Abstract
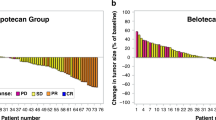
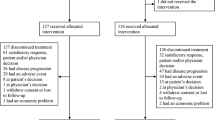
The semisynthetic taxoid docetaxel was investigated in a phase II study in non-chemotherapy pretreated patients with metastatic urothelial cell cancer. Thirty patients (median age 61, range 45-72) were treated with docetaxel 100 mg m(-2) administered as a 1-h infusion every 3 weeks. Of 29 evaluable patients, four achieved a complete response and five a partial response, for an overall response rate of 31%. The median duration of response was 6 months (range 4-51+). A total of 104 cycles were administered. The median number of cycles given was three (range 1-9). Toxic effects of docetaxel mainly consisted of neutropenia, which, however, rarely caused infectious complications (5%). Fluid retention or neuropathy necessitated treatment cessation in two patients. We conclude that docetaxel is an effective agent in urothelial cell cancer, and should be further tested in combination chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Wit, R., Kruit, W., Stoter, G. et al. Docetaxel (Taxotere): an active agent in metastatic urothelial cancer; results of a phase II study in non-chemotherapy-pretreated patients. Br J Cancer 78, 1342–1345 (1998). https://doi.org/10.1038/bjc.1998.681
Issue Date:
DOI: https://doi.org/10.1038/bjc.1998.681
This article is cited by
-
Pharmacoethnicity of docetaxel-induced severe neutropenia: integrated analysis of published phase II and III trials
International Journal of Clinical Oncology (2013)
-
The novel use of intravesical docetaxel for the treatment of non-muscle invasive bladder cancer refractory to BCG therapy: a single institution experience
World Journal of Urology (2009)