Abstract
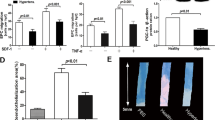
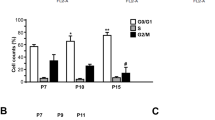
The functional impairment associated with atherogenic factors, including hypertension, constitutes a limitation to the ability of endothelial progenitor cells (EPCs) to repair. In addition, estrogens have been shown to play a role in reendothelialization after vascular injury. We investigated the effects of estrogens on differentiation and senescence of EPCs derived from bone marrow (BM-EPCs) in spontaneously hypertensive rats (SHR/Izm). Bone marrow (BM) cells were obtained from the tibias and femurs of age-matched, male SHR/Izm and Wistar-Kyoto rats (WKY/Izm). The number of differentiated, adherent BM-EPCs derived from SHR/Izm was significantly smaller than the number derived from WKY/Izm. 17β-Estradiol (E2) significantly increased the number of adherent BM-EPCs from SHR/Izm, and this effect was significantly attenuated by pharmacological phosphatidylinositol 3-kinase (PI3-K) blockers. Immunoblotting analysis revealed that E2 treatment led to phosphorylation of Akt. Senescence, as assessed by acidic β-galactosidase staining, occurred at a significantly greater rate in the BM-EPCs from SHR/Izm than in those from WKY/Izm, but E2 treatment dramatically delayed the senescence of BM-EPCs from SHR/Izm. A polymerase chain reaction (PCR)-ELISA based assay revealed that telomerase activity in BM-EPCs from SHR/Izm was significantly lower than in those from WKY/Izm, but that E2 treatment significantly augmented it. Both MTS and colony forming unit assay revealed that E2 treatment significantly augmented the functional activity in BM-endothelial cell (EC)-like cells from SHR/Izm compared to that in control BM-EC-like cells (no treatment). In conclusion, the differentiation of BM-EPCs derived from SHR/Izm was significantly decreased compared with that of BM-EPCs from WKY/Izm. In addition, the rate of senescence was significantly greater in the BM-EPCs from SHR/Izm than in those from WKY/Izm. Estrogen was shown to augment differentiation and delay the onset of senescence in BM-EPCs from SHR/Izm.
Similar content being viewed by others
Article PDF
References
Asahara T, Murohara T, Sullivan M, et al: Isolation of putative progenitor endothelial cells to angiogenesis. Science 1997; 275: 964–967.
Shi Q, Rafii S, Wu MH, Wijelath ES, et al: Evidence for circulating bone marrow-derived endothelial cells. Blood 1998; 92: 362–367.
Kong D, Melo LG, Mangi AA, et al: Enhanced inhibition of neointimal hyperplasia by genetically engineered endothelial progenitor cells. Circulation 2004; 109: 1769–1775.
Iwakura A, Luedemann C, Shastry S, et al: Estrogen-mediated, endothelial nitric oxide synthase-dependent mobilization of bone marrow-derived endothelial progenitor cells contributes to reendotheliazation after arterial injury. Circulation 2004; 108: 3115–3121.
Hill JM, Zalos G, Halcox JP, et al: Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med 2003; 348: 593–600.
Vasa M, Fichtlscherer S, Aicher A, et al: Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circ Res 2001; 89: e1–e7.
Hayflick L, Moorhead P : The serial cultivation of human diploid strains. Exp Cell Res 1961; 25: 585–621.
Murasawa S, Llevadot J, Silver M, Isner JM, Losordo DW, Asahara T : Constitutive human telomerase reverse transcriptase expression enhances regenerative properties of endothelial progenitor cells. Circulation 2002; 106: 1133–1139.
Iwakura A, Luedemann C, Shastry S, et al: Estrogen-mediated, endothelial nitric oxide synthase-dependent mobilization of bone marrow-derived endothelial progenitor cells contributes to reendotheliazation after arterial injury. Circulation 2003; 108: 3115–3121.
Strehlow K, Werner N, Berweiler J, et al: Estrogen increases bone marrow-derived endothelial progenitor cell production and diminishes neointima formation. Circulation 2003; 107: 3059–3065.
Bunag RD : Validation in awake rats of a tail-cuff method for measuring systolic pressure. J Appl Physiol 1973; 34: 279–282.
Imanishi T, McBride J, Ho Q, O'Brien KD, Schwartz SM, Han DK : Expression of cellular FLICE-inhibitory protein in human coronary arteries and in a rat vascular injury model. Am J Pathol 2000; 156: 125–137.
Dimri GP, Lee X, Basile G, et al: A biomarker that identifies senescent human cells in culture and in aging skin in vitro. Proc Natl Acad Sci U S A 1995; 92: 9363–9367.
Imanishi T, Hano T, Sawamura T, Nishio I : Oxidized low-density lipoprotein induces endothelial progenitor cell senescence, leading to cellular dysfunction. Clin Exp Pharmacol Physiol 2004; 31: 407–413.
Imanishi T, Hano T, and Nishio I : Angiotensin II accelerates endothelial progenitor cell senescence through induction of oxidative stress. J Hypertens 2005; 23: 97–104.
Dimmeler S, Aisher A, Vasa M, et al: HMG-CoA reductase inhibitors (statins) increase endothelial progenitor cells via the PI 3-kinase/Akt pathway. J Clin Invest 2001; 108: 391–397.
Kawamoto A, Gwon HC, Iwaguro H, et al: Therapeutic potential of ex vivo expanded endothelial progenitor cells for myocardial ischemia. Circulation 2001; 103: 634–637.
Assmus B, Schachinger V, Teupe C, et al: Transplatation of Progenitor Cell and Regeneration Enhancement in Acute Myocardial Infarction (TOPCARE-AMI). Circulation 2002; 106: 3009–3017.
Tepper OM, Galiano RD, Capla JM, et al: Human endothelial progenitor cells from type II diabetes exhibit impaired proliferation, adhesion, and incorporation into vascular structures. Circulation 2002; 106: 2781–2786.
Ikeda Y, Fukuda N, Wada M, et al: Development of angiogenic cell and gene therapy by transplantation of umbilical cord blood with vascular endothelial growth factor gene. Hypertens Res 2004; 27: 119–128.
Imanishi T, Hano T, Matsuo Y, Nishio I : Oxidized LDL inhibits vascular endothelial growth factor-induced endothelial progenitor cell differentiation. Clin Exp Pharmacol Physiol 2003; 30: 665–670.
Simoncini T, Rabkin E, Liao JK : Molecular basis of cell membrane estrogen receptor interaction with phosphatidylinositol 3-kinase in endothelial cells. Arterioscler Thromb Vasc Biol 2003; 23: 198–203.
Greider CW, Blackburn EH : Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 1985; 43: 405–413.
Greider CW, Blackburn EH : A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature 1989; 337: 331–337.
Crow JP, Beckman JS : The importance of superoxide in nitric oxide-dependent toxicity: evidence for peroxynitrite-mediated injury. Adv Exp Med Biol 1996; 387: 147–161.
Bodnar AG, Ouellette M, Frolkis M, et al: Extension of life-span by introduction of telomerase into normal human cells. Science 1998; 279: 349–352.
Broccoli D, Young JW, de Lange T : Telomerase activity in normal and malignant hematopoietic cells. Proc Natl Acad Sci U S A 1995; 92: 9082–9086.
Hiyama K, Hirai Y, Kyoizumi S, et al: Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J Immunol 1995; 155: 3711–3715.
Morrison SJ, Prowse KR, Ho P, Weissman IL : Telomerase activity in hematopoietic cells is associated with self-renewal potential. Immunity 1996; 5: 207–216.
Feng J, Funk WD, Wang SS, et al: The RNA component of human telomerase. Science 1995; 269: 1236–1241.
Nakamura TM, Morin GB, Chapman KB, et al: Telomerase catalytic subunit homologs from fission yeast and human. Science 1997; 277: 955–959.
Greider CW : Telomerase activity, cell proliferation, and cancer. Proc Natl Acad Sci U S A 1998; 95: 90–92.
Imanishi T, Hano T, Nishio I : Estrogen reduces endothelial progenitor cell senescenes through augmentation of telomerase activity. J Hypertens 2005; 23: 1699–1706.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Imanishi, T., Kobayashi, K., Hano, T. et al. Effect of Estrogen on Differentiation and Senescence in Endothelial Progenitor Cells Derived from Bone Marrow in Spontaneously Hypertensive Rats. Hypertens Res 28, 763–772 (2005). https://doi.org/10.1291/hypres.28.763
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.28.763
Keywords
This article is cited by
-
Fuchs endothelial corneal dystrophy and corneal endothelial diseases: East meets West
Eye (2020)
-
Endothelial progenitor cells as the target for cardiovascular disease prediction, personalized prevention, and treatments: progressing beyond the state-of-the-art
EPMA Journal (2020)
-
Effects of dihydrotestosterone on adhesion and proliferation via PI3-K/Akt signaling in endothelial progenitor cells
Endocrine (2014)
-
Endothelial progenitor cells in cardiovascular diseases: from biomarker to therapeutic agent
Regenerative Medicine Research (2013)
-
Estrogen and progesterone play pivotal roles in endothelial progenitor cell proliferation
Reproductive Biology and Endocrinology (2012)