Abstract
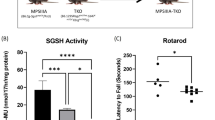
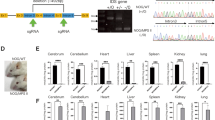
Mucopolysaccharidosis type I (MPS I) is an autosomal recessive inherited disease caused by deficiency of the glycosidase α-L-iduronidase (IDUA). Deficiency of IDUA leads to lysosomal accumulation of glycosaminoglycans (GAG) heparan and dermatan sulfate and associated multi-systemic disease, the most severe form of which is known as Hurler syndrome. Since 1981, the treatment of Hurler patients has often included allogeneic BMT from a matched donor. However, mouse models of the disease were not developed until 1997. To further characterize the MPS-I mouse model and to study the effectiveness of BMT in these animals, we engrafted a cohort (n=33) of 4–8-week-old Idua−/− animals with high levels (88.4±10.3%) of wild-type donor marrow. Engrafted animals displayed an increased lifespan, preserved cardiac function, partially restored IDUA activity in peripheral organs and decreased GAG accumulation in both peripheral organs and in the brain. However, levels of GAG and GM3 ganglioside in the brain remained elevated in comparison to unaffected animals. As these results are similar to those observed in Hurler patients following BMT, this murine-transplantation model can be used to evaluate the effects of novel, more effective methods of delivering IDUA to the brain as an adjunct to BMT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Neufeld EF, Muenzer J . The mucopolysaccharidoses. In: Scriver ALB CR, Sly WS, Beaudet AL, Valle D (eds). The Metabolic and Molecular Bases of Inherited Disease. McGraw Hill: New York, 2001, pp 3421–3452.
Scott HS, Litjens T, Nelson PV, Thompson PR, Brooks DA, Hopwood JJ et al. Identification of mutations in the alpha-L-iduronidase gene (IDUA) that cause Hurler and Scheie syndromes. Am J Hum Genet 1993; 53: 973–986.
Kornfeld S . Trafficking of lysosomal enzymes. Faseb J 1987; 1: 462–468.
Fratantoni JC, Hall CW, Neufeld EF . Hurler and Hunter syndromes: mutual correction of the defect in cultured fibroblasts. Science 1968; 162: 570–572.
Unger EG, Durrant J, Anson DS, Hopwood JJ . Recombinant alpha-L-iduronidase: characterization of the purified enzyme and correction of mucopolysaccharidosis type I fibroblasts. Biochem J 1994; 304 (Pt 1): 43–49.
Tsukimura T, Tajima Y, Kawashima I, Fukushige T, Kanzaki T, Kanekura T et al. Uptake of a recombinant human alpha-L-iduronidase (laronidase) by cultured fibroblasts and osteoblasts. Biol Pharm Bull 2008; 31: 1691–1695.
Rome LH, Weissmann B, Neufeld EF . Direct demonstration of binding of a lysosomal enzyme, alpha-L-iduronidase, to receptors on cultured fibroblasts. Proc Natl Acad Sci USA 1979; 76: 2331–2334.
Hobbs JR, Hugh-Jones K, Barrett AJ, Byrom N, Chambers D, Henry K et al. Reversal of clinical features of Hurler's disease and biochemical improvement after treatment by bone-marrow transplantation. Lancet 1981; 2: 709–712.
Boelens JJ, Rocha V, Aldenhoven M, Wynn R, O’Meara A, Michel G et al. Risk factor analysis of outcomes after unrelated cord blood transplantation in patients with hurler syndrome. Biol Blood Marrow Transplant 2009; 15: 618–625.
Staba SL, Escolar ML, Poe M, Kim Y, Martin PL, Szabolcs P et al. Cord-blood transplants from unrelated donors in patients with Hurler's syndrome. N Engl J Med 2004; 350: 1960–1969.
Bjoraker KJ, Delaney K, Peters C, Krivit W, Shapiro EG . Long-term outcomes of adaptive functions for children with mucopolysaccharidosis I (Hurler syndrome) treated with hematopoietic stem cell transplantation. J Dev Behav Pediatr 2006; 27: 290–296.
Dusing SC, Rosenberg A, Hiemenz JR, Piner S, Escolar M . Gross and fine motor skills of children with Hurler syndrome (MPS-IH) post umbilical cord blood transplantation: a case series report. Pediatr Phys Ther 2005; 17: 264–267.
Guffon N, Souillet G, Maire I, Straczek J, Guibaud P . Follow-up of nine patients with Hurler syndrome after bone marrow transplantation. J Pediatr 1998; 133: 119–125.
Whitley CB, Belani KG, Chang PN, Summers CG, Blazar BR, Tsai MY et al. Long-term outcome of Hurler syndrome following bone marrow transplantation. Am J Med Genet 1993; 46: 209–218.
Tolar J, Grewal SS, Bjoraker KJ, Whitley CB, Shapiro EG, Charnas L et al. Combination of enzyme replacement and hematopoietic stem cell transplantation as therapy for Hurler syndrome. Bone Marrow Transpl 2008; 41: 531–535.
Prasad VK, Kurtzberg J . Transplant outcomes in mucopolysaccharidoses. Semin Hematol 2010; 47: 59–69.
Vellodi A, Young E, Cooper A, Lidchi V, Winchester B, Wraith JE . Long-term follow-up following bone marrow transplantation for Hunter disease. J Inherit Metab Dis 1999; 22: 638–648.
Ziegler R, Shapiro E . Metabolic and neurodegenerative diseases across the life span. In: Donder J, Hunter SJ (eds) Principles and Practice of Lifespan Developmental Neuropsychology. Cambridge University Press: New York, 2007, pp 427–448.
Clarke LA, Russell CS, Pownall S, Warrington CL, Borowski A, Dimmick JE et al. Murine mucopolysaccharidosis type I: targeted disruption of the murine alpha-L-iduronidase gene. Hum Mol Genet 1997; 6: 503–511.
Ohmi K, Greenberg D, Rajavel K, Ryazantsev S, Li H, Neufeld E . Activated microglia in cortex of mouse models of mucopolysaccharidoses I and IIIB. PNAS 2003; 100: 1902–1907.
Garcia-Rivera MF, Colvin-Wanshura LE, Nelson MS, Nan Z, Khan SA, Rogers TB et al. Characterization of an immunodeficient mouse model of mucopolysaccharidosis type I suitable for preclinical testing of human stem cell and gene therapy. Brain Res Bull 2007; 74: 429–438.
Zheng Y, Rozengurt N, Ryazantsev S, Kohn DB, Satake N, Neufeld EF . Treatment of the mouse model of mucopolysaccharidosis I with retrovirally transduced bone marrow. Mol Genet Metab 2003; 79: 233–244.
Visigalli I, Delai S, Politi LS, Di Domenico C, Cerri F, Mrak E et al. Gene therapy augments the efficacy of hematopoietic cell transplantation and fully corrects mucopolysaccharidosis type I phenotype in the mouse model. Blood 2010; 116: 5130–5139.
Ohmi K, Greenberg DS, Rajavel KS, Ryazantsev S, Li HH, Neufeld EF . Activated microglia in cortex of mouse models of mucopolysaccharidoses I and IIIB. Proc Natl Acad Sci USA 2003; 100: 1902–1907.
Gori JL, Podetz-Pedersen K, Swanson D, Karlen AD, Gunther R, Somia NV et al. Protection of mice from methotrexate toxicity by ex vivo transduction using lentivirus vectors expressing drug-resistant dihydrofolate reductase. J Pharmacol Exp Ther 2007; 322: 989–997.
Wolf DA, Lenander AW, Nan Z, Belur LR, Whitley CB, Gupta P et al. Direct gene transfer to the CNS prevents emergence of neurologic disease in a murine model of mucopolysaccharidosis type I. Neurobiol Dis 2011; 43: 123–133.
Tolar J, Braunlin E, Riddle M, Peacock B, McElmurry RT, Orchard PJ et al. Gender-related dimorphism in aortic insufficiency in murine mucopolysaccharidosis type I. J Heart Valve Dis 2009; 18: 524–529.
Kuhn HG, Dickinson-Anson H, Gage FH . Neurogenesis in the dentate gyrus of the adult rat: age-related decrease of neuronal progenitor proliferation. J Neurosci 1996; 16: 2027–2033.
Braunlin E, Mackey-Bojack S, Panoskaltsis-Mortari A, Berry JM, McElmurry RT, Riddle M et al. Cardiac functional and histopathologic findings in humans and mice with mucopolysaccharidosis type I: implications for assessment of therapeutic interventions in hurler syndrome. Pediatr Res 2006; 59: 27–32.
Peters C, Balthazor M, Shapiro EG, King RJ, Kollman C, Hegland JD et al. Outcome of unrelated donor bone marrow transplantation in 40 children with Hurler syndrome. Blood 1996; 87: 4894–4902.
Souillet G, Guffon N, Maire I, Pujol M, Taylor P, Sevin F et al. Outcome of 27 patients with Hurler's syndrome transplanted from either related or unrelated haematopoietic stem cell sources. Bone Marrow Transpl 2003; 31: 1105–1117.
Breider MA, Shull RM, Constantopoulos G . Long-term effects of bone marrow transplantation in dogs with mucopolysaccharidosis I. Am J Pathol 1989; 134: 677–692.
Ellinwood NM, Colle MA, Weil MA, Casal ML, Vite CH, Wiemelt S et al. Bone marrow transplantation for feline mucopolysaccharidosis I. Mol Genet Metab 2007; 91: 239–250.
Shull RM, Breider MA, Constantopoulos GC . Long-term neurological effects of bone marrow transplantation in a canine lysosomal storage disease. Pediatr Res 1988; 24: 347–352.
Shull R, Lu X, Dube I, Lutzko C, Kruth S, Abrams-Ogg A et al. Humoral immune response limits gene therapy in canine MPS I. Blood 1996; 88: 377–379.
Krivit W, Sung JH, Shapiro EG, Lockman LA . Microglia: the effector cell for reconstitution of the central nervous system following bone marrow transplantation for lysosomal and peroxisomal storage diseases. Cell Transplant 1995; 4: 385–392.
Walkley SU . Secondary accumulation of gangliosides in lysosomal storage disorders. Semin Cell Dev Biol 2004; 15: 433–444.
Acknowledgements
We thank B Koniar for animal care, S Sandberg and R Cooksley for animal genotyping and J Gori for guidance in performing the transplant procedure. This work was supported by NIH grant P01 HD032652 (CBW, RSM), NIH grant R01NS048606 (PG and WCL), the US Department of Veterans Affairs (PG), the Minnesota Veterans Research Institute (PG) and training grant T32 DA022616 (DAW).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wolf, D., Lenander, A., Nan, Z. et al. Increased longevity and metabolic correction following syngeneic BMT in a murine model of mucopolysaccharidosis type I. Bone Marrow Transplant 47, 1235–1240 (2012). https://doi.org/10.1038/bmt.2011.239
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2011.239