Abstract
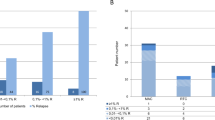
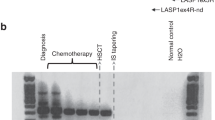
Allo-SCT is the only potentially curative regimen for myelodysplastic syndromes (MDSs), but it is associated with a high relapse risk. The role of chimerism analysis for prediction of relapse is yet to be determined. To assess the clinical usefulness of mixed chimerism (MC) for predicting hematological relapse, 75 consecutively transplanted patients with MDS were analyzed with regard to lineage-specific chimerism, encompassing CD33+ cells in peripheral blood (PB, n=49) and CD34+ cells in BM (n= 35). A cutoff of 5% recipient cells was used to discriminate complete donor chimerism from MC. A total of 19 patients (25%) experienced hematological relapse after a median of 5 (1–31) months. Sensitivity for detection of relapse was 59% for CD33+ PB cells and 92% for CD34+ BM cells with corresponding specificities of 91% and 65%. CD34+ BM cells were analyzed before relapse in seven patients, five of whom showed MC at a median of 2.5 (0.5–7) months before relapse. In contrast, 8 of 18 patients showed MC involving CD33 PB with a median of one month (0.5–2) before relapse. Here, we provide a model for early detection of relapse after SCT in MDS, in which serial characterization of both CD33+ PB cells and CD34+ BM cells gives an opportunity for early treatment before overt relapse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hofmann WK, Heil G, Zander C, Wiebe S, Ottmann OG, Bergmann L et al. Intensive chemotherapy with idarubicin, cytarabine, etoposide, and G-CSF priming in patients with advanced myelodysplastic syndrome and high-risk acute myeloid leukemia. Ann Hematol 2004; 83: 498–503.
Martino R, Iacobelli S, Brand R, Jansen T, van Biezen A, Finke J et al. Retrospective comparison of reduced-intensity conditioning and conventional high-dose conditioning for allogeneic hematopoietic stem cell transplantation using HLA-identical sibling donors in myelodysplastic syndromes. Blood 2006; 108: 836–846.
Sierra J, Perez WS, Rozman C, Carreras E, Klein JP, Rizzo JD et al. Bone marrow transplantation from HLA-identical siblings as treatment for myelodysplasia. Blood 2002; 100: 1997–2004.
Castro-Malaspina H, Harris RE, Gajewski J, Ramsay N, Collins R, Dharan B et al. Unrelated donor marrow transplantation for myelodysplastic syndromes: outcome analysis in 510 transplants facilitated by the National Marrow Donor Program. Blood 2002; 99: 1943–1951.
de Witte T, Hermans J, Vossen J, Bacigalupo A, Meloni G, Jacobsen N et al. Haematopoietic stem cell transplantation for patients with myelo-dysplastic syndromes and secondary acute myeloid leukaemias: a report on behalf of the Chronic Leukaemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT). Br J Haematol 2000; 110: 620–630.
Alessandrino EP, Della Porta MG, Bacigalupo A, Van Lint MT, Falda M, Onida F et al. WHO classification and WPSS predict posttransplantation outcome in patients with myelodysplastic syndrome: a study from the Gruppo Italiano Trapianto di Midollo Osseo (GITMO). Blood 2008; 112: 895–902.
Sutton L, Chastang C, Ribaud P, Jouet JP, Kuentz M, Attal M et al. Factors influencing outcome in de novo myelodysplastic syndromes treated by allogeneic bone marrow transplantation: a long-term study of 71 patients Societe Francaise de Greffe de Moelle. Blood 1996; 88: 358–365.
Nevill TJ, Fung HC, Shepherd JD, Horsman DE, Nantel SH, Klingemann HG et al. Cytogenetic abnormalities in primary myelodysplastic syndrome are highly predictive of outcome after allogeneic bone marrow transplantation. Blood 1998; 92: 1910–1917.
Deeg HJ, Shulman HM, Anderson JE, Bryant EM, Gooley TA, Slattery JT et al. Allogeneic and syngeneic marrow transplantation for myelodysplastic syndrome in patients 55 to 66 years of age. Blood 2000; 95: 1188–1194.
Appelbaum FR, Anderson J . Allogeneic bone marrow transplantation for myelodysplastic syndrome: outcomes analysis according to IPSS score. Leukemia 1998; 12 (Suppl 1): S25–S29.
Zipperer E, Pelz D, Nachtkamp K, Kuendgen A, Strupp C, Gattermann N et al. The hematopoietic stem cell transplantation comorbidity index is of prognostic relevance for patients with myelodysplastic syndrome. Haematologica 2009; 94: 729–732.
Armand P, Kim HT, Cutler CS, Ho VT, Koreth J, Alyea EP et al. Prognostic impact of elevated pretransplantation serum ferritin in patients undergoing myeloablative stem cell transplantation. Blood 2007; 109: 4586–4588.
Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 1997; 89: 2079–2088.
Malcovati L, Porta MG, Pascutto C, Invernizzi R, Boni M, Travaglino E et al. Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J Clin Oncol 2005; 23: 7594–7603.
NCCN. Practical guidelines in oncology. www.nccn.org 2009.
Roux E, Abdi K, Speiser D, Helg C, Chapuis B, Jeannet M et al. Characterization of mixed chimerism in patients with chronic myeloid leukemia transplanted with T-cell-depleted bone marrow: involvement of different hematologic lineages before and after relapse. Blood 1993; 81: 243–248.
Mattsson J, Uzunel M, Tammik L, Aschan J, Ringden O . Leukemia lineage-specific chimerism analysis is a sensitive predictor of relapse in patients with acute myeloid leukemia and myelodysplastic syndrome after allogeneic stem cell transplantation. Leukemia 2001; 15: 1976–1985.
Barrios M, Jimenez-Velasco A, Roman-Gomez J, Madrigal ME, Castillejo JA, Torres A et al. Chimerism status is a useful predictor of relapse after allogeneic stem cell transplantation for acute leukemia. Haematologica 2003; 88: 801–810.
Huisman C, de Weger RA, de Vries L, Tilanus MG, Verdonck LF . Chimerism analysis within 6 months of allogeneic stem cell transplantation predicts relapse in acute myeloid leukemia. Bone Marrow Transplant 2007; 39: 285–291.
Bornhaeuser M, Oelschlaegel U, Bug G, Platzbecker U, Lutterbeck K, Kiehl MG et al. Monitoring of donor chimerism in CD34+ peripheral blood progenitors allows to detect minimal residual disease after allogeneic stem cell transplantation -results of a randomized trial. ASH Ann Meet Abstr 2008; 112: 340.
Platzbecker U, Radke J, Kiani A, von Bonin M, Mohr B, Oelschlagel U et al. (eds). 5-Azacitidine treatment for imminent relapse in patients with high-risk myelodysplastic syndromes or acute myeloid leukaemia after allogeneic peripheral blood stem cell transplantation. 35th Annual Meeting of the European-Group-for-Blood-and-Marrow-Transplantation; 2009. Nature Publishing Group: Goteborg, SWEDEN.
Ringden O, Remberger M, Ruutu T, Nikoskelainen J, Volin L, Vindelov L et al. Increased risk of chronic graft-versus-host disease, obstructive bronchiolitis, and alopecia with busulfan versus total body irradiation: long-term results of a randomized trial in allogeneic marrow recipients with leukemia. Nordic Bone Marrow Transplantation Group. Blood 1999; 93: 2196–2201.
Uzunel M, Remberger M, Sairafi D, Hassan Z, Mattsson J, Omazic B et al. Unrelated versus related allogeneic stem cell transplantation after reduced intensity conditioning. Transplantation 2006; 82: 913–919.
Storb R, Deeg HJ, Whitehead J, Appelbaum F, Beatty P, Bensinger W et al. Methotrexate and cyclosporine compared with cyclosporine alone for prophylaxis of acute graft versus host disease after marrow transplantation for leukemia. N Engl J Med 1986; 314: 729–735.
Cutler C, Li S, Ho VT, Koreth J, Alyea E, Soiffer RJ et al. Extended follow-up of methotrexate-free immunosuppression using sirolimus and tacrolimus in related and unrelated donor peripheral blood stem cell transplantation. Blood 2007; 109: 3108–3114.
Remberger M, Svahn BM, Hentschke P, Lofgren C, Ringden O . Effect on cytokine release and graft-versus-host disease of different anti-T cell antibodies during conditioning for unrelated haematopoietic stem cell transplantation. Bone Marrow Transplant 1999; 24: 823–830.
Ringden O, Okas M, Uhlin M, Uzunel M, Remberger M, Mattsson J . Unrelated cord blood and mismatched unrelated volunteer donor transplants, two alternatives in patients who lack an HLA-identical donor. Bone Marrow Transplant 2008; 42: 643–648.
Bader P, Niethammer D, Willasch A, Kreyenberg H, Klingebiel T . How and when should we monitor chimerism after allogeneic stem cell transplantation? Bone Marrow Transplant 2005; 35: 107–119.
Westwood NB, Mufti GJ . Apoptosis in the myelodysplastic syndromes. Curr Hematol Rep 2003; 2: 186–192.
Kolb HJ . Donor leukocyte transfusions for treatment of leukemic relapse after bone marrow transplantation. EBMT Immunology and Chronic Leukemia Working Parties. Vox Sang 1998; 74 (Suppl 2): 321–329.
Campregher PV, Gooley T, Scott BL, Moravec C, Sandmaier B, Martin PJ et al. Results of donor lymphocyte infusions for relapsed myelodysplastic syndrome after hematopoietic cell transplantation. Bone Marrow Transplant 2007; 40: 965–971.
Czibere A, Bruns I, Kroger N, Platzbecker U, Lind J, Zohren F et al. 5-Azacytidine for the treatment of patients with acute myeloid leukemia or myelodysplastic syndrome who relapse after allo-SCT: a retrospective analysis. Bone Marrow Transplant 2010; 45: 872–876.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Tobiasson, M., Olsson, R., Hellström-Lindberg, E. et al. Early detection of relapse in patients with myelodysplastic syndrome after allo-SCT. Bone Marrow Transplant 46, 719–726 (2011). https://doi.org/10.1038/bmt.2010.179
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.179