Abstract
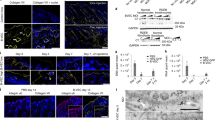
Mutations of the LAMB3 gene cause a lethal form of junctional epidermolysis bullosa (JEB). We hypothesized that early intra-amniotic gene transfer in a severe murine model of JEB would improve or correct the skin phenotype. Time-dated fetuses from heterozygous LAMB3IAP breeding pairs underwent ultrasound guided intra-amniotic injection of lentiviral vector encoding the murine LAMB3 gene at embryonic day 8 (E8). Gene expression was monitored by immunohistochemistry. The transgenic laminin-β3 chain was shown to assemble with its endogenous partner chains, resulting in detectable amounts of laminin-332 in the basement membrane zone of skin and mucosa. Ultrastructually, the restoration of ∼60% of hemidesmosomal structures was also noted. Although we could correct the skin phenotype in 11.9% of homozygous LAMB3IAP mice, none survived beyond 48 h. However, skin transplants from treated E18 homozygous LAMB3IAP fetuses maintained normal appearance for 6 months with persistence of normal assembly of laminin-332. These results demonstrate for the first time long-term phenotypic correction of the skin pathology in a severe model of JEB by in vivo prenatal gene transfer. Although survival remained limited due to the limitations of this mouse model, this study supports the potential for treatment of JEB by prenatal gene transfer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mavilio F, Pellegrini G, Ferrari S, Di Nunzio F, Di Iorio E, Recchia A et al. Correction of junctional epidermolysis bullosa by transplantation of genetically modified epidermal stem cells. Nat Med 2006; 12: 1397–1402.
Remington J, Wang X, Hou Y, Zhou H, Burnett J, Muirhead T et al. Injection of recombinant human type VII collagen corrects the disease phenotype in a murine model of dystrophic epidermolysis bullosa. Mol Ther 2009; 17: 26–33.
Wagner JE, Ishida-Yamamoto A, McGrath JA, Hordinsky M, Keene DR, Woodley DT et al. Bone marrow transplantation for recessive dystrophic epidermolysis bullosa. N Engl J Med 2010; 363: 629–639.
Wong T, Gammon L, Liu L, Mellerio JE, Dopping-Hepenstal PJ, Pacy J et al. Potential of fibroblast cell therapy for recessive dystrophic epidermolysis bullosa. J Invest Dermatol 2008; 128: 2179–2189.
Uitto J, McGrath JA, Rodeck U, Bruckner-Tuderman L, Robinson EC . Progress in epidermolysis bullosa research: toward treatment and cure. J Invest Dermatol 2010; 130: 1778–1784.
Chino T, Tamai K, Yamazaki T, Otsuru S, Kikuchi Y, Nimura K et al. Bone marrow cell transfer into fetal circulation can ameliorate genetic skin diseases by providing fibroblasts to the skin and inducing immune tolerance. Am J Pathol 2008; 173: 803–814.
Tolar J, Ishida-Yamamoto A, Riddle M, McElmurry RT, Osborn M, Xia L et al. Amelioration of epidermolysis bullosa by transfer of wild-type bone marrow cells. Blood 2009; 113: 1167–1174.
Muhle C, Jiang QJ, Charlesworth A, Bruckner-Tuderman L, Meneguzzi G, Schneider H . Novel and recurrent mutations in the laminin-5 genes causing lethal junctional epidermolysis bullosa: molecular basis and clinical course of Herlitz disease. Hum Genet 2005; 116: 33–42.
Laimer M, Lanschuetzer CM, Diem A, Bauer JW . Herlitz junctional epidermolysis bullosa. Dermatol Clin 2010; 28: 55–60.
Zanjani ED, Anderson WF . Prospects for in utero human gene therapy. Science 1999; 285: 2084–2088.
Schneider H, Coutelle C . In utero gene therapy: the case for. Nat Med 1999; 5: 256–257.
Douar AM, Adebakin S, Themis M, Pavirani A, Cook T, Coutelle C . Foetal gene delivery in mice by intra-amniotic administration of retroviral producer cells and adenovirus. Gene Therapy 1997; 4: 883–890.
Waddington SN, Buckley SM, Bernloehr C, Bossow S, Ungerechts G, Cook T et al. Reduced toxicity of F-deficient Sendai virus vector in the mouse fetus. Gene Therapy 2004; 11: 599–608.
Sato M, Tanigawa M, Kikuchi N . Nonviral gene transfer to surface skin of mid-gestational murine embryos by intraamniotic injection and subsequent electroporation. Mol Reprod Dev 2004; 69: 268–277.
Yoshizawa J, Li XK, Fujino M, Kimura H, Mizuno R, Hara A et al. Successful in utero gene transfer using a gene gun in midgestational mouse fetuses. J Pediatr Surg 2004; 39: 81–84.
Muhle C, Neuner A, Park J, Pacho F, Jiang Q, Waddington SN et al. Evaluation of prenatal intra-amniotic LAMB3 gene delivery in a mouse model of Herlitz disease. Gene Therapy 2006; 13: 1665–1676.
Endoh M, Koibuchi N, Sato M, Morishita R, Kanzaki T, Murata Y et al. Fetal gene transfer by intrauterine injection with microbubble-enhanced ultrasound. Mol Ther 2002; 5: 501–508.
Ito M, Liu Y, Yang Z, Nguyen J, Liang F, Morris RJ et al. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat Med 2005; 11: 1351–1354.
Levy V, Lindon C, Harfe BD, Morgan BA . Distinct stem cell populations regenerate the follicle and interfollicular epidermis. Dev Cell 2005; 9: 855–861.
Cotsarelis G, Sun TT, Lavker RM . Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 1990; 61: 1329–1337.
Fuchs E, Tumbar T, Guasch G . Socializing with the neighbors: stem cells and their niche. Cell 2004; 116: 769–778.
Lavker RM, Sun TT . Epidermal stem cells: properties, markers, and location. Proc Natl Acad Sci USA 2000; 97: 13473–13475.
Byrne C, Hardman M, Nield K . Covering the limb--formation of the integument. J Anat 2003; 202: 113–123.
Endo M, Zoltick PW, Peranteau WH, Radu A, Muvarak N, Ito M et al. Efficient in vivo targeting of epidermal stem cells by early gestational intraamniotic injection of lentiviral vector driven by the keratin 5 promoter. Mol Ther 2008; 16: 131–137.
Kuster JE, Guarnieri MH, Ault JG, Flaherty L, Swiatek PJ . IAP insertion in the murine LamB3 gene results in junctional epidermolysis bullosa. Mamm Genome 1997; 8: 673–681.
Endo M, Zoltick PW, Chung DC, Bennett J, Radu A, Muvarak N et al. Gene transfer to ocular stem cells by early gestational intraamniotic injection of lentiviral vector. Mol Ther 2007; 15: 579–587.
Henriques-Coelho T, Gonzaga S, Endo M, Zoltick PW, Davey M, Leite-Moreira AF et al. Targeted gene transfer to fetal rat lung interstitium by ultrasound-guided intrapulmonary injection. Mol Ther 2007; 15: 340–347.
Kim MG, Lee G, Lee SK, Lolkema M, Yim J, Hong SH et al. Epithelial cell-specific laminin 5 is required for survival of early thymocytes. J Immunol 2000; 165: 192–201.
Aumailley M, Smyth N . The role of laminins in basement membrane function. J Anat 1998; 193 (Pt 1): 1–21.
Stanley JR, Rubinstein N, Klaus-Kovtun V . Epidermolysis bullosa acquisita antigen is synthesized by both human keratinocytes and human dermal fibroblasts. J Invest Dermatol 1985; 85: 542–545.
Woodley DT, Krueger GG, Jorgensen CM, Fairley JA, Atha T, Huang Y et al. Normal and gene-corrected dystrophic epidermolysis bullosa fibroblasts alone can produce type VII collagen at the basement membrane zone. J Invest Dermatol 2003; 121: 1021–1028.
Amano S, Nishiyama T, Burgeson RE . A specific and sensitive ELISA for laminin 5. J Immunol Methods 1999; 224: 161–169.
Hata D, Miyazaki M, Seto S, Kadota E, Muso E, Takasu K et al. Nephrotic syndrome and aberrant expression of laminin isoforms in glomerular basement membranes for an infant with Herlitz junctional epidermolysis bullosa. Pediatrics 2005; 116: e601–e607.
Laimer M, Lanschützer CM, Nischler E, Klausegger A, Diem A, Pohla-Gubo G et al. [Hereditary blistering diseases. Symptoms, diagnosis and treatment of epidermolysis bullosa]. Hautarzt 2009; 60: 378–388.
Endo M, Henriques-Coelho T, Zoltick PW, Stitelman DH, Peranteau WH, Radu A et al. The developmental stage determines the distribution and duration of gene expression after early intra-amniotic gene transfer using lentiviral vectors. Gene Therapy 2010; 17: 61–71.
Sakai N, Waterman EA, Nguyen NT, Keene DR, Marinkovich MP . Observations of skin grafts derived from keratinocytes expressing selectively engineered mutant laminin-332 molecules. J Invest Dermatol 2010; 130: 2147–2150.
Stitelman DH, Endo M, Bora A, Muvarak N, Zoltick PW, Flake AW et al. Robust in vivo transduction of nervous system and neural stem cells by early gestational intra amniotic gene transfer using lentiviral vector. Mol Ther 2010; 18: 1615–1623.
Uitto J, Pfendner E, Jackson LG . Probing the fetal genome: progress in non-invasive prenatal diagnosis. Trends Mol Med 2003; 9: 339–343.
Weissman A, Itskovitz-Eldor J, Jakobi P . Sonographic measurement of amniotic fluid volume in the first trimester of pregnancy. J Ultrasound Med 1996; 15: 771–774.
Matsui C, Wang CK, Nelson CF, Bauer EA, Hoeffler WK . The assembly of laminin-5 subunits. J Biol Chem 1995; 270: 23496–23503.
Reynolds ES . The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 1963; 17: 208–212.
Qin XF, An DS, Chen IS, Baltimore D . Inhibiting HIV-1 infection in human T cells by lentiviral-mediated delivery of small interfering RNA against CCR5. Proc Natl Acad Sci USA 2003; 100: 183–188.
Kaufman CK, Sinha S, Bolotin D, Fan J, Fuchs E . Dissection of a complex enhancer element: maintenance of keratinocyte specificity but loss of differentiation specificity. Mol Cell Biol 2002; 22: 4293–4308.
Nakano A, Chao SC, Pulkkinen L, Murrell D, Bruckner-Tuderman L, Pfendner E et al. Laminin 5 mutations in junctional epidermolysis bullosa: molecular basis of Herlitz vs non-Herlitz phenotypes. Hum Genet 2002; 110: 41–51.
Billingham RE, Krohn PL, Medawar PB . Effect of cortisone on survival of skin homografts in rabbits. Br Med J 1951; 1: 1157–1163.
Acknowledgements
We thank Keith Alcorn (The Children's Hospital of Philadelphia) for animal support, Ray Meade (The Biomedical Imaging Core/Electron Microscopy Resource Laboratory of the University of Pennsylvania) for electron microscopy support and Xiaohua Chen (The Children's Hospital of Philadelphia) for qPCR support. These studies were supported by NIH/NIAMS Grant R01 AR54876 (JU) and by funds from the Department of Surgery and the Ruth and Tristram C Colket Chair in Pediatric Surgery, Children's Hospital of Philadelphia (AWF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Endo, M., Zoltick, P., Radu, A. et al. Early intra-amniotic gene transfer using lentiviral vector improves skin blistering phenotype in a murine model of Herlitz junctional epidermolysis bullosa. Gene Ther 19, 561–569 (2012). https://doi.org/10.1038/gt.2011.135
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.135
Keywords
This article is cited by
-
Therapeutic potential of adenovirus-encoding brain-derived neurotrophic factor for spina bifida aperta by intra-amniotic delivery in a rat model
Gene Therapy (2020)
-
Transamniotic mesenchymal stem cell therapy for neural tube defects preserves neural function through lesion-specific engraftment and regeneration
Cell Death & Disease (2020)
-
In Utero Gene Therapy and Genome Editing
Current Stem Cell Reports (2018)
-
A New Mouse Model of Junctional Epidermolysis Bullosa: The LAMB3 628G>A Knockin Mouse
Journal of Investigative Dermatology (2015)