Abstract
Microspherophakia seems to be the most specific feature of the Weill–Marchesani Syndrome, which could be due to mutations within the ADAMTS10 gene. As the locus responsible for isolated microspherophakia is still unknown, because the reported cases are rare, we checked whether the ADAMTS10 gene is involved in isolated microspherophakia in a Tunisian family. A consanguineous family (MSP-M), including six family members and two patients, presented with decreased vision secondary to bilateral isolated microspherophakia. A linkage analysis was carried out using microsatellite markers flanking the ADAMTS10 candidate gene. In the MSP-M family, isolated microspherophakia is likely inherited as an autosomal-recessive disease. Using a homozygosity-mapping strategy, haplotypic analysis using four STRs showed an exclusion of linkage between the ADAMTS10 gene and the disease locus in this family. Our study suggests that isolated microspherophakia and the Weill–Marchesani Syndrome are not allelic to the ADAMTS10 gene.
Similar content being viewed by others
Introduction
Microspherophakia (MSP) is a rare disease, characterized by a bilateral microphakia and spherophakia, smaller and more spherical lenses than normal, an increased anteroposterior thickness and highly myopic eyes.1 Lens dislocation or subluxation may occur, leading to defective accommodation.1
From 1901 to 2009, 30 sporadic cases of microspherophakia were reported.2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 It may occur in association with ectopia lentis and glaucoma as an isolated familial anomaly with either an autosomal-dominant14 or an autosomal-recessive pattern of inheritance.6 None of these reports made a mention of associated extraocular abnormalities.
Microspherophakia is often described in association with systemic diseases, mainly in Weill-Marchesani (MIM 277600, MIM 134797) and Marfan (MIM 154700) syndromes.
It seems to be the most specific feature of the Weill–Marchesani syndrome (WMS), which is characterized by short stature, brachydactyly, joint stiffness, microspherophakia and ectopia of lenses.1 WMS results from mutations within ADAMTS10 (a disintegrin and metalloprotease with thrombospondin motifs) in an autosomal-recessive form (MIM 277600) and fibrillin-1 (FBN1) in autosomal-dominant WMS (MIM 134797). Both these genes are involved in connective tissue organization,15, 16 and the metalloprotease protein has a major role in growth as well as in skin, lens and heart development.16
The syndromic forms likely suggest that the cause of microspherophakia is an anomaly of the mesodermal portions of the ciliary body and zonules.14 In a histopathological study of human microspherophakia in WMS, zonular fibers were rudimentary in character, abnormally long and somewhat degenerated.17 However, molecular data confirmed the physiopathology of MSP.
As the locus responsible for isolated MSP is still unknown, because the reported cases are rare, we checked whether the ADAMTS10 gene is involved in isolated microspherophakia. In this study, we report the clinical and genetic investigation of isolated microspherophakia in a consanguineous Tunisian family.
Materials and methods
Patients
The study included eight members of a consanguineous family (MSP-M) from Central Tunisia (two affected siblings and six unaffected members). The patients are born to a first-cousin marriage between healthy parents. All the members were examined at the Fattouma Bourguiba Hospital.
The diagnosis of isolated MSP was made on the basis of an ophthalmological examination including visual acuity and pupillary measurements, a slit-lamp examination of anterior chambers, ocular pressure measurement by Goldmann applanation tonometry and vitreous examination and a careful general physical examination.
Genetic investigation
In accordance with the Declaration of Helsinki, informed consent was obtained from all family members. Blood samples were obtained for DNA extraction from peripheral blood leukocytes by the salting out method. The PCR amplification reaction was carried out in a volume of 25 μl containing 10 ng of DNA, 10 × PCR buffer, 0.2 mM of each dNTP, 1.5 mM MgCl2, 0.5 μM of one of each primer and 0.5 U Amplitaq DNA polymerase (Invitrogen, Carlsbad, CA, USA). DNA was denatured for 5 min at 96 °C, followed by 40 cycles with denaturation at 94 °C for 30 s, annealing at the optimal annealing temperature depending on the primer pair used for 35 s, elongation at 72 °C for 40 s and terminal elongation for 10 min at 72 °C.
To test linkage to the 19p13.3–p13.2 region, patients and family members were subtyped with four microsatellite markers, D19S216 (AFM164ZB8), D19S884 (AFMa299zc5), D19S916 (AFMb325wa5) and D19S221 (AFM224YE9), flanking the candidate gene, ADAMTS10. Genotyping was performed as described elsewhere.18
Statistical methods
A two-point lod score was calculated using Linkage v5.1 program.19 An autosomal-recessive mode of inheritance, a complete penetrance and a disease gene frequency of 0.0001 were specified for all analyses. Allele frequencies were set equal for all markers. The recombination frequency was considered to be equal for both sexes.
Results
Clinical data
Case 1. (Proband): A 26-year-old woman presented with a history of gradual painless visual deterioration in both eyes. She did not have a cardiovascular, musculoskeletal or metabolic disease and has been healthy since birth. There was no family history of Marfan's syndrome, WMS or metabolic disease. The patient had high myopia (−15.00 in the right eye and −14.25 in the left eye). The best-corrected visual acuities were 20/100 and 20/200, respectively.
Intraocular pressure was normal in both eyes. The corneas of both eyes were clear. The anterior chambers were shallow with a central iridocorneal touch in the left eye (Figure 1). The crystalline lenses were clear and there was bilateral MSP with mild nasal and anterior displacement. The zonules were intact. The remaining anterior segment structures were normal (Figure 2). The patient underwent uneventful lens removal and in the sulcus intraocular lens implantation in both eyes. Visual acuity improved postoperatively to 20/25 in the right eye and 20/30 in the left eye. Ocular examination remained unchanged 24 months later.
Case 2: A 17-year-old boy, the proband's brother, was examined systematically. His medical history was unremarkable. The patient had high myopia (−12.00 in the right eye and −13.50 in the left eye). The best-corrected visual acuities were 20/60 and 20/100, respectively.
Intraocular pressure was normal in both eyes. The corneas of both eyes were clear. The anterior chambers were shallow with a central iridocorneal touch in both eyes. The crystalline lenses were clear and there was bilateral MSP with no significant lateral displacement. The zonules were intact. The remaining anterior segment structures were normal. The patient underwent uneventful lens removal and in the sulcus intraocular lens implantation in both eyes. Visual acuity improved postoperatively to 20/25 in both eyes. Ocular examination remained unchanged 18 months later.
Exclusion of linkage to the ADAMTS10 gene
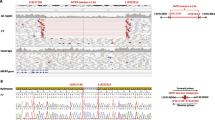
Genotypic analysis showed that the affected siblings, IV.3 and IV.5, were heterozygous for all tested markers overlapping the ADAMTS10 gene (Figure 3). This is not consistent with the fact that in autosomal-recessive diseases, affected children from consanguineous families are expected to be homozygous for the defective allele inherited from a common ancestor and for the genetic interval encompassing the causative gene. Furthermore, patients IV-3 and IV-5 did not share a common disease haplotype. They also had the same haplotype as their unaffected siblings IV-1 and IV-6, respectively. These genotyping results suggest that the family MSP-M is not linked to the tested candidate locus.
Two-point linkage analysis confirms exclusion of the region around markers D19S216 (LOD= −2.00 at the recombination fraction θ=0.024), D19S884 (LOD= −2.00 at θ=0.059), D19S916 (LOD= −2.01 at θ=0.052) and D19S221 (LOD= −2.00 at θ=0.054).
Consequently, the ADAMTS10 gene is not responsible for the MSP in the MSP-M family.
Taking into account recent reports on the involvement of fibrillin type-1 gene FBN1 in autosomal-dominant WMS and in dominant and recessive Marfan20 (MIM 154700), and on protein interaction between ADAMTS10 and FBN1,21 and on the linkage of FBN1 in isolated skeletal features of Marfan,22 linkage analysis using microsatellite markers flanking this gene was performed. Although the affected sibs shared the same haplotype, no homozygosity by descent was observed. This is not in favor of linkage to this gene under the hypothesis of an autosomal-recessive inheritance.
Discussion
Although MSP may occur frequently as a part of several syndromes, the isolated microspherophakia is rare and its disease-causing mechanism remains unknown. Since 1901, isolated MSP disease was described in different ethnic groups, mostly in the Asian continent.3, 9, 10, 11, 12, 13, 14
We report here a clinical and genetic investigation of isolated MSP in a consanguineous Tunisian family in which the parents were unaffected and both sexes were affected in the siblings, suggesting an autosomal-recessive inheritance. This observation was in accordance with previous reports in German–Swiss and French MSP families.6, 23, 24
Ocular features of the two affected patients were similar to those seen in WMS. Moreover, the normality of the general physical examination and the absence of major lateral displacement argue against other causes of MSP or ectopia lentis, particularly Marfan's syndrome. The similarity of ocular involvement between our patients and patients with WMS led us to check whether the autosomal-recessive WMS gene (ADAMTS10) is also involved in isolated MSP in this Tunisian family. A haplotypic analysis using four microsatellite markers clearly showed an exclusion of linkage between the ADAMTS10 gene and the disease locus in this family. Therefore, WMS and MSP were not allelic disorders to the ADAMTS10 gene.
In conclusion, isolated MSP in this Tunisian family shares a similar clinical ocular appearance with MSP secondary to WMS. This first molecular study of isolated MSP suggests that these two disorders are not allelic to the ADAMTS10 gene. Nevertheless, the involvement of other WMS loci in MSP cannot be ruled out.
Conflict of interest
The authors declare no conflict of interest.
References
Duke-Elder, S. in System of Ophthalmology Vol. III. Normal and Abnormal Development. Part 2. Congenital Deformities 694–696 (The CV Mosby Co, St Louis, 1963).
Shapira, T. M. Microphakia and spherophakia with glaucoma. Am. J. Ophthal. 17, 726–735 (1934).
Nirankari, M. S. & Maudgal, M. C. Microphakia. Brit. J. Ophthal. 43, 314–316 (1959).
Bronner, A. & Benck, P. Microspherophakia with zonular agenesis. Bull. Soc. Ophtalmol. Fr. 65, 648–654 (1965).
Hudelo, J. Microspherophakia. Bull. Soc. Ophtalmol. Fr. 66, 49–57 (1966).
Saraux, H., Claux, J. M. & Gaboriaux, J. La microsphérophaquie_Problèmes génétiques et thérapeutiques. Bull. Mem. Soc. Fr. Ophtalmol. 81, 352–357 (1968).
Viallefont, M. M., Boudet, C., Costeau, J. & Raynaud, C. A propos d'un cas isolé de microsphérophakie. Bull. Soc. Ophtalmol. Fr. 68, 784–787 (1968).
Michaud, C. Microspherophakia. Can. J. Ophthalmol. 5, 252–254 (1970).
Chan, R. T. & Collin, H. B. Microspherophakia. Clin. Exp. Optom. 85, 294–299 (2002).
Fan, D. S., Young, A. L., Yu, C. B., Chiu, T. Y., Chan, N. R. & Lam, D. S. Isolated microspherophakia with optic disc colobomata. J. Cataract Refract. Surg. 29, 1448–1452 (2003).
Khokhar, S., Pangtey, M. S., Sony, P. & Panda, A. Phacoemulsification in a case of microspherophakia. J. Cat. Refract. Surg. 29, 845–847 (2003).
Pal, N., Sharma, Y. R., Azad, R. & Chandra, P. Isolated bilateral microspherophakia with giant retinal tear and rhegmatogenous retinal detachment. J. Pediatr. Ophthalmol. Strabismus. 42, 238–240 (2005).
Kaushik, S., Sachdev, N., Pandav, S. S., Gupta, A. & Ram, J. Bilateral acute angle closure glaucoma as a presentation of isolated microspherophakia in an adult: case report. BMC Ophthalmol. 6, 29 (2006).
Johnson, V. P., Grayson, M. & Christian, J. C. Dominant microspherophakia. Arch. Ophthalmol. 85, 534–542 (1971).
Sakai, L. Y., Keene, D. R., Glanville, R. W. & Bachinger, H. P. Purification and partial characterization of fibrillin, a cysteine-rich structural component of connective tissue microfibrils. J. Biol. Chem. 266, 14763–14770 (1991).
Dagoneau, N., Benoist-Lasselin, C., Huber, C., Faivre, L., Mégarbané, A., Alswaid, A. et al. ADAMTS10 mutations in autosomal recessive Weill-Marchesani syndrome. Am. J. Hum. Genet. 75, 801–806 (2004).
Nagata, M., Takagi, S., Yamasaki, A., Tsunematsu, S., Kumagami, T., Itamochi, C. et al. Histopathological study of microspherophakia in the Weill-Marchesani syndrome. Jpn. J. Ophthalmol. 39, 89–95 (1995).
Bouchlaka, C., Abdelhak, S., Amouri, A., Ben Abid, H., Hadiji, S., Frikha, M. et al. Fanconi anemia in Tunisia: high prevalence of group A and identification of new FANCA mutations. J. Hum. Genet. 48, 352–361 (2003).
Ott, J. Analysis of Human Genetic Linkage 2nd edn. (Johns Hopkins University Press, Baltimore, 1991).
de Vries, B. B. A., Pals, G., Odink, R. & Hamel, B. C. J. Homozygosity for a FBN1 missense mutation: clinical and molecular evidence for recessive Marfan syndrome. Eur. J. Hum. Genet. 15, 930–935 (2007).
Kutz, W. E., Wang, L. W., Keene, D. R., Sakai, L. Y. & Apte, S. S. ADAMTS10 interacts with fibrillin in Weill-Marchesani Syndrome. ASMB Meeting Abstracts/Matrix Biology 25, S1–S94 (2006).
Milewicz, D. M., Grossfield, J., Cao, S. N., Kielty, C., Covitz, W. & Jewett, T. A mutation in FBN1 disrupts profibrillin processing and results in isolated skeletal features of the Marfan Syndrome. J. Clin. Invest. 95, 2373–2378 (1995).
Franceschetti, A. Ueber Mikrophakie und deren Erbgang. Klin. Mbl. Augenheilk. 85, 285 (1930).
Brasseur, G., Gruber, D., Charlin, J. F., Hubault, D. & Langlois, J. Microspherophakia--therapeutic aspects apropos of a familial case. Bull. Soc. Fr. Ophtalmol. 85, 1221–1224 (1985).
Acknowledgements
We are grateful to the patients and their family members for their cooperation in this study. This work was supported by the Tunisian Ministry of Higher Education, Scientific and Technological Research and the Ministry of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ben Yahia, S., Ouechtati, F., Jelliti, B. et al. Clinical and genetic investigation of isolated microspherophakia in a consanguineous Tunisian family. J Hum Genet 54, 550–553 (2009). https://doi.org/10.1038/jhg.2009.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhg.2009.75