Abstract
Aims
To determine the effects of argon green panretinal laser photocoagulation on retinal nerve fibre layer thickness, threshold visual fields, and Estermann full-binocular visual fields over time in diabetic retinopathy.
Method
Prospective, pilot clinical study. Time-domain optical coherence tomography (TD-OCT) of the optic nerve head and 24-2 SITA-Fast Humphrey/Estermann visual fields (HVF, EVFs) recorded at baseline, 10 weeks, and 6 months post laser. Quantitative field analysis of central 10°, 24°, and binocular visual fields.
Results
A total of 10 eye samples were subjected to uncomplicated multiple-session 100 ms panretinal laser using 2000 burns, 300-μm spot, and mean power of 136 mW (SD ±39.3). TD-OCT detected and quantified an increase in mean retinal nerve fibre layer thickness at 10 weeks (+8 μm; P<0.05) and progressive thinning at 6 months (−4 μm; P<0.05) compared with baseline. Mean threshold sensitivities, and 10° and 24° HVF improved at both time points in the majority (9 of 10 and 8 of 10) of patients. EVFs showed no significant change with treatment.
Conclusions
This pilot study shows that conventional argon laser panretinal photocoagulation may increase the retinal nerve fibre layer thickness in the short term, presumably related to laser-induced axonal injury, with progressive thinning of nerve fibre layer over the long term. The 10° and 24° visual fields improved significantly after laser with no adverse effects on the UK standard driving fields.
Similar content being viewed by others
Introduction
The use of retinal laser photocoagulation for advanced proliferative diabetic retinopathy (PDR) was reported by Meyer-Schwickerath.1 The laser light is absorbed primarily by melanosomes within the retinal pigment epithelium (RPE), leading to coagulation of adjacent photoreceptors and RPE cells by thermal mechanisms. Immediate consequences are reductions in oxygen consumption within the outer retina and formation of laser burns. The burns thin the outer retina and create window defects that allow oxygen diffusion from the choroid to the inner retina.2
Early xenon arc and krypton laser photocoagulation produced almost full-thickness burns with significant visual complications.3 The DRS (Diabetic Retinopathy Study) established that single-spot argon green laser panretinal photocoagulation (PRP) reduces PDR progression and severe visual loss.4
In the United Kingdom, the Driver and Vehicle Licensing Agency (DVLA) has designated both a minimum visual acuity and a visual field standard. For visual fields, there should be no ‘significant’ loss within the central 20° zone or within a 120° zone along the horizontal meridian using the Estermann visual field (EVF) test.5
PRP remains the gold standard treatment for PDR, and the Early Treatment Diabetic Retinopathy Study (ETDRS) recommends application of up to 2000 visible end-point burns on the retina.6 It is recognised that the laser scar expansion in the retina may be associated with photoreceptor loss, RPE hypertrophy, and visual field loss. The annual scar expansion rate (16.5%) continues to increase up to 4 years after PRP.7 After ETDRS protocol PRP, between 12 and 30% of patients may fail DVLA binocular visual field testing.8, 9 Vision testing and driving field assessment are requested locally by the DVLA to assess the patient's fitness to drive after self-declaration of bilateral laser photocoagulation, which may be repeated as required with further PRP.
High-resolution imaging of the peripapillary, retinal nerve fibre layer is possible using time-domain optical coherence tomography (TD-OCT) and scanning laser polarimetry (GDx VCC). Nerve fibre layer thickness may be quantified and correlated with visual field change in patients with glaucoma, and more recently, these tools investigated early retinopathy alterations in sub-clinical diabetic patients.10, 11
This paper describes a pilot study of argon green PRP in a cohort of diabetic patients using a current smaller spot laser strategy in PDR. The main aims of our study were to assess the effects of conventional laser on the retinal nerve fibre layer and to better understand the laser–neural tissue interaction, as well as to investigate effects of conventional burns on central and binocular driving fields over time.
Materials and methods
Subjects
We conducted a prospective, non-randomised pilot study in a diabetic eye clinic (Bradford Royal Infirmary, Bradford, UK) between March 2007 and February 2008. A consecutive series of 8 patients underwent unilateral (n=6) or bilateral (n=4) PRP for PDR. Patients gave informed consent according to the Declaration of Helsinki, and all investigations were performed as part of routine NHS care. We certify that all applicable institutional and governmental regulations regarding the ethical use of human volunteers were followed during this research. The study was approved by the local research ethics committee. Intraocular pressure and optic disc cupping were examined retrospectively in all eyes.
Argon green photocoagulation treatment
Multiple-session PRP used conventional argon green laser (514 nm) in two sessions, 2 weeks apart. We used the Transequator lens (Volk Optical Inc., Mentor, OH, USA) for all treatments, with a system spot size of 200 μm that gives an aerial laser spot size of 300 μm. Threshold laser treatment was designated by a grey–white, ETDRS burn. Primary treatment involved 2000 burns placed at one burn-width apart of 300 μm size and 100 ms pulse duration, 2 weeks apart. The area of laser coverage extended from two disc diameters outside the vascular arcades up to the ora serrata in all four quadrants, sparing one disc diameter zone in the peripapillary zone. Laser was not delivered to any patient on the same day as fluorescein angiography. At 3 months, the PDR activity was re-assessed, and additional PRP was delivered using similar parameters if required. Re-treatment of previous laser burns, and overlapping burns were avoided.
Time-domain optical coherence tomography
The Stratus OCT3 system (Carl Zeiss Meditec Inc., Dublin, CA, USA) was used to measure retinal nerve fibre layer thickness in the peripapillary zone. A circular scan was taken for each eye, and scanning was repeated until an optimal image capture that met the following criteria was achieved. The signal intensity should be ⩾7, the circular scan should be centrally aligned at the optic disc to measure the nerve fibre layer edges, with clear retina disc and vessel image clarity. The scan with highest signal intensity was analysed. All patients were imaged within 1 week of laser treatment, at 10 weeks, and at 6 months post laser.
Visual field assessment
The 24-2 SITA Fast and EVFs were recorded at baseline, 10 weeks, and 6 months post laser. A quantitative field analysis of central 10° and 24° was conducted. Full, binocular EVFs were examined by a member (FDG) of both the Visual Standards Sub-Committee of the Royal College of Ophthalmologists and the Vision Panel for the DVLA (UK). Estermann fields were analysed in a masked and non-identifiable manner, using the 2008 RCO definition for the minimum field for safe driving.5
Statistical analysis
We performed statistical analyses using Statistica (version 6, Reed Business Information Inc., New York, NY, USA). We used the two-tailed t-test to explore changes in nerve fibre layer thickness, central 24-2 field threshold values, and Estermann field scores after treatment at specified time points. The null hypothesis was rejected for P-values <0.05.
Results
A total of 10 eye samples obtained from 8 diabetic patients (mean age 48 years; range 33–62) were treated with PRP. There were no patients with glaucoma in this study. All patients completed follow-up and investigations that included six male and four female eye samples. Seven eye samples were of Asian origin and three were Caucasian. The mean baseline glycosylated haemoglobin level for this cohort was 9.7±2.6% (range 7.9–12), and there were no significant changes at the end of follow-up.
Five eye samples had neovascularisation at the disc greater than standard Airlie House photograph 10A, with 1 of 5 having an associated vitreous haemorrhage; 2 eye samples had neovascularisation elsewhere greater than half-disc diameter; and 3 eye samples had neovascularisation elsewhere less than half-disc diameter. The mean power used for primary PRP treatment was 137 mW (SD ±43). At the 10-week visit, two eye samples (20%) required additional fill-in PRP using an average of 1500 burns. Complete regression of PDR was achieved in 90% of eye samples at 6 months. One patient with ongoing poor glycaemic control developed cystoid macular oedema (CMO) 6 months after primary PRP, unrelated to any recent laser photocoagulation.
Pre-treatment visual acuity was 6/12 or better in 7 eyes, and >6/12 in 3 eyes. At 6 months after laser, vision was stable in 60, and 30% of eyes had improved vision (range 6/6 to 6/18). One eye lost two lines of Snellen vision secondary to CMO. Intraocular pressure remained within the normal range for all eyes throughout the study. We did not observe any significant changes in optic disc cupping during the study.
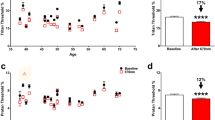
Serial nerve fibre layer thickness was analysed using Stratus OCT software (Carl Zeiss, Meditec Inc., Dublin, CA, USA). There were no significant changes in thickness measurements for superior or inferior quadrants at any time point. Ten weeks after laser, the mean 360° nerve fibre layer thickness (Figure 1) increased significantly by 8 μm (97.7±20.1 μm) compared with baseline (89.7±15.4 μm; P<0.05). The temporal quadrant (Figure 2) also demonstrated a significant thickening at 10 weeks compared with baseline (+11.2 μm, P<0.05).
At 6 months after PRP, there was progressive thinning of nerve fibre layer below the baseline levels (Figure 1). Mean 360° nerve fibre layer thickness reduced significantly by 3.99 μm (85.7±16.5 μm; P<0.05) compared with baseline thickness. In the nasal quadrant (Figure 3), there was a localised and significant reduction in nerve fibre layer thickness below baseline (58.8±17.3 μm, 6 months; 70.9±12.2 μm; P<0.05).
After treatment, improvement in mean 24-2 threshold values (excluding blind spot locations) of >1 dB was observed in 8 of 10 cases, worsening of >1 dB in 1 of 10 cases, and a stable value in 1 of 10 cases. Similar analysis of threshold values within the central 10° (outside the treatment area) found that 9 of 10 cases showed an improvement in >1 dB, whereas 1 of 10 cases had worsening of >1 dB. No detectable correlation was found between retinal nerve fibre layer changes and 24-2 SITA-Fast Humphrey (HVF) plots. Mean threshold levels for the inferior field alone increased with a trend for improvement after PRP.
Estermann field plots both pre and post laser showed little if any loss (Figure 4), the average number of seen stimuli being 117/120 (range 120/120 to 101/120). There was little change in the average number of seen stimuli being 117/120, 117/120, and 116/120 for the baseline, 10-week and 6-month measures, respectively. Masked grading of Estermann fields showed no failures of DVLA standards in any patients at any time point.
Discussion
Laser photocoagulation may cause a thermal rise and thermal spread in the outer retina. Laser induces intra-retinal inflammation, which can lead to macular oedema and visual loss.12, 13 A number of theories for PRP-induced inflammation exist. These include leukocyte–endothelial cell interactions that lead to inflammatory maculopathy after PRP in animal models, and correlation of laser-induced increased cytokine release with retinal capillary hyper-permeability.14
Laser-induced tissue reactions after PRP appear as cone-shaped lesions within the outer retina, and tend to spare inner retinal architecture. Over time, the outer retina appears to be the main site of laser burn formation.15 At longer pulse durations, laser burns may cause fibrotic reactions in animal models.16
Blankenship17 reported temporal thickening of the nerve fibre layer in the rabbit retina after experimental laser photocoagulation. With time, laser-induced damage to retinal ganglion cells results in the loss of nerve fibre layer and thinning within peripapillary nerve fibre layer zones.18 To test our hypothesis that PRP causes axonal damage and progressive loss over time, we examined visual fields in our patients to determine whether nerve fibre layer alterations are associated with any functional loss.
We used a multiple-session PRP protocol reflecting the current laser practice of UK retinologists. Some centres use 500 μm as a standard laser strategy rather than our current laser strategy of 300 μm; however, there are risks of 16.5% expansion rate with large laser spots and increased risks of people failing driving fields using a 500-μm spot size.7, 8, 9 Although laser powers appear lower than those of the published literature, standard pulse duration was used to ensure ETDRS burn intensity. No natural history data are available for RNFL changes in PDR. Furthermore, at the time of conception of this study in 2007, no published studies were available on nerve fibre layer change post PRP in diabetics. More recently, Maia et al19 described nerve fibre layer changes in very severe non-proliferative DR and PDR using 1600 burns titrated over three sessions. A significant increase in nerve fibre layer within the temporal quadrant was observed up to 6 months post PRP, without any reduction in the nerve fibre layer after 6 months. In a retrospective study, Kim and Cho20 compared peripapillary nerve fibre layer thickness between a control group, mild-to-moderate non-proliferative DR, and a group with severe non-proliferative DR using a mean of 1577 PRP burns. They reported a significant reduction (2.12 μm) in nerve fibre layer at 6 months post laser compared with baseline. No data were available on the nerve fibre layer between baseline and 6-month time points.
Similar to these papers, our study confirms that the nerve fibre layer thickens after treatment and that this thickening persisted at 10 weeks. It is noteworthy that the nerve fibre layer remains thickened beyond the 30 days reported by Blankenship.17 We hypothesise that thermal diffusion around the 100-ms burn may damage inner retinal axons. Such sub-lethal axonal damage may cause a disruption of the mid-flow axonal flow. It is recognised that axonal damage and interruption of mid-flow axonal flow causes axonal oedema. PRP may cause a large amount of axon damage, which can lead to global thickening of the nerve fibre layer as observed after PRP. Furthermore, concurrent poor glycaemic control may also contribute to the ongoing laser-induced inflammation at 10 weeks in our patients.
Progressive thinning of the nerve fibre layer after PRP has been demonstrated in histopathological studies.17, 18 After PRP, there is increased oxygenation of the inner retina from the choroid, increasing intra-retinal perfusion around the ganglion cell axons.21 We hypothesise that direct laser damage and axonal oedema may lead to axonal cell death. Unlike this, no significant reductions in nerve fibre layer thickness were observed at 6 months in non-proliferative DR without laser treatment.20
Diabetic patients may have sub-clinical, underlying visual field defects secondary to pre-existing severe non-proliferative DR or PDR.22 Laser treatment for PDR induces visual field defects as the ETDRS reported significant worsening and constriction of the visual field at 4 months after full, scatter PRP.6 Henricsson and Heijl23 found significant deteriorations in the visual field in 94% patients at 4 months. The extent of visual field loss has been attributed to higher laser power intensity and greater retinal laser coverage.8, 9
In this study, an increase in mean threshold sensitivity was observed in a majority of the patients, and such increases were also present for the untreated central 10°. Our laser technique was modelled on ETDRS guidelines but used lower power than did the EDTRS and Henricsson and Heijl studies, which may have contributed to significant functional benefits. Improved sensitivity, especially at the untreated central area may be the result of a reduction in oedema after PRP, although part of the improvement may be attributable to learning effects.
In the United Kingdom, the DVLA evaluates binocular visual fields to assess a patient's ‘fitness to drive.’5 In the 1990s, a number of studies investigated PRP and driving visual fields in the United Kingdom. Buckley et al24 retrospectively studied different types of laser PRP and DVLA field testing. Overall, 50% failed the DVLA standards using treatments ranging from 164 to 5917 photocoagulation burns. In particular, xenon arc posed a greater risk to visual field than did argon or diode laser, as xenon produced full-thickness retinal burns.
In 1992, Hulbert and Vernon25 reported results of PRP using different pulse durations on visual fields. They advocated routine use of a 200-μm spot for primary PRP in PDR to achieve regression of PDR and maintenance of driving visual fields. Similar recommendations were reported by Mackie et al,8 after 19% of patients studied were found to fail the DVLA standards. We used a 100-ms pulse duration, 300-μm spot, and low laser power. Using a 1 burn-width spot spacing and 2000 burns, a full PRP across a satisfactory area of retina can be applied according to the ETDRS guidelines, and still preserving binocular fields to driving standards.
The Pascal Photocoagulator (OptiMedica, Santa Clara, CA, USA) was introduced in 2005 for retinal photocoagulation.26 It semi-automates the procedure using a brief pulse duration combined with rapid raster scan application of multiple spots that allows shorter treatment delivery times and a shorter duration of 0.02–0.05 s. In terms of modern laser PRP practice, the use of 400–500-μm spot sizes are relevant to Pascal users, and would also be relevant to current laser strategies in PDR.
We suggest that for prediction of visual field damage after PRP, one should consider the impact of laser power and fluence rather than spot size alone.16 Fluence is calculated as Power × Time/Area. A strength of this study is our use of standardised parameters for laser PRP. Other studies published have used various protocols and significantly higher laser powers; however, each treated eye in our study received an equivalent intensity burn as per the ETDRS guidelines. Our reduced laser powers resulted in lower fluence being applied to the retina without compromises in clinical efficacy. There are no data regarding laser power or pulse duration parameters from the published studies mentioned.8, 9, 25 It could be argued that a gentle and slow burn at 100 ms would lead to greater thermal spread within the retina, and this may produce nerve fibre layer changes observed in this study. Recent work using the Pascal laser at 10–20 ms has shown more localised laser burns within the outer retina, hence potentially less risk of nerve fibre layer damage.15 Al-Hussainy et al27 reported reduced pain responses using single-spot 20 ms compared with single-spot 100 ms pulse duration laser PRP in diabetic retinopathy.
There is an increased risk of laser burn expansion over time with PRP fill-in treatments.7 After primary PRP, top-up PRP burns may contaminate previous burns and further damage nerve fibre layer and visual fields. In our patients, there was no significant change in visual field over time and this may reflect the PRP protocol. The optimal strategy for maintaining binocular visual fields may be to use a scatter PRP regimen with smaller and less intense laser burns.25 These laser parameters may prevent burns from enlarging over time and permit safe re-treatments.
The visual field changes demonstrated in our pilot study bring into focus the optimal timing of binocular visual field testing for the DVLA in the United Kingdom. At present, the DVLA requests patients to undergo Estermann field tests once a patient notifies the agency of bilateral PRP. Binocular DVLA visual field performed within 6 months of PRP may actually reflect pre-existing visual field abnormalities due to severe retinal ischaemia or PDR at presentation. Some of these changes may improve after PRP as observed in this cohort with increased central threshold measures. It is recognised that visual field testing soon after PRP may cause false-negative errors.
PRP is a successful treatment for PDR, which can cause increased retinal nerve fibre layer thickness at 10 weeks and a reduction at 6 months. With modern, careful application of PRP, central visual field sensitivity improvement can be observed. In addition, such treatment does not cause debarring defects for driving as per the current DVLA standards. Patients may require visual field screening in the community, perhaps annually, as there may be late-onset binocular visual field loss in the long term.

References
Meyer-Schwickerath G . Light-coagulation. Buech Augenartz 1959; 33: 1.
Stefansson E . Oxygen and diabetic eye disease. Graefes Arch Clin Exp Ophthalmol 1990; 228: 120–123.
Marshall J, Bird AC . A comparative histopathological study of argon and krypton laser irradiations of the human retina. Br J Ophthalmol 1979; 63: 657–668.
Diabetic Retinopathy Study Research Group. Photocoagulation treatment of proliferative diabetic retinopathy. The second report of diabetic retinopathy study findings. Ophthalmology 1978; 85: 82–106.
Drivers Medical Group. At a glance guide to the current medical standards of fitness to drive. DVLA: Swansea, UK, 2009.
Early photocoagulation for diabetic retinopathy. ETDRS Report 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1991; 98: 766–785.
Maeshima K, Utsugi-Sutah N, Otani T, Kishi S . Progressive enlargement of scattered photocoagulation scars in diabetic retinopathy. Retina 2004; 24: 507–511.
Mackie SW, Webb LA, Hutchinson BM, Hammer HM, Barrie T, Walsh G . How much blame can be placed on laser photocoagulation for failure to attain driving standards? Eye 1995; 9: 517–525.
Pearson AR, Tanner V, Keightley SJ, Casswell AG . What effect does laser photocoagulation have on driving visual fields in diabetics? Eye 1998; 12: 64–68.
Lan YW, Henson DB, Kwartz A . The correlation between optic nerve head topographic measurements, peripapillary nerve fibre layer thickness, and visual field indices in glaucoma. Br J Ophthalmol 2003; 87 (9): 1135–1141.
Medeiros FA, Moura FC, Vessani RM, Susanna Jr R . Axonal loss after traumatic optic neuropathy documented by optical coherence tomography. Am J Ophthalmol 2003; 135: 406–408.
McDonald HR, Schatz H . Visual loss following panretinal photocoagulation for proliferative diabetic retinopathy. Ophthalmology 1985; 92: 388–393.
McDonald HR, Schatz H . Macular edema following panretinal photocoagulation. Retina 1985; 5: 5–10.
Nonaka A, Kiryu J, Tsujikawa A, Yamashiro K, Nishijima K, Kamizuru H et al. Inflammatory response after scatter laser photocoagulation in nonphotocoagulated retina. Invest Ophthalmol Vis Sci 2002; 43: 1204–1209.
Muqit MMK, Gray JCB, Marcellino GR, Henson DB, Young LB, Charles SJ et al. Fundus autofluorescence and Fourier-domain optical coherence tomography imaging of 10 and 20 millisecond Pascal® Retinal Photocoagulation Treatment. Br J Ophthalmol 2008; 93 (4): 518–525.
Jain A, Blumenkranz MS, Paulus Y, Wiltberger MW, Andersen DE, Huie P et al. Effect of pulse duration on size and character of the lesion in retinal photocoagulation. Arch Ophthalmol 2008; 126: 78–85.
Blankenship GW . Red krypton and blue-green argon panretinal laser photocoagulation for proliferative diabetic retinopathy: a laboratory and clinical comparison. Trans Am Ophthalmol Soc 1986; 84: 967–1003.
Brooks DE, Komàromy AM, Källberg ME . Comparative retinal ganglion cell and optic nerve morphology. Vet Ophthalmol 1999; 2: 3–11.
Maia Jr OO, Vessani RM, Takahashi WY, Susanna Jr R . Examination of the retinal nerve fiber layer in diabetic retinopathy treated by argon laser panphotocoagulation. Arq Bras Oftalmol 2008; 71 (2): 187–190.
Kim HY, Cho HK . Peripapillary retinal nerve fiber layer thickness change after panretinal photocoagulation in patients with diabetic retinopathy. Korean J Ophthalmol 2009; 23 (1): 23–26.
Flower RW, Fryczkowski AW, McLeod DS . Variability in choriocapillaris blood flow distribution. Invest Ophthalmol Vis Sci 1995; 36 (7): 1247–1258.
Chee CK, Flanagan DW . Visual field loss with capillary non-perfusion in preproliferative and early proliferative diabetic retinopathy. Br J Ophthalmol 1993; 77: 726–730.
Henricsson M, Heijl A . The effect of panretinal laser photocoagulation on visual acuity, visual fields and on subjective visual impairment in preproliferative and early proliferative diabetic retinopathy. Acta Ophthalmol (Copenh) 1994; 72: 570–575.
Buckley SA, Jenkins L, Benjamin L . Fields, DVLC and panretinal photocoagulation. Eye 1992; 6: 623–625.
Hulbert MFG, Vernon SA . Passing the DVLC field regulations following bilateral pan-retinal photocoagulation in diabetics. Eye 1992; 6: 456–460.
Blumenkranz MS, Yellachich D, Anderson DE, Wiltberger MW, Mordaunt D, Marcellino GR et al. New instrument: semiautomated patterned scanning laser for retinal photocoagulation. Retina 2006; 26: 370–375.
Al-Hussainy S, Dodson PM, Gibson JM . Pain response and follow-up of patients undergoing panretinal laser photocoagulation with reduced exposure times. Eye 2008; 22 (1): 96–99.
Acknowledgements
This research is supported by the Manchester Academic Health Sciences Centre (MAHSC) and the NIHR Manchester Biomedical Research Centre. This work has been presented at the European Association for Vision and Eye Research meeting, Slovenia, in September 2009.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Muqit, M., Wakely, L., Stanga, P. et al. Effects of conventional argon panretinal laser photocoagulation on retinal nerve fibre layer and driving visual fields in diabetic retinopathy. Eye 24, 1136–1142 (2010). https://doi.org/10.1038/eye.2009.308
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2009.308
Keywords
This article is cited by
-
The effects of visual-field loss from panretinal photocoagulation of proliferative diabetic retinopathy on performance in a driving simulator
Eye (2023)
-
Impact of photoreceptor density in a 3D simulation of panretinal laser photocoagulation
BMC Ophthalmology (2021)
-
Attaining functional levels of visual acuity after vitrectomy for retinal detachment secondary to proliferative diabetic retinopathy
Scientific Reports (2020)
-
The inner retinal structures of the eyes of children with a history of retinopathy of prematurity
Eye (2018)
-
The range of peripapillary retinal nerve fibre layer and optic disc parameters, in children aged up to but not including 18 years of age who were born prematurely: protocol for a systematic review
Systematic Reviews (2016)