly inhibited human skin fibroblast migration through the transwell chamber compared with DMSO-treated and -untreated cells (Figures 4c and d). Further, fibroblasts were transfected with two series of ILK siRNA to block ILK expression, using untreated and control siRNA as controls. The migration was detected by transwell assay after transfection. The data show that ILK siRNA transfection significantly inhibited fibroblast migration.
Human skin fibroblast migration was inhibited by QLT0267 or LY294002 treatments. (a and b) Human skin fibroblasts were isolated from adult normal skin and treated with QLT0267 or LY294002 to inhibit the catalytic activity of ILK and AKT (using untreated cells and DMSO-treated cells as controls). We determined human skin fibroblast migration with a scratched cell migration assay. Photographs were taken at 0, 12, and 24 h after scratching. We calculated the migration rate using the following formula: (1−(current denuded zone area/initial denuded zone area)) × 100. (c and d) Human skin fibroblasts were treated as above. We determined the migration ability 24 h after treatment using a transwell cell migration assay. Cells migrating through the polycarbonate membrane were counted by detecting the average cell number in five randomly chosen fields using a light microscope (*P<0.05 compared with untreated cells, #P<0.05 compared with DMSO-treated cells). (e and f) Human skin fibroblasts were transfected with two series of ILK siRNA to specifically target ILK gene, control siRNA-transfected and -untreated fibroblasts were as control. The migration ability was determined 24 h after treatment using a transwell cell migration assay. The number of migration cells was calculated as above. (*P<0.05 compared with untreated cells, #P<0.05 compared with control siRNA-transfected cells). DMSO, dimethyl sulfoxide; ILK, integrin-linked kinase; siRNA, small interfering RNA.
Role of ILK–PI3K/AKT Pathway in Differentiation of Fibroblasts to Myofibroblasts
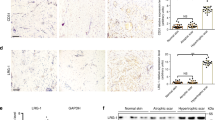
Myofibroblasts, which specifically generated α-SMA protein, have a critical role in the contraction of wound granulation tissue. TGF-β1 is the key cytokine promoting the transition of fibroblasts to myofibroblasts. To investigate the effects of ILK on fibroblast differentiation, we utilized the ILK inhibitor QLT0267 (at a final concentration of 5 μM) and PI3K/AKT inhibitor LY294002 (at a final concentration of 50 μM) to attenuate their activity in human skin fibroblasts, respectively, and detected α-SMA expression using the western blot. Human skin fibroblasts were treated with TGF-β1 10 ng/l as well as combined with QLT0267 (at a final concentration of 5 μM) or LY294002 (at a final concentration of 50 μM). We detected α-SMA and ILK expression using immunofluorescence and western blot. We found that QLT0267 and LY294002 did not alter the expression of ILK in fibroblasts, and directly attenuated the kinase activity (Figures 5b and c). Our data show that in addition to QLT0267, LY294002 treatment also depleted α-SMA expression in human skin fibroblasts effectively (Figure 5c). TGF-β1 treatment enhanced ILK and α-SMA expression, which was abolished by treatment with QLT0267 or LY294002 (Figures 5a and b).
ILK/AKT pathway mediates TGF-β1-induced α-SMA expression and myofibroblast differentiation in human skin fibroblasts. (a) Human skin fibroblasts were treated with TGF-β1 10 ng/l for 24 h, and detected ILK expression using immunofluorescence staining. Image acquisition was performed using laser confocal microscopy (*P<0.05 compared with untreated cells). (c) We treated human skin fibroblasts with QLT0267 or LY294002 for 24 h (untreated cells and DMSO-treated cells served as control), and determined ILK and α-SMA expressions using the western blot (*P<0.05 compared with untreated cells, #P<0.05 compared with DMSO-treated cells). (b) We cultured human skin fibroblasts using TGF-β1 10 ng/l treatment alone and in combination with QLT0267 or LY294002. We determined ILK and α-SMA expression 24 h after treatment using the western blot (*P<0.05 compared with untreated cells, #P<0.05 compared with DMSO-treated cells). (d) Human skin fibroblasts were transfected with two series of ILK siRNA to specifically target ILK gene, using control siRNA-transfected and -untreated fibroblasts as controls. We determined the expression of ILK, AKT, p-AKT, and α-SMA 24 h after transfection using western blot (*P<0.05 compared with untreated cells, #P<0.05 compared with control siRNA-transfected cells). α-SMA, alpha smooth muscle actin; DMSO, dimethyl sulfoxide; ILK, integrin-linked kinase; siRNA, small interfering RNA; TGF-β1, transforming growth factor-β1.
To determine the role of ILK, fibroblasts were transfected with two series of ILK siRNA to block the ILK expression, using untreated and control siRNA as the control. The expression of ILK, AKT, p-AKT, and α-SMA was detected by western blot. Data show that ILK siRNA transfection significantly suppressed the expression of ILK. Conversely, untreated and control siRNA transfection failed to alter ILK expression. Meanwhile, we found that the phosphorylated AKT was markedly decreased after ILK siRNA transfection, without significantly altering the total AKT expression. Further, the expression of α-SMA was significantly inhibited by silencing ILK expression (Figure 5d).
DISCUSSION
Wound contraction is essential for open-wound repair. Wound contraction reportedly accounts for 88% of wound closure, whereas scar deposition only contributes to 12%. The driving force of wound contraction is mainly generated by fibroblast migration and constriction and drawing the surrounding tissues into the lesion by attaching to ECMs.26 Several factors including multiple cell types, ECM components, various cytokines, and cytoskeletal components, are involved in wound contraction.1
Integrins mediate the communication between cells and ECMs. ECMs bind to the transmembrane integrin receptors and activate the integrin signaling pathway. Consequently, extracellular signals are transferred into the cytoplasm to regulate various cellular functions.27 Fetal and adult human skin fibroblasts display intrinsic differences in integrin expression and contractile capacity following TGF-β stimulation, and integrins may contribute to the differences in wound contraction between fetal subjects and adults.28 ILK is the key kinase in the integrin signaling pathway and further phosphorylates downstream AKT, glycogen synthase kinase-3β (GSK-3β), MAPK, and other kinases to regulate cell proliferation, apoptosis, migration, and cycle. ILK reportedly has important roles in malignant tumor formation and invasion, tissue and organ fibrosis, angiopoiesis, regeneration after injury, and skin follicle morphogenesis.29, 30 A previous study in vitro showed that ILK may be involved in scar contracture, and ILK overexpression in cells markedly enhances cell migration and FPCL contractility.31 Our in vivo study results indicate that ILK mediates wound healing and contributes to wound contraction. Complete ILK function is crucial for wound contraction. Further, we isolated normal human skin fibroblasts to investigate the mechanism of ILK in wound contraction in vitro.
Collagen gel without fibroblasts showed no contractibility. However, FPCL-containing fibroblasts exhibited various degrees of contractility. FPCL is utilized generally to simulate the process of wound contraction in vitro. A contracted FPCL containing 0.5 × 105 cell per ml appears as a flattened disc. However, an FPCL containing 5 × 105 cell per ml has a doughnut-like appearance due to the high density of cells accumulating at the periphery. Monoclonal α2β1 integrin antibody anti-CD49b significantly inhibited the contraction of FPCLs (0.5 × 105 cell per ml) by 50%, but not that of FPCLs at a higher density of 5 × 105 cell per ml. Fibroblast interaction with collagen fibers in the FPCLs (0.5 × 105 cell per ml) is α2β1 integrin dependent.32 Therefore, we selected the cell concentration of 0.5 × 105 cell per ml to construct FPCLs. Isolated wound fibroblasts enhanced FPCL contractility compared with normal dermal fibroblasts. Wound fluid also stimulated FPCL contraction by normal dermal fibroblasts, but not by wound fibroblasts.33 This result indicated differences in wound contraction between wound fibroblasts and normal dermal fibroblasts. Our results in vitro confirmed that fibroblasts with impaired ILK or AKT function led to deficient FPCL contractility, suggesting that ILK and AKT are required for wound contraction.
The migration of wound fibroblasts are crucial for wound contraction, especially in the early phases. Following injury, fibroblasts at the periphery of a wound proliferate abundantly and migrate toward the center. The healthy tissues surrounding the wound are drawn into the lesion.1 By regulating the migration of fibroblasts, we may control wound contraction. ILK effects on cell migration and proliferation have been confirmed in various cell types, such as malignant tumor cells, cardiac stem cells, and endothelial cells. ILK knockdown impairs melanoma cell migration, which is associated with reduced stress fiber formation, cell spread, and cell adhesion.34 The downstream Pi3 K/AKT pathway was also involved in vascular endothelial growth factor-mediated cardiac stem cell migration.35 In ILK knockout endothelial cells, the proliferation was significantly inhibited by obstructing the transition from the G0–G1-phase to S-phase and suppressing cyclin D1-expression. The migration was suppressed by disrupting the F-actin assembly.36
Our study confirmed that the ILK/AKT pathway was possibly involved in wound contraction by regulating wound fibroblast migration. ILK-mediated regulation of migration may be related to fibronectin. ILK knockdown strongly inhibited intestinal cell spread and migration by decreasing fibronectin deposition, and the inhibition was reversed with exogenously deposited fibronectin.37 Further, the ILK/Pi3K/AKT mediated fibronectin peptide-induced actin filament rearrangement to form cell motility structures, and increase cell migration and invasion.38 Besides, PI3K/AKT acts via IKK α/β to increase p65 phosphorylation at Ser536 and enhance NF-κB transactivation, which leads to β1-integrin expression and increased cell migration.39, 40, 41
TGF-β signaling has an important role in wound contraction. TGF-β type II receptor knockout results in reduced collagen deposition, remodeling, and wound contraction. The mediators of cell–matrix interaction, including α1, α2, and β1 integrins, were reduced by blocking TGF-β signaling.42 Fibroblasts overexpressing the TGF-β1 gene increase α-SMA expression and enhance FPCL contraction, and the TGF-β/Smad pathway is involved in inducing α-SMA expression.18, 43 α-SMA is characteristically expressed by myofibroblasts in the skin and is considered a marker of differentiation of fibroblasts to myofibroblasts. Myofibroblast differentiation is widely considered a crucial factor contributing to wound and scar contraction.44 Smad3 knockout blocks TGF-β1-induced α-SMA expression, and α-SMA-depleted human lung fibroblasts lack the ability to contract the collagen gels with or without TGF-β1, indicating that Smad3 is required for α-SMA expression and α-SMA is required for gel contraction.45 Recent studies demonstrated that ILK mediates pathological fibrosis, tumorigenesis, and metastasis by mediating TGF-β-induced EMT in diverse cell types, such as mammary epithelial cells and renal tubular epithelial cells. α-SMA is a marker of EMT, and ILK knockout significantly interrupts TGF-β-induced EMT as well as α-SMA expression.46
Our results indicate that the ILK/AKT pathway probably mediates TGF-β1-induced α-SMA expression and myofibroblast differentiation in dermal fibroblasts. A previous study determined that ILK-deficient fibroblasts treated with TGF-β1 exhibited attenuation of Smad 2 and 3 phosphorylation accompanied by impaired transcriptional activation of Smad targets, such as α-SMA. Therefore, ILK regulates the differentiation of fibroblasts to myofibroblasts is probably attributed to the TGF-β/Smad pathways.47, 48
The role of ILK as a kinase is disputed, because ILK lacks specific amino-acid residues thought to be essential for phosphotransferase activity.49 However, similar deficiencies in canonical residues occur in the catalytic domains of other kinases such as Wnk1, VRK3, CASK, and ErbB3, which possess protein kinase activity.50 In our study, we found that the phosphorylation activity of AKT was significantly inhibited by ILK siRNA transfection, suggesting that, at least in vitro, ILK is indeed a protein kinase. QLT0267 was initially synthesized and widely used as an ILK inhibitor, because QLT0267 effectively suppressed the downstream AKT and GSK-3β phosphorylation by the inhibition of ILK.51 It does not target other components of the PI3K pathway including GSK-3β, PDK1, and AKT subunits (Product Monograph of QLT0267, Dermira Inc.). However, a recent study found that the specificity of QLT0267 was limited. QLT0267 inhibited both ILK and FLT-3 in acute myeloid leukemia samples. However, further evidence is needed to determine whether QLT0267 directly inhibited FLT-3 or indirectly via ILK-related pathways.52 In our study, we observed that QLT0267 effectively inhibited ILK downstream AKT phosphorylation in a concentration-dependent manner.
In conclusion, our in vivo study demonstrated for the first time that ILK mediated the entire process of rat skin wound healing. A positive correlation existed between the ILK expression levels and the rate of wound contraction. ILK inhibitors effectively inhibited wound contraction and re-epithelialization, consequently delaying wound healing. Further, in our in vitro study, we found that the ILK inhibitor QLT0267 and the AKT inhibitor LY294002 displayed similar function, including inhibition of FPCL contraction, suppression of normal dermal fibroblast migration, and TGF-β1-induced α-SMA expression. When ILK expression was directly blocked by ILK siRNA transfection, the migration and α-SMA expression of normal dermal fibroblasts were significantly suppressed as well. Our study suggests that ILK has a crucial role in wound and FPCL contraction, probably due to the regulation of wound fibroblast migration and fibroblast-to-myofibroblast differentiation. Therefore, ILK is a potential molecular target in regulating wound contraction and treating excessive scar contracture. The ILK inhibitor QLT0267 and the AKT inhibitor LY294002 may find clinical application in the future.
References
Nedelec B, Ghahary A, Scot PG et al. Control of wound contraction. Basic and clinical features. Hand Clin 2000:16:289–302.
Yang L, Witten TM, Pidaparti RM . A biomechanical model of wound contraction and scar formation. J Theor Biol 2013:332:228–248.
Robert TT, Murray JD . Mechanistic model of wound contraction. J Surg Res 1993:55:233.
Bell E, Ivarsson B, Merrill C . Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc Natl Acad Sci USA 1979:76:1274–1278.
Ahmed N, Riley C, Rice G et al. Role of integrin receptors for fibronectin, collagen and laminin in the regulation of ovarian carcinoma functions in response to a matrix microenvironment. Clin Exp Metastasis 2005:22:391–402.
Hannigan GE, Hagesteijn LC, Gibbon FL et al. Regulation of cell adhesion and anchorage-dependent growth by a new β1-integrin-linked protein kinase. Nature 1996:379:91–96.
Legate KR, Montanez E, Kudlacek O et al. ILK, PINCH and parvin: the tIPP of integrin signalling. Nat Rev Mol Cell Biol 2006:7:20–31.
Tu Y, Li F, Goicoechea S et al. The LIM-only protein PINCH directly interacts with integrin-linked kinase and is recruited to integrin-rich sites in spreading cells. Mol Cell Biol 1999:19:2425–2434.
Tu Y, Li F, Wu C . Nck-2, a novel Src homology2/3-containing adaptor protein that interacts with the LIM-only protein PINCH and components of growth factor receptor kinase-signaling pathways. Mol Biol Cell 1998:9:3367–3382.
McDonald PC, Fielding AB, Dedhar S . Integrin-linked kinase-essential roles in physiology and cancer biology. J Cell Sci 2008:121:3121–3132.
Nikolopoulos SN, Turner CE . Molecular dissection of actopaxin-integrin-linked kinase-Paxillin interactions and their role in subcellular localization. J Biol Chem 2002:277:1568–1575.
Delcommenne M, Tan C, Gray V et al. Phosphoinositide-3-OHkinase-dependent regulation of glycogen synthase kinase 3 and protein kinase B/AKT by the integrin-linked kinase. Proc Natl Acad Sci USA 1998:95:11211–11216.
Troussard AA, Tan C, Yoganathan TN et al. Cell-extracellular matrix interactions stimulate the AP-1 transcription factor in an integrin-linked kinase- and glycogen synthase kinase 3-dependent manner. Mol Cell Biol 1999:19:7420–7427.
Lynch DK, Ellis CA, Edwards PA et al. Integrin-linked kinase regulates phosphorylation of serine 473 of protein kinase B by an indirect mechanism. Oncogene 1999:18:8024–8032.
Hannigan GE, McDonald PC, Walsh MP et al. Integrin-linked kinase: not so 'pseudo' after all. Oncogene 2011:30:4375–4385.
Mamta S, Don R, Sarah H et al. Role of elevated plasma transforming growth factor-beta 1 levels in wound healing. Am J Pathol 1999:154:1115–1124.
Massague J . How cells read TGF-beta signals. Nat Rev Mol Cell Biol 2000:1:169–178.
Campaner AB, Ferreira LM, Gragnani A et al. Upregulation of TGF-beta1 expression may be necessary but is not sufficient for excessive scarring. J Invest Dermatol 2006:126:1168–1176.
Serrano I, McDonald PC, Lock FE et al. Role of the integrin-linked kinase (ILK)/Rictor complex in TGF beta-1-induced epithelial-mesenchymal transition (EMT). Oncogene 2013:32:50–60.
Li Y, Tan X, Dai C et al. Inhibition of integrin-linked kinase attenuates renal interstitial fibrosis. J Am Soc Nephrol 2009:20:1907–1918.
Shafiei MS, Rockey DC . The role of integrin-linked kinase in liver wound healing. J Biol Chem 2006:281:24863–24872.
Mills J, Digicaylioglu M, Legg AT et al. Role of integrin-linked kinase in nerve growth factor-stimulated neurite outgrowth. J Neurosci 2003:23:1638–1648.
Bock-Marquette I, Saxena A, White MD et al. Thymosin β4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair. Nature 2004:432:466–472.
Zhang Z, Garronb TM, Jian LX et al. Recombinant human decorin inhibits TGF-beta1-induced contraction of collagen lattice by hypertrophic scar fibroblasts. Burns 2009:35:527–537.
Kuhn DM, Balkis M, Chandra J et al. Uses and limitations of the XTT assay in studies of Candida growth and metabolism. J Clin Microbiol 2003:41:506–508.
Berry DP, Harding KG, Stanton MR et al. Human Wound Contraction: Collagen Organization, Fibroblasts, and Myofibroblasts. Plastic Reconstr Surg 1998:102:124–131.
Hynes RO . Integrins: versatility, modulation, and signaling in cell adhesion. Cell 1992:69:11–25.
Moulin V, Tam BY, Castilloux G et al. Fetal and adult human skin fibroblasts display intrinsic differences in contractile capacity. J Cell Physiol 2001:188:211–222.
Lorenz K, Grashoff C, Torka R et al. Integrin-linked kinase is required for epidermal and hair follicle morphogenesis. J Cell Biol 2007:177:501–513.
Kogata N, Tribe RM, Fässler R et al. Integrin-linked kinase controls vascular wall formation by negatively regulating Rho/ROCK-mediated vascular smooth muscle cell contraction. Genes Dev 2009:23:2278–2283.
Levinson H, Turner C, Ehrlich H . Integrin-linked kinase: a possible role in scar contracture. Ann Plast Surg 2004:52:204–211.
Ehrlich HP, Rittenberg T . Differences in the mechanism for high- versus moderate-density fibroblast-populated collagen lattice contraction. J Cell Physiol 2000:185:432–439.
Schäffer MR, Tantry U, Ahrendt GM et al. Stimulation of fibroblast proliferation and matrix contraction by wound fluid. Int J Biochem Cell Biol 1997:29:231–239.
Wong RP, Ng P, Dedhar S et al. The role of integrin-linked kinase in melanoma cell migration, invasion, and tumor growth. Mol Cancer Ther 2007:6:1692–1700.
Tang J, Wang J, Kong X et al. Vascular endothelial growth factor promotes cardiac stem cell migration via the PI3K/Akt pathway. Exp Cell Res 2009:315:3521–3531.
Guo L, Yu W, Li X et al. Targeting of integrin-linked kinase with a small interfering RNA inhibits endothelial cell migration, proliferation and tube formation in vitro. Ophthalmic Res 2009:42:213–220.
Gagné D, Groulx JF, Benoit YD et al. Integrin-linked kinase regulates migration and proliferation of human intestinal cells under a fibronectin-dependent mechanism. J Cell Physiol 2010:222:387–400.
Qian Y, Zhong X, Flynn DC et al. ILK mediates actin filament rearrangements and cell migration and invasion through PI3K/Akt/Rac1 signaling. Oncogene 2005:24:3154–3165.
Zhao Y, Wang H, Li X, Cao M et al. Ang II-AT1R increases cell migration through PI3K/AKT and NF-kappaβ pathways in breast cancer. J Cell Physiol 2014:229:1855–1862.
Huang CY, Fong YC, Lee CY et al. CCL5 increases lung cancer migration via PI3K, Akt and NF-kappaβpathways. Biochem Pharmacol 2009:77:794–803.
Fong YC, Li TM, Wu CM et al. BMP-2 increases migration of human chondrosarcoma cells via PI3K/Akt pathway. J Cell Physiol 2008:217:846–855.
Martinez-Ferrer M, Afshar-Sherif AR, Uwamariya C et al. Dermal transforming growth factor-beta responsiveness mediates wound contraction and epithelial closure. Am J Pathol 2010:176:98–107.
Hu B, Wu Z, Phan SH . Smad3 mediates transforming growth factor-beta-induced alpha-smooth muscle actin expression. Am J Respir Cell Mol Biol 2003:29:397–404.
Hinz B . Formation and function of the myofibroblast during tissue repair. J Invest Dermatol 2007:127:526–537.
Kobayashi T, Liu X, Wen FQ et al. Smad3 mediates TGF-beta1-induced collagen gel contraction by human lung fibroblasts. Biochem Biophys Res Commun 2006:339:290–295.
Lee YI, Kwon YJ, Joo CK . Integrin-linked kinase function is required for transforming growth factor beta-mediated epithelial to mesenchymal transition. Biochem Biophys Res Commun 2004:316:997–1001.
Vi L, de Lasa C, DiGuglielmo GM et al. Integrin-linked kinase is required for TGF-β1 induction of dermal myofibroblast differentiation. J Invest Dermatol 2011:131:586–593.
Boo S, Dagnino L . Integrins as modulators of transforming growth factor beta signaling in dermal fibroblasts during skin regeneration after injury. Adv Wound Care (New Rochelle) 2013:2:238–246.
Boudeau J, Miranda-Saavedra D, Barton GJ et al. Emerging roles of pseudokinases. Trends Cell Biol 2006:16:443–452.
Taylor SS, Kornev AP . Yet another ‘active’ pseudokinase, Erb3. Proc Natl Acad Sci USA 2010:107:8047–8048.
Eke I, Leonhardt F, Storch K et al. The small molecule inhibitor QLT0267 Radiosensitizes squamous cell carcinoma cells of the head and neck. PLoS ONE 2009:4:e6434.
Muranyi AL, Dedhar S, Hogge DE . Combined inhibition of integrin linked kinase and FMS-like tyrosine kinase 3 is cytotoxic to acute myeloid leukemia progenitor cells. Exp Hematol 2009:37:450–460.
Acknowledgements
This research was supported by the Key Science & Technology Brainstorm Project of Guangzhou, China (201300000178).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Integrin-linked kinase (ILK) is a key mediator in integrin signaling, which regulates the interactions between fibroblasts and extracellular matrix in wounds. This study found that ILK participates in wound and the fibroblast-populated collagen lattice contraction, probably due to the regulation of fibroblast migration and fibroblast-to-myofibroblast differentiation. Therefore, ILK may be a potential molecular target in regulating wound contraction and treating excessive scar contracture.
Rights and permissions
About this article
Cite this article
Li, G., Li, YY., Sun, JE. et al. ILK–PI3K/AKT pathway participates in cutaneous wound contraction by regulating fibroblast migration and differentiation to myofibroblast. Lab Invest 96, 741–751 (2016). https://doi.org/10.1038/labinvest.2016.48
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2016.48
This article is cited by
-
Ketotifen directly modifies the fibrotic response of human skin fibroblasts
Scientific Reports (2024)
-
Human Parathyroid Hormone Analog (3–34/29–34) promotes wound re-epithelialization through inducing keratinocyte migration and epithelial–mesenchymal transition via PTHR1-PI3K/AKT activation
Cell Communication and Signaling (2023)
-
Endothelial ILK induces cardioprotection by preventing coronary microvascular dysfunction and endothelial-to-mesenchymal transition
Basic Research in Cardiology (2023)
-
Staphylococcus aureus impairs cutaneous wound healing by activating the expression of a gap junction protein, connexin-43 in keratinocytes
Cellular and Molecular Life Sciences (2021)
-
Moxibustion Promotes Formation of Granulation in Wound Healing Process through Induction of Transforming Growth Factor-β in Rats
Chinese Journal of Integrative Medicine (2020)