Abstract
Aim:
To investigate the effect of icariin on the expression of peroxisome proliferator-activated receptor γ coactivator-1 alpha (PGC-1α), peroxisome proliferator-activated receptor alpha (PPARα), and nuclear respiratory factor 1 (NRF-1) on cardiomyocyte differentiation of murine embryonic stem (ES) cells in vitro.
Methods:
The cardiomyocytes derived from murine ES cells were verified by immunocytochemistry using confocal laser scanning microscopy. Cardiac-specific sarcomeric proteins (ie α-actinin, troponin T) were evaluated when embryoid bodies (EB) were treated with icariin or retinoid acid. The expression of PGC-1α, PPARα, and NRF-1 were analyzed using both semiquantitative RT-PCR and Western blotting in cardiomyocyte differentiation. The phosphorylation of the p38 mitogen-activated protein kinase (MAPK) was studied in the differentiation process, and its specific inhibitor SB203580 was employed to confirm the function of the p38 MAPK on icariin-induced cardiac differentiation.
Results:
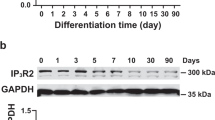
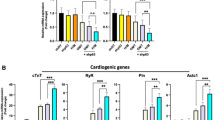
The application of icariin significantly induced the cardiomyocyte differentiation of EB as indicated by the promoted expression of α-actinin and troponin T. The expression of PGC-1α, PPARα, and NRF-1 increased coincidently in early differentiation and the increase was dose-dependently upregulated by icariin treatment. The phosphorylation of the p38 MAPK peaked on d 6 and decreased after d 8, and the activation was further enhanced and prolonged when the EB were subjected to icariin, which was concurrent with the elevation of PGC-1α, PPARα, and NRF-1. Moreover, the inhibition of the p38 MAPK pathway by SB203580 efficiently abolished icariin-stimulated cardiomyocyte differentiation and resulted in the capture of the upregulation of PGC-1α, PPARα, and NRF-1.
Conclusion:
Taken together, icariin promoted the expression of PGC-1α, PPARα, and NRF-1 during cardiomyocyte differentiation of murine ES cells in vitro and the effect was partly responsible for the activation of the p38 MAPK.
Similar content being viewed by others
Article PDF
References
Scarpulla RC . Transcriptional activators and coactivators in the nuclear control of mitochondrial function in mammalian cells. Gene 2002; 286: 81–9.
Scarpulla RC . Nuclear activators and coactivators in mammalian mitochondrial biogenesis. Biochim Biophys Acta 2002; 1576: 1–14.
Barger PM, Kelly DP . PPAR signaling in the control of cardiac energy metabolism. Trends Cardiovasc Med 2000; 10: 238–45.
Brandt JM, Djouadi F, Kelly DP . Fatty acids activate transcription of the muscle carnitine palmitoyltransferase I gene in cardiac myocytes via the peroxisome proliferator-activated receptor alpha. J Biol Chem 1998; 273: 23786–92.
Gulick T, Cresci S, Caira T, Moore DD, Kelly DP . The peroxi-some proliferator-activated receptor regulates mitochondrial fatty acid oxidative enzyme gene expression. Proc Natl Acad Sci USA 1994; 91: 11012–6.
Yu GS, Lu YC, Gulick T . Co-regulation of tissue-specific alternative human carnitine palmitoyltransferase Ibeta gene promoters by fatty acid enzyme substrate. J Biol Chem 1998; 273: 32901–9.
Puigserver P, Spiegelman BM . Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev 2003; 24: 78–90.
Knutti D, Kralli A . PGC-1, a versatile coactivator. Trends Endocrinol Metab 2001; 12: 360–5.
Lehman JJ, Barger PM, Kovacs A, Saffitz JE, Medeiros DM, Kelly DP . Peroxisome proliferator-activated receptor gamma coactivator-1 promotes cardiac mitochondrial biogenesis. J Clin Invest 2000; 106: 847–56.
Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell 1999; 98: 115–24.
Vega RB, Huss JM, Kelly DP . The coactivator PGC-1 cooperates with peroxisome proliferator-activated receptor alpha in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes. Mol Cell Biol 2000; 20: 1868–76.
Kliewer SA, Forman BM, Blumberg B, Ong ES, Borgmeyer U, Mangelsdorf DJ, et al. Differential expression and activation of a family of murine peroxisome proliferator-activated receptors. Proc Natl Acad Sci USA 1994; 91: 7355–9.
Akimoto T, Pohnert SC, Li P, Zhang M, Gumbs C, Rosenberg PB, et al. Exercise stimulates Pgc-1alpha transcription in skeletal muscle through activation of the p38 MAPK pathway. J Biol Chem 2005; 280: 19587–93.
Puigserver P, Rhee J, Lin J, Wu Z, Yoon JC, Zhang C Y, et al. Cytokine stimulation of energy expenditure through p38 MAP kinase activation of PPARgamma coactivator-1. Mol Cell 2001; 8: 971–82.
Knutti D, Kressler D, Kralli A . Regulation of the transcriptional coactivator PGC-1 via MAPK-sensitive interaction with a repressor. Proc Natl Acad Sci USA 2001; 98: 9713–8.
He W, Sun H, Yang B, Zhang D, Kabelitz D . Immunoregulatory effects of the herba Epimediia glycoside icariin. Arzneimittel-forschung 1995; 45: 910–3.
Zhu D, Qu L, Zhang X, Lou Y . Icariin-mediated modulation of cell cycle and p53 during cardiomyocyte differentiation in embryonic stem cells. Eur J Pharmacol 2005; 514: 99–110.
Zhu DY, Lou YJ . Inducible effects of icariin, icaritin, and desmethylicaritin on directional differentiation of embryonic stem cells into cardiomyocytes in vitro. Acta Pharmacol Sin 2005; 26: 477–85.
Boheler KR, Czyz J, Tweedie D, Yang HT, Anisimov SV, Wobus AM . Differentiation of pluripotent embryonic stem cells into cardiomyocytes. Circ Res 2002; 91: 189–201.
Miller-Hance WC, LaCorbiere M, Fuller SJ, Evans SM, Lyons G, Schmidt C, et al. In vitro chamber specification during embryonic stem cell cardiogenesis. Expression of the ventricular myosin light chain-2 gene is independent of heart tube formation. J Biol Chem 1993; 268: 25244–52.
Wobus AM, Wallukat G, Hescheler J . Pluripotent mouse embryonic stem cells are able to differentiate into cardiomyocytes expressing chronotropic responses to adrenergic and cholinergic agents and Ca2+channel blockers. Differentiation 1991; 48: 173–82.
Metzger JM, Lin WI, Samuelson LC . Vital staining of cardiac myocytes during embryonic stem cell cardiogenesis in vitro. Circ Res 1996; 78: 547–52.
Wei H, Juhasz O, Li J, Tarasova YS, Boheler KR . Embryonic stem cells and cardiomyocyte differentiation: phenotypic and molecular analyses. J Cell Mol Med 2005; 9: 804–17.
Wobus AM, Kaomei G, Shan J, Wellner MC, Rohwedel J, Ji G, et al. Retinoic acid accelerates embryonic stem cell-derived cardiac differentiation and enhances development of ventricular cardio-myocytes. J Mol Cell Cardiol 1997; 29: 1525–39.
Irrcher I, Adhihetty PJ, Sheehan T, Joseph AM, Hood DA . PPARgamma coactivator-1alpha expression during thyroid hormone- and contractile activity-induced mitochondrial adaptations. Am J Physiol Cell Physiol 2003; 284: C1669–77.
Attardi G, Schatz G . Biogenesis of mitochondria. Annu Rev Cell Biol 1988; 4: 289–333.
Makinde AO, Kantor PF, Lopaschuk GD . Maturation of fatty acid and carbohydrate metabolism in the newborn heart. Mol Cell Biochem 1998; 188: 49–56.
Braissant O, Wahli W . Differential expression of peroxisome proliferator-activated receptor-alpha, -beta, and -gamma during rat embryonic development. Endocrinology 1998; 139: 2748–54.
Steinmetz M, Quentin T, Poppe A, Paul T, Jux C . Changes in expression levels of genes involved in fatty acid metabolism: upregulation of all three members of the PPAR family (alpha, gamma, delta) and the newly described adiponectin receptor 2, but not adiponectin receptor 1 during neonatal cardiac development of the rat. Basic Res Cardiol 2005; 100: 263–9.
Kraft CS, LeMoine CM, Lyons CN, Michaud D, Mueller CR, Moyes CD . Control of mitochondrial biogenesis during myogenesis. Am J Physiol Cell Physiol 2006; 290: C1119–27.
Marin-Garcia J, Ananthakrishnan R, Goldenthal MJ . Heart mi-tochondrial DNA and enzyme changes during early human development. Mol Cell Biochem 2000; 210: 47–52.
Herzberg NH, Middelkoop E, Adorf M, Dekker HL, Van Galen MJ, Van den Berg M, et al. Mitochondria in cultured human muscle cells depleted of mitochondrial DNA. Eur J Cell Biol 1993; 61: 400–8.
Barger PM, Browning AC, Garner AN, Kelly DP . p38 mitogen-activated protein kinase activates peroxisome proliferator-activated receptor alpha: a potential role in the cardiac metabolic stress response. J Biol Chem 2001; 276: 44495–501.
Davidson SM, Morange M . Hsp25 and the p38 MAPK pathway are involved in differentiation of cardiomyocytes. Dev Biol 2000; 218: 146–60.
Eriksson M, Leppa S . Mitogen-activated protein kinases and activator protein 1 are required for proliferation and cardio-myocyte differentiation of P19 embryonal carcinoma cells. J Biol Chem 2002; 277: 15992–6001.
Aouadi M, Bost F, Caron L, Laurent K, Le Marchand Brustel Y, Binetruy B . p38 mitogen-activated protein kinase activity commits embryonic stem cells to either neurogenesis or cardio-myogenesis. Stem Cells 2006; 24: 1399–406.
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the National Natural Sciences Foundation of China (No 30171121, 30472112, and 30672564), the Key Grant of the Chinese Ministry of Education (No 03088), and the International Joint Key Grant of Zhejiang Province (No 2003C24005).
Rights and permissions
About this article
Cite this article
Ding, L., Liang, Xg., Zhu, Dy. et al. Icariin promotes expression of PGC-1α, PPARα, and NRF-1 during cardiomyocyte differentiation of murine embryonic stem cells in vitro. Acta Pharmacol Sin 28, 1541–1549 (2007). https://doi.org/10.1111/j.1745-7254.2007.00648.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00648.x
Keywords
This article is cited by
-
Transcriptome analysis of intestine from alk-SMase knockout mice reveals the effect of alk-SMase
Cancer Cell International (2022)