Abstract
Aim:
To determine whether adrenomedullin (ADM) attenuates vascular calcification (VC) by inducing osteopontin (OPN) expression.
Methods:
A VC model of rat aorta was induced with vitamin D3 plus nicotine (VDN), and vascular smooth muscle cell (VSMC) calcification was induced with beta-glycerophosphate. Von Kossa staining and alizarin red staining were assessed. Alkaline phosphatase (ALP) activity was measured. Immunohistochemical analysis was used to detect alpha-actin, while RT-PCR and Western blot analysis were used to quantify OPN expression.
Results:
Administration of ADM greatly reduced VC in VDN-treated aortas compared with controls, which was confirmed in calcified VSMCs. The decrease in alpha-actin expression was ameliorated by ADM both in vivo and in vitro. Moreover, mRNA and protein expression levels of OPN were significantly up-regulated in calcified aortas, and ADM increased OPN expression in calcified aortas. Furthermore, ADM up-regulated OPN expression in normal aortas and VSMCs. The ADM-mediated effects were similar to that of forskolin, which activates adenylyl cyclase; additionally, while the PKA inhibitor H89 and Ca2+ chelator Fura-2 blocked the effect of ADM.
However, the MEK/ERK inhibitor PD98509 had no effect on ADM induction of OPN mRNA expression. An OPN polyclonal antibody inhibited ADM-mediated attenuation of VC.
Conclusion:
ADM up-regulates OPN expression and thus attenuates VC via PKA. ADM appears to be an endogenous cardiovascular protective peptide and may represent a new therapeutic target for VC treatment.
Similar content being viewed by others
Introduction
Vascular calcification (VC) is a common occurrence in patients who have hypertension, atherosclerosis, diabetes, chronic renal failure, aging, aortic stenosis, and prosthetic valve replacement; VC is also currently believed to be an important risk factor in cardiovascular disease1, 2, 3, 4. Calcified vessels are resistant to vascular vasodilatation and more rigid than normal vessels, and promote thrombus formation and atherosclerotic plaque rupture. Previously, calcification was generally considered a result of passive calcium deposition in the extracellular matrix and cells5, 6. However, VC is currently believed to be an active, regulated process similar to osteogenesis4, 7, 8. During VC, vascular cells, including vascular smooth muscle cells (VSMCs), pericytes, and macrophages, revert to an osteoblast-like phenotype. This osteoblast-like phenotype is characterized by increased alkaline phosphatase (ALP) activity, matrix vesicle formation, and overexpression of bone morphogenetic proteins (BMPs), such as BMP-2, as well as bone matrix proteins, such as osteopontin (OPN), osteonectin, and osteocalcin4, 7, 8, 9, 10, 11. However, some BMPs, such as OPN and gamma-carboxyglutamic acid (Gla) protein (cMGP), are endogenous inhibitors of VC12, 13, 14, 15.
OPN is a phosphorylated glycoprotein generated or secreted by osteoblasts, osteoclasts, macrophages, T cells, hematopoietic cells, VSMCs, fibroblasts, and myocardial cells16. In calcified human aortic valves, OPN protein expression is upregulated and colocalizes with valvular calcific deposits17. OPN and cMGP were found to be expressed at calcification sites within atherosclerotic lesions and in microvessels with calciphylaxis18. Additionally, plasma OPN levels were significantly higher in patients with coronary calcification19. Previous in vitro and in vivo studies have demonstrated that OPN inhibits the formation and growth of hydroxyapatite and other biominerals and that phosphorylation of OPN is an important factor for regulating the OPN-mediated mineralization process20. OPN expression was also greatly upregulated in a cMGP knockdown-induced arterial calcification model, where OPN accumulated adjacent to the mineral and colocalized to surrounding cells in the calcified media. Interestingly, mice deficient in both cMGP and OPN exhibited a significant increase in arterial calcification compared to cMGP knockdown alone and died significantly earlier21. However, no spontaneous calcification was observed in OPN−/− VSMCs under normal culture conditions, while significantly higher calcification was observed in OPN−/− VSMCs cultured with a VC-inducing agent. Retroviral transduction of mouse OPN cDNA into OPN−/− VSMCs rescued the calcification phenotype of the cells22. These studies have shown that OPN plays a key role in inhibiting ectopic calcification. OPN may interact with transglutaminase 2 and lead to protein polymerization, which facilitates macrophage adhesion to the calcified elastic lamellae and promotes clearance of the ectopic mineral deposits23.
Recent studies have also demonstrated that paracrine/autocrine factors help maintain circulatory homeostasis and mediate the pathogenesis of cardiovascular disease. Among these factors, vasoactive peptides, such as adrenomedullin (ADM), intermedin, ghrelin, parathyroid hormone-related peptide, and C-type natriuretic peptide, are endogenous factors that inhibit VC15, 24, 25, 26, 27. However, endothelin and angiotensin II have opposite effects28, 29. Thus, examination of the endogenous peptides may help reveal novel mechanisms and therapeutic targets for VC.
ADM, which was initially isolated from human pheochromocytomas cells, can elevate platelet cyclic adenosine 3′,5′-monophosphate (cAMP) levels and induce a potent and long-lasting hypotensive effect30. Circulating ADM is mainly synthesized and secreted from vascular endothelial cells and VSMCs. Considerable evidence has shown that ADM has a wide range of autocrine, paracrine and endocrine mechanisms, including vasodilatory, anti-apoptotic, angiogenic, anti-fibrotic, natriuretic, diuretic, and positive inotropic mechanisms. ADM is therefore considered an important regulatory peptide that helps to regulate cardiovascular homeostasis. The ADM levels present in plasma and cardiovascular tissues are elevated to compensate for changes during cardiovascular diseases, such as atherosclerosis, hypertension, heart failure, acute myocardial infarction, and pulmonary hypertension31, 32. ADM function is mediated by the 7-transmembrane G-protein-coupled calcitonin receptor-like receptor (CRLR), which co-assembles with the subtypes 2 and 3 of a family of receptor activity modifying proteins (RAMPs), thus forming a receptor–co-receptor system. Binding of ADM to CRLR can activate several signaling pathways, including cAMP, calcium, cyclic guanosine 3′,5′-monophosphate, phosphatidylinositol 3-kinase/Akt, and mitogen-activated protein kinase (MEK)/extracellular signal-related kinase (ERK)31, 32. In VSMCs, the cAMP/protein kinase A (PKA) pathway regulates the vasodilatory effects of ADM. ADM causes an increase in the cAMP level of VSMCs by activating PKA, which ultimately decreases the calcium content of VSMCs33.
Our previous studies have shown that mRNA expression of ADM and its receptor gene are upregulated in calcified VSMCs in vitro, as well as in rat calcified vessels in vivo34, 35, 36. Administration of ADM significantly attenuated VC both in vivo and in vitro through the cAMP/PKA signaling pathway37, 38. However, the mechanism of how ADM attenuates VC is still unknown. We hypothesized that ADM upregulates OPN and thus ameliorates VC. In the present study, we investigated the ability of ADM to inhibit VC and the effect of ADM on phenotype transition in vitamin D3 plus nicotine (VDN)-induced calcified rat aortas and calcified VSMCs. We also investigated the effect of ADM on OPN mRNA and protein expression levels in normal and calcified aortas, as well as VSMCs. Moreover, we studied the effects of an OPN polyclonal antibody and assessed its ability to inhibit the ADM-mediated attenuation of VC.
Materials and methods
Materials
All animal care and experimental protocols were in compliance with the Regulation of Animal Management in China (documentation Number 55, 2001, the Ministry of Health of China) and were approved by the Animal Care Committee of the Peking University Health Science Center. Male Sprague–Dawley rats (150–180 g) were supplied by the Animal Center of Peking University Health Science Center. Synthesized human ADM1-52 was provided by Phoenix Pharmaceuticals (Belmont, CA, USA). The ERK inhibitor PD98059, PKA inhibitor H89, Ca2+ chelator Fura-2, beta-glycerophosphate, nicotine, alizarin red, and anti-alpha-SM actin antibody were from Sigma (St. Louis, MO, USA). Anti-OPN and beta-actin antibodies as well as all of the secondary antibodies, were from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The nitrocellulose membrane was from Hybond-C (Amersham Life Science, England), and Trizol reagent and the enhanced chemiluminescence kit were from Beijing Applygen Technologies (Beijing, China). The dNTPs, M-MuLV reverse transcriptase, Taq DNA polymerase, RNasin, and Oligo(dT)15 primer were from Promega (Madison, WI, USA). The ALP assay kits were from Nanjing Jiancheng Biological Co (Nanjing, China). All of the chemicals and reagents were of analytical grade.
The PCR primers were synthesized by SBS Co (Beijing, China). The forward and reverse PCR primers (rat) were OPN, 5′-AGA CCA GCC ATG AGT CAA GTC A-3′ and 5′-TGA AAC TCG TGG CTC TGA TGT T-3′; and beta-actin, 5′-GAG ACC TTC AAC ACC CCA GCC-3′ and 5′-TCG GGG CAT CGG AAC CGC TCA-3′, respectively.
Rat model of VC
The rat VC model was established by our laboratory as previously described25, 26. Briefly, 19 rats were used for treatment groups of VC, VC+ADM, or a vehicle control. Seventeen random rats were given vitamin D3 (300,000 IU/kg in arachis oil, intramuscularly) plus nicotine (25 mg/kg in 5 mL peanut oil, intragastrically) at 09:00 on day 1, and nicotine was re-administered at 19:00 on the same day. Seven randomly chosen rats were treated with ADM (100 ng·kg−1·h−1, 4 weeks) 24 h later, and the remaining VC rats served as the calcification controls. An additional 2 rats were given corresponding solvents as a vehicle control. ADM was administered subcutaneously in pure water through an Alzet Mini-osmotic Pump (Alzetw model 2004, DURECT Corp., Cupertino, CA, USA). After four weeks, blood sample was collected in heparinized syringes from the abdominal aorta and transferred to tubes to measure ALP activity. All of the animals were killed by exsanguination, and the aortas were quickly removed.
VSMC calcification model
To culture the VSMCs, we used the explant culture method with a few modifications, which have been previously described39, 40. Briefly, the rat thoracic aortas were cut into small pieces (about 2 mm3 each) after partial removal of the external connective tissue, placed in DMEM medium supplemented with 4 g/L glucose, 10 mmol/L sodium pyruvate, and 20% fetal bovine serum (FBS), and incubated at 37 oC in an incubator containing 95% air and 5% CO2 for 10 days. VSMCs that migrated from the explants were maintained in growth medium DMEM containing 10 mmol/L sodium pyruvate and 15% FBS. Staining for the presence of alpha-actin was used to confirm that there was a positive response. VSMCs at passages 5 to 8 were used for all of the experiments. Confluent cells were inoculated on 24-well plates (1×104 cells/mL) in DMEM that contained 10 mmol/L sodium pyruvate supplemented with 15% FBS without (growth medium) or with 10 mmol/L beta-glycerophosphate (calcification medium). At concentrations ranging from 10−9–10−7 mol/L, ADM was added to the medium after treatment with beta-glycerophosphate for 4 days. The medium was replaced with fresh medium every 3 days. After 14 days the medium was removed, and the cell layer was washed with cold phosphate-buffered saline. The cells were collected and stored at −70 oC until further use. The cells were divided into groups and treated as follows: 1) the control group, which was cultured in culture medium; 2) the calcification group, which was cultured in calcification medium; 3) the ADM treatment group, which was cultured in calcification medium with the respective ADM peptides (this group was divided into 2 subgroups based on the peptide concentration used: 10−9 or 10 –7 mol/L); and 4) the ADM agent treatment group, where 20 μmol/L Forskolin, 100 nmol/L H89, 30 μmol/L PD98059 or 2 μmol/L Fura-2 was added to the medium of the calcification group 40 min before supplementation with 10−7 mol/L ADM. After incubation for 10 days, the experiment was terminated and the cells were analyzed.
Detection of VC
Von Kossa staining for calcification25 and alizarin red staining for calcium phosphate salt deposition41 were performed as previously described with minor modifications. The ALP activity in plasma and aortic tissue was measured with an ALP assay kit (Jiancheng Bioengineering Co, Nanjing, China) and the results were normalized to the total protein concentration25, 26.
Immunohistochemical staining of alpha-actin
The VSMCs were fixed, permeabilized, blocked as previously described15, and subsequently stained with an anti-alpha-actin antibody (1:400 mouse anti-rat alpha-actin, Sigma).
Semi-quantitative RT-PCR analysis of OPN mRNA expression
RNA was extracted from the aortas and VSMCs with a Trizol reagent and quantified with an ultraviolet spectrophotometer (Biophotometer, Eppendorf, Germany). RNA (2 μg) was used to reverse-transcribe cDNA with the M-MuLV reverse transcriptase and Oligo(dT)15 primer. The total PCR reaction volume was 25 μL. For OPN expression analysis, the PCR program consisted of denaturation at 95 oC for 5 min, then PCR at 94 oC for 30 s; 60 oC for 30 s; and 72 oC for 60 s for 30 cycles with an extension at 72 oC for 5 min. Beta-actin primers (1 μL of a 200 nmol/L stock) instead of OPN primers were added to 2 μL of the PCR product to analyze beta-actin expression. The beta-actin expression PCR program consisted of denaturation at 95 oC for 5 min, then PCR at 94 oC for 30 s; 55 oC for 30 s; and 72 oC for 40 s for 30 cycles with an extension at 72 oC for 5 min. The DNA PCR product (5 μL) was analyzed by electrophoresis on a 1.5% agarose gel with ethidium bromide staining. The ratio of the optical density of the 2 bands (871 bp for OPN and 291 bp for beta-actin) was measured by a Gel Documentation System (Bio-Rad, Hercules, CA); the data are expressed as the ratio of OPN mRNA to beta-actin mRNA.
Western blot analysis of OPN protein expression
Protein extracts from the aortas and VSMCs were resuspended in sample buffer that contained 2% SDS, 2% mercaptoethanol, 50 mmol/L Tris–HCl (pH 6.8), 10% glycerol and 0.05% bromophenol blue. The protein mixture was then placed in boiling water for 10 min and briefly centrifuged at a low speed. The denatured protein samples were resolved on a 12% Tris/glycine SDS-polyacrylamide gel in running buffer containing 25 mmol/L Tris, 192 mmol/L glycine, and 0.1% SDS. The proteins were then transferred to a nitrocellulose membrane for 3 h at 4 oC at 200 mA in a transfer buffer containing 20 mmol/L Tris–HCl (pH 8.0), 150 mmol/L glycine, and 20% methanol. Non-specific protein binding was blocked by incubating the membrane with 5% non-fat dry milk in TBS-T [20 mmol/L Tris–HCl (pH 7.6), 150 mmol/L NaCl, and 0.02% Tween 20] for 1 h at room temperature with agitation. A goat anti-rat OPN primary antibody was added to the membrane at a 1:1000 dilution in TBS-T and incubated at 4 oC overnight with agitation. The secondary antibody was diluted in TBS-T (1:2000 dilution) and applied to the membrane, and the reaction was incubated at room temperature for 1 h with agitation. Between each of the three proceeding steps (primary antibody, secondary antibody, and visualization) the membrane was washed 3 times for 10 min each with TBS-T at room temperature. The membrane was immediately visualized on a phosphorimager after the addition of an enhanced chemiluminescence substrate.
Statistical analysis
The results were normalized to the total protein concentration and are presented as means±SD. The results for variables measured more than once in each animal (eg, calcified nodules) were averaged to produce a single value for each animal before further analysis. Student's t test was used to compare the results between 2 groups. A one-way ANOVA, followed by a Student–Newman–Keuls test, was used to compare the results between more than 2 groups. A P<0.05 was considered statistically significant, and P<0.01 was considered very statistically significant.
Results
ADM treatment attenuated VC
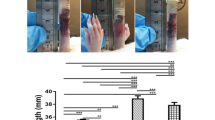
To investigate the effect of ADM on VC, rats with VC were treated with human ADM1–52. Von Kossa staining revealed the presence of dispersed, calcified nodules among the elastic fibers in calcified aortas but not in the control or ADM-treated aortas (Figure 1A). The activity of ALP, the functional phenotype marker of osteoblasts, was increased in the plasma (133±2.7 vs 1.3±0.8 U/L, P<0.01) and aortas (930±732 vs 212±45 U/g protein, P<0.05) of VC rats compared with the controls, and ADM treatment decreased the activity in both the plasma (61±1.9 vs 133±2.7 U/L, P<0.01, Figure 1B) and the aortas (448±265 vs 930±732 U/g protein, P<0.05, Figure 1C).
Adrenomedullin (ADM) inhibits vascular calcification induced by vitamin D3 plus nicotine (VDN) in rat aortas and in vascular smooth muscle cells (VSMCs). (A) von Kossa staining of aortas; (B) and (C) alkaline phosphatase (ALP) activity in the plasma and aortas, respectively; (D) alizarin-red staining of VSMCs. bP<0.05, cP<0.01 vs Control. eP<0.05, fP<0.01 vs Ca (Con, control; Ca, calcification).
In cultured VSMCs, alizarin-red staining showed that ADM treatment significantly decreased the calcium phosphate salt deposition that is associated with VC induction by beta-glycerophosphate (Figure 1D).
AMD prevented the loss of lineage markers in VSMCs
Because the phenotype transition of SMCs is associated with VC in vitro and in vivo, we investigated the expression of SM lineage markers. Compared with the controls, media from arteries with VC showed significantly decreased levels of SM alpha-actin with both immunostaining (0.04±0.01 vs 0.11±0.01, P<0.01) and Western blot analysis (1.03±0.09 vs 1.47±0.10, P<0.01); however, ADM administration prevented reduction of the SM alpha-actin levels (immunostaining, 0.10±0.02 vs 0.04±0.01, P<0.01; Western blot analysis, 1.21±0.06 vs 0.10±0.02, P<0.05; Figure 2A and B).
ADM inhibits the phenotype transition in calcified rat aortas and in VSMCs. (A) and (B) Immunochemistry and Western blot analysis, respectively, of α-actin protein expression in aortas. (C) Immunochemistry staining of α-actin in VSMCs. cP<0.01 vs Control. eP<0.05, fP<0.01 vs Ca (Con, control; Ca, calcification).
We further confirmed that ADM treatment prevents the loss of SM lineage markers in vitro by studying cultured VSMCs. The level of the SM lineage marker SM alpha-actin was strikingly decreased in calcified VSMC cultures by day 7, and ADM treatment prevented this decrease (Figure 2C).
ADM increased OPN mRNA and protein expression
We next investigated the mRNA and protein expression levels of OPN, which is an important endogenous inhibitor of VC. Semi-quantitative RT-PCR results showed that the OPN mRNA expression level was 48.8% (P<0.01) higher in VC aortas compared with the controls. Treatment with ADM (10−7 mol/L) for 8 h significantly increased the OPN mRNA expression level in the control and VC aortas by 37.4% and 46% (both P<0.01), respectively; additionally, the OPN mRNA level in ADM-treated VC aortas was 58.6% higher than that in the controls (P<0.01, Figure 3A). Similarly, the OPN protein concentration was 55.6% higher in VC aortas than in the controls (P<0.01). ADM treatment increased the OPN protein concentration in the control and calcified VSMCs by 44% and 80%, respectively; additionally, the OPN protein level was 94% higher in the VC aortas compared with the controls (all P<0.01, Figure 3B). We further assessed the in vitro effect of ADM on OPN expression in normal VSMCs. Semi-quantitative RT-PCR results showed that treatment with ADM at a dose of 10−9 or 10−7 mol/L for 8 h increased the OPN mRNA level by 50% and 122%, respectively, compared with the controls; the OPN mRNA level was 48.4% higher for the 10−7 mol/L of ADM compared to the 10−9 mol/L ADM (all P<0.01, Figure 3C). The OPN protein levels were increased by 54% and 185% with 10−9 and 10−7 mol/L ADM treatment, respectively, for 24 h, and the OPN protein level was 86% higher at the 10−7 mol/L dose compared to the 10−9 mol/L dose (all P<0.01, Figure 3D).
ADM inhibits aortic calcification by upregulating osteopontin (OPN). (A and B) RT-PCR and Western blot analysis of mRNA and protein expression of OPN, respectively, in aortas. (C) and D) mRNA and protein expression of OPN in VSMCs. cP<0.01 vs Con; fP<0.01 vs Ca; iP<0.01 vs ADM (Con, control; Ca, calcification).
Upregulation of OPN mRNA expression by ADM depends on cAMP/PKA and Ca2+ signaling
Because ADM can activate the cAMP/PKA signaling pathway in VSMCs and ameliorate VC, we investigated whether ADM upregulates OPN expression via activation of the cAMP/PKA signaling pathway. Semi-quantitative RT-PCR results showed that ADM and the adenylyl cyclase agonist forskolin (20 μmol/L) increase the OPN mRNA expression levels by 81% and 145%, respectively (both P<0.01), compared with the controls; upregulation with forskolin treatment was 36% higher than ADM-induced upregulation (P<0.01, Figure 4). Pretreatment with the PKA inhibitor H89 (100 nmol/L) prior to ADM treatment caused a 26% decrease in the OPN mRNA level compared to ADM treatment alone (P<0.05). Pretreatment with the intracellular Ca2+ chelator Fura-2 (2 μmol/L) prior to ADM treatment caused a 22% decrease in the OPN mRNA level compared to ADM treatment alone (P<0.05). However, the OPN mRNA expression level was not significantly altered when the cells were pretreated with the ERK inhibitor PD98059 (30 μmol/L) prior to ADM treatment.
ADM upregulates OPN mRNA expression through the cyclic adenosine 3′,5′-monophosphate (cAMP)/protein kinase A (PKA) signaling pathway. Forskolin, an adenylyl cyclase agonist; H89, a PKA inhibitor; Fura-2, a Ca2+ chelator; and PD98059, a mitogen-activated protein kinase kinase (MEK)/extracellular signal-related kinase (ERK) inhibitor. cP<0.01 vs Con; eP<0.05 vs ADM 10−7 mol/L.
An OPN polyclonal antibody block VC inhibition by ADM
To determine if the ADM mechanism is dependent upon OPN function, we assessed the effect of ADM treatment during VC in conjunction with an OPN polyclonal antibody. In calcified VSMCs, ADM treatment significantly attenuated the increase in calcium phosphate salt deposition and ALP activity (12.1±5.9 vs 29.1±12.7 IU/g protein, P<0.05). However, calcium phosphate salt deposition and ALP activity was significantly higher when the cells were pretreated with the OPN polyclonal antibody before the addition of ADM (28.7±9.3 for OPN pretreatment vs 12.1±5.9 IU/g protein for ADM treatment alone, P<0.05, Figure 5).
Discussion
In the present study, we discovered that ADM treatment attenuated calcification and prevented the loss of lineage markers in both rat aortas and VSMCs. Interestingly, ADM up-regulated OPN expression in calcified and normal aortas, as well as VSMCs. The effects of ADM on OPN expression were similar to that of the adenylyl cyclase activator forskolin, and the PKA inhibitor H89 blocked ADM modulation of OPN expression. The Ca2+ chelator Fura-2 also blocked the ADM-induced effects, although the ERK inhibitor PD98059 had no significant effect. Moreover, the ability of AMD to prevent VC was blocked by an OPN polyclonal antibody. These results suggest that ADM increases OPN expression through a cAMP/PKA and Ca2+-dependent signaling pathway, which ultimately decreases VC.
ADM is an endogenous, bioactive, 52 amino acid peptide. ADM and its receptor CRLR/RAMPs are widely expressed in tissues that include the vasculature, lung, heart and adipose tissue. ADM is mainly produced by vascular endothelial cells, VSMCs, and macrophages. Considerable evidence exists for a wide range of autocrine, paracrine and endocrine ADM mechanisms that maintain cardiovascular homeostasis31, 32. Previous studies have shown that the mRNA expression levels of ADM and its receptor, CRLR/RAMP2, 3, are upregulated in calcified rat aortas and VSMCs34, 35, 36. We found that exogenous ADM treatment attenuated aortic calcification that was induced by VDN, and similar results were discovered in calcified VSMCs that were induced by beta-glycerophosphate. Our results also confirm that ADM ameliorates calcium deposition, increases ALP activity in VC aortas, and inhibits calcium deposition in calcified VSMCs. All of these results indicate that ADM attenuates VC in the aorta and VSMCs. ADM was previously thought to be an endogenous defensive factor against VC, although the mechanism was unknown.
Ample evidence has suggested that an increase in ALP activity and promotion of osteogenic transition of contractile VSMCs are related to VC4, 27. Thus, arterial calcification is thought to be associated with a loss of SM lineage markers and gain of an osteogenic phenotype, as indicated by increased expression of OPN, BMP-2, osteonectin, and osteocalcin4, 7, 8, 9, 10, 11. In the present study, we saw strikingly decreased levels of SM alpha-actin, enhanced ALP activity, and increased expression of OPN in calcified aortas in vivo. Additionally, in calcified VSMCs the SM alpha-actin expression levels were downregulated. ADM treatment increased the levels of SM alpha-actin both in vivo and in vitro. The results suggest that ADM attenuates the osteogenic transition of contractile VSMCs.
During VC, as VSMCs switch to an osteoblast-like phenotype that expresses BMPs, some proteins, such as OPN and cMGP, are important inhibitors of VC12, 13, 14, 15. OPN is a potent physiological inhibitor of mineralization − it prevents ectopic calcium deposition and is a potent inducible inhibitor of VC42. Previous studies have shown that there are increased OPN protein levels in patients with signs of VC and in several VC models in vivo and in vitro15, 17, 18, 19, 43; additionally, a decrease in OPN expression enhances VC both in vivo and in vitro21, 22. Furthermore, re-expression of OPN in OPN−/− VSMCs rescues the calcification phenotype of these cells22. Thus, OPN may be an important inhibitor of arterial calcification. Our results show that there was an upregulation in OPN mRNA and protein expression in calcified aortas and that exogenous ADM treatment further induced OPN expression. Moreover, ADM also enhanced OPN expression in normal aortas and VSMCs. Finally, an OPN polyclonal antibody blocked the ameliorating effects of ADM on VC. These results suggest that ADM upregulates OPN expression, thus attenuating VC.
In VSMCs, ADM can activate the cAMP/PKA signaling pathway33. Our previous studies have demonstrated that ADM inhibits VC via activating the cAMP/PKA signaling pathway both in vivo and in vitro37, 38. Moreover, two cAMP-response element binding protein (CREBP) binding sites on the promoter of OPN and CREBP can be activated by PKA44. Therefore, we investigated whether ADM also enhances OPN expression through the cAMP/PKA signaling pathway. The PKA inhibitor H89 blocked the ADM-mediated increase in OPN expression, and the adenylyl cyclase agonist forskolin had an effect that was similar to ADM treatment in VSMCs. These results demonstrate that ADM may trigger the cAMP/PKA signaling pathway and activate CREBP, thus inducing OPN expression. The Ca2+ ion chelator Fura-2 also inhibited the ADM-induced upregulation of OPN, although the ERK inhibitor PD98059 had no effect. These results suggest that ADM enhances OPN expression through a cAMP/PKA and Ca2+-dependent, but not a MEK/ERK, signaling pathway.
Author contribution
Yan CAI, Chun-shui PAN, Chao-shu TANG and Yong-fen QI designed the research; Yan CAI, Chun-shui PAN and Xiao-hui DUAN performed the research; Yan CAI, Chun-shui PAN and Xu TENG analyzed the data; and Xu TENG, Chao-shu TANG and Yong-fen QI wrote the paper.
References
Tsuchihashi K, Takizawa H, Torii T, Ikeda R, Nakahara N, Yuda S, et al. Hypoparathyroidism potentiates cardiovascular complications through disturbed calcium metabolism: possible risk of vitamin D(3) analog administration in dialysis patients with end-stage renal disease. Nephron 2000; 84: 13–20.
London GM, Marchais SJ, Guerin AP, Metivier F . Arteriosclerosis, vascular calcifications and cardiovascular disease in uremia. Curr Opin Nephrol Hypertens 2005; 14: 525–31.
Parfitt AM . Soft-tissue calcification in uremia. Arch Intern Med 1969; 124: 544–56.
Johnson RC, Leopold JA, Loscalzo J . Vascular calcification: pathobiological mechanisms and clinical implications. Circ Res 2006; 99: 1044–59.
Iribarren C, Sidney S, Sternfeld B, Browner WS . Calcification of the aortic arch: risk factor and association with coronary heart disease, stroke, and peripheral vascular disease. JAMA 2000; 283: 2810–5.
Wexler L, Brundage B, Crouse J, Detrano R, Fuster V, Addahi JM, et al. Coronary artery calcification: pathophysiology, epidemiology, imaging methods, and clinical implications. A statement for health professionals from the American Heart Association. Circulation 1996; 94: 1175–92.
Shao JS, Cai J, Towler DA . Molecular mechanisms of vascular calcification: lessons learned from the aorta. Arterioscler Thromb Vasc Biol 2006; 26: 1423–30.
Demer LL, Tintut Y . Vascular calcification: pathobiology of a multifaceted disease. Circulation 2008; 117: 2938–48.
Steitz SA, Speer MY, Curinga G, Yang HY, Haynes P, Aebersold R, et al. Smooth muscle cell phenotypic transition associated with calcification: upregulation of Cbfa1 and downregulation of smooth muscle lineage markers. Circ Res 2001; 89: 1147–54.
Tyson KL, Reynolds JL, McNair R, Zhang Q, Weissberg PL, Shanahan CM . Osteo/chondrocytic transcription factors and their target genes exhibit distinct patterns of expression in human arterial calcification. Arterioscler Thromb Vasc Biol 2003; 23: 489–94.
Moe SM, Duan D, Doehle BP, O'Neill KD, Chen NX . Uremia induces the osteoblast differentiation factor Cbfa1 in human blood vessels. Kidney Int 2003; 63: 1003–11.
Jahnen-Dechent W, Schäfer C, Ketteler M, McKee MD . Mineral chaperones: a role for fetuin-A and osteopontin in the inhibition and regression of pathologic calcification. J Mol Med 2008; 86: 379–89.
Engelse MA, Neele JM, Bronckers AL, Pannekoek H, de Vries CJ . Vascular calcification: expression patterns of the osteoblast-specific gene core binding factor alpha-1 and the protective factor matrix gla protein in human atherogenesis. Cardiovasc Res 2001; 52: 281–9.
Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, Behringer RR, et al. Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature 1997; 386: 78–81.
Cai Y, Xu MJ, Teng X, Zhou YB, Chen L, Zhu Y, et al. Intermedin inhibits vascular calcification by increasing the level of matrix gamma-carboxyglutamic acid protein. Cardiovasc Res 2010; 85: 864–73.
Okamoto H . Osteopontin and cardiovascular system. Mol Cell Biochem 2007; 300: 1–7.
Mohler ER 3rd, Adam LP, McClelland P, Graham L, Hathaway DR . Detection of osteopontin in calcified human aortic valves. Arterioscler Thromb Vasc Biol 1997; 17: 547–52.
Canfield AE, Farrington C, Dziobon MD, Boot-Handford RP, Heagerty AM, Kumar SN, et al. The involvement of matrix glycoproteins in vascular calcification and fibrosis: an immunohistochemical study. J Pathol 2002; 196: 228–34.
Aryan M, Kepez A, Atalar E, Hazirolan T, Haznedaroglu I, Akata D, et al. Association of plasma osteopontin levels with coronary calcification evaluated by tomographic coronary calcium scoring. J Bone Miner Metab 2009; 27: 591–7.
Gericke A, Qin C, Spevak L, Fujimoto Y, Butler WT, Sørensen ES, et al. Importance of phosphorylation for osteopontin regulation of biomineralization. Calcif Tissue Int 2005; 77: 45–54.
Speer MY, McKee MD, Guldberg RE, Liaw L, Yang HY, Tung E, et al. Inactivation of the osteopontin gene enhances vascular calcification of matrix Gla protein-deficient mice: evidence for osteopontin as an inducible inhibitor of vascular calcification in vivo. J Exp Med 2002; 196: 1047–55.
Speer MY, Chien YC, Quan M, Yang HY, Vali H, McKee MD, et al. Smooth muscle cells deficient in osteopontin have enhanced susceptibility to calcification in vitro. Cardiovasc Res 2005; 66: 324–33.
Kaartinen MT, Murshed M, Karsenty G, McKee MD . Osteopontin upregulation and polymerization by transglutaminase 2 in calcified arteries of Matrix Gla protein-deficient mice. J Histochem Cytochem 2007; 55: 375–86.
Huang Z, Li J, Jiang Z, Qi Y, Tang C, Du J . Effects of adrenomedullin, C-type natriuretic peptide, and parathyroid hormone-related peptide on calcification in cultured rat vascular smooth muscle cells. J Cardiovasc Pharmacol 2003; 42: 89–97.
Cai DY, Yu F, Jiang W, Jiang HF, Pan CS, Qi YF, et al. Adrenomedullin(27–52) inhibits vascular calcification in rats. Regul Pept 2005; 129: 125–32.
Li GZ, Jiang W, Zhao J, Pan CS, Cao J, Tang CS, et al. Ghrelin blunted vascular calcification in vivo and in vitro in rats. Regul Pept 2005; 129: 167–76.
Jono S, Nishizawa Y, Shioi A, Morii H . 1,25-Dihydroxyvitamin D3 increases in vitro vascular calcification by modulating secretion of endogenous parathyroid hormone-related peptide. Circulation 1998; 98: 1302–6.
Essalihi R, Ouellette V, Hao Dao H, McKee MD, Moreau P . Phenotypic modulation of vascular smooth muscle cells during medial arterial calcification: a role for endothelin? J Cardiovasc Pharmacol 2004; 44: S147–S150.
Jaffe IZ, Mendelsohn ME . Angiotensin II and aldosterone regulate gene transcription via functional mineralocortocoid receptors in human coronary artery smooth muscle cells. Circ Res 2005; 96: 643–50.
Kitamura K, Kangawa K, Kawamoto M, Ichiki Y, Nakamura S, Matsuo H, et al. Adrenomedullin: a novel hypotensive peptide isolated from human pheochromocytoma. Biochem Biophys Res Commun 1993; 192: 553–60.
Yanagawa B, Nagaya N . Adrenomedullin: molecular mechanisms and its role in cardiac disease. Amino Acids. 2007; 32: 157–64.
Gibbons C, Dackor R, Dunworth W, Fritz-Six K, Caron KM . Receptor activity-modifying proteins: RAMPing up adrenomedullin signaling. Mol Endocrinol 2007; 21: 783–96.
Ishizaka Y, Ishizaka Y, Tanaka M, Kitamura K, Kangawa K, Minamino N, et al. Adrenomedullin stimulates cyclic AMP formation in rat vascular smooth muscle cells. Biochem Biophys Res Commun 1994; 200: 642–6.
Qi YF, Wang SH, Zhang BH, Bu DF, Shu TC, Du JB . Changes in amount of ADM mRNA and RAMP2 mRNA in calcified vascular smooth muscle cells. Peptides 2003; 24: 287–94.
Pan CS, Qi YF, Wang SH, Zhao J, Bu DF, Li GZ, et al. Alterations of adrenomedullin and its receptor system components in calcified vascular smooth muscle cells. Regul Pept 2004; 120: 77–83.
Pan CS, Qi YF, Wu SY, Jiang W, Li GZ, Tang CS . The role of adrenomedullin and its receptor system in cardiovascular calcification of rat induced by vitamin D(3) plus nicotine. Peptides 2004; 25: 601–8.
Huang Z, Li J, Jiang Z, Qi Y, Tang C, Du J . Effects of adrenomedullin, C-type natriuretic peptide, and parathyroid hormone-related peptide on calcification in culture rat vascular smooth muscle cells. J Cardiovasc Pharmacol 2003; 42: 89–97.
Zhang B, Tang C, Jiang Z, Qi Y, Pang Y, Du J . Effects of adrenomedullin on vascular calcification in rats. Z Kardiol 2002; 91: 568–74.
Shioi A, Nishizawa Y, Jono S, Koyama H, Hosoi M, Morii H . Beta-glycerophosphate accelerates calcification in cultured bovine vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 1995; 15: 2003–9.
Schurgers LJ, Spronk HM, Skepper JN, Hackeng TM, Shanahan CM, Vermeer C, et al. Post-translational modifications regulate matrix Gla protein function: importance for inhibition of vascular smooth muscle cell calcification. J Thromb Haemost 2007; 5: 2503–11.
Proudfoot D, Skepper JN, Shanahan CM, Weissberg PL . Calcification of human vascular cells in vitro is correlated with high levels of matrix Gla protein and low levels of osteopontin expression. Arterioscler Thromb Vasc Biol 1998; 18: 379–88.
Scatena M, Liaw L, Giachelli CM . Osteopontin: a multifunctional molecule regulating chronic inflammation and vascular disease. Arterioscler Thromb Vasc Biol 2007; 27: 2302–9.
Ohmori R, Momiyama Y, Taniguchi H, Takahashi R, Kusuhara M, Nakamura H, et al. Plasma osteopontin levels are associated with the presence and extent of coronary artery disease. Atherosclerosis 2003; 170: 333–7.
Jalvy S, Renault MA, Leen LL, Belloc I, Bonnet J, Gadeau AP, et al. Autocrine expression of osteopontin contributes to PDGF-mediated arterial smooth muscle cell migration. Cardiovasc Res 2007; 75: 738–47.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No 30470693, 30871013 to Yong-fen QI) , the State Major Basic Research Development Program of the People's Republic of China (No 2006CB503807 to Yong-fen QI), and the Beijing Natural Science Foundation of the People's Republic of China (No 7052041 to Yong-fen QI).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cai, Y., Teng, X., Pan, Cs. et al. Adrenomedullin up-regulates osteopontin and attenuates vascular calcification via the cAMP/PKA signaling pathway. Acta Pharmacol Sin 31, 1359–1366 (2010). https://doi.org/10.1038/aps.2010.89
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2010.89
Keywords
This article is cited by
-
Flavocoxid Ameliorates Aortic Calcification Induced by Hypervitaminosis D3 and Nicotine in Rats Via Targeting TNF-α, IL-1β, iNOS, and Osteogenic Runx2
Cardiovascular Drugs and Therapy (2022)
-
C-type natriuretic peptide inhibiting vascular calcification might involve decreasing bone morphogenic protein 2 and osteopontin levels
Molecular and Cellular Biochemistry (2014)
-
Frontiers of vascular biology and disease research
Acta Pharmacologica Sinica (2010)