Abstract
Aim:
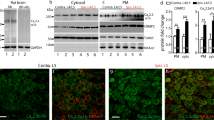
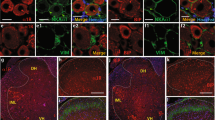
The present study aimed to elucidate the role of T-subtype calcium channels (Cav3.1, Cav3.2, and Cav3.3) in the pathogenesis of neuropathic pain at spinal level.
Methods:
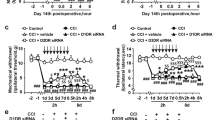
The chronic compression of the dorsal root ganglion (CCD) rat model was adopted. The antisense oligonucleotide of Cav3.1, Cav3.2, and Cav3.3 or normal saline (NS) were intrathecally administered twice per day from the first day to the fourth day after operation. Paw mechanical withdrawal threshold and paw thermal withdrawal latency were measured to evaluate the tactile allodynia and thermal hyperalgesia, respectively.
Results:
CCD rats developed reliable tactile allodynia and thermal hyperalgesia after operation. Intrathecal administration of antisense oligonucleotide of Cav3.2 and Cav3.3 significantly relieved tactile allodynia and thermal hyperalgesia in CCD rats, but not Cav3.1.
Conclusion:
Cav3.2 and Cav3.3 subtype calcium channels in the spinal cord may play an important role in the pathogenesis of neuropathic pain, which may contribute to the management of the neuropathic pain.
Similar content being viewed by others
Article PDF
References
Woolf CJ, Salter MW . Neuronal plasticity: increasing the gain in pain. Science 2000; 288: 1765–9.
Bridges D, Thompson SWN, Rice ASC . Mechanisms of neuropathic pain. Br J Anaesth 2001; 87: 12–26.
Perez-Reyes E . Molecular physiology of low-voltage-activated t-type calcium channels. Physiol Rev 2003; 83: 117–61.
Bertolino M, Llinas RR . The central role of voltage-activated and receptor-operated calcium channels in neuronal cells. Annu Rev Pharmacol Toxicol 1992; 32: 399–421.
Kostyuk PG . Low-voltage activated calcium channels: achievements and problems. Neuroscience 1999; 92: 1157–63.
Slobodan MT, Adam M, Vesna JT . Mechanical and thermal antinociception in rats following systemic administration of mibefradil, a T-type calcium channel blocker. Brain Res 2002; 951: 336–40.
Ikeda Heinke HB, Ruscheweyh R, Sandkuhler J . Synaptic plasticity in spinal lamina I projection neurons that mediate hyper-algesia. Science 2003; 299: 1237–40.
Dogrul A, Gardell LR, Ossipov MH, Tulunay FC, Lai J, Porreca F . Reversal of experimental neuropathic pain by T-type calcium channel blockers. Pain 2003; 105: 159–68.
Kim C, Jun K, Lee T, Kim SS, McEnery MW, Chin H, et al. Altered nociceptive response in mice deficient in the alpha (1B) subunit of the voltage-dependent calcium channel. Mol Cell Neurosci 2001; 18: 235–45.
Yaksh TL, Rudy TA . Chronic catheterization of the spinal sub-arachnoid space. Physiol Behav 1976; 17: 1031–6.
Hu SJ, Xing JL . An experimental model for chronic compression of dorsal root ganglion produced by intervertebral foramen stenosis in the rat. Pain 1998; 77: 15–23.
Hargreaves K, Dubner R, Brown F, Flores C, Joris J . A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988; 32: 77–88.
Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL . Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 1994; 53: 55–63.
Khosravani H, Altier C, Simms B, Hamming KS, Snutch TP, Mezeyova J . Gating effects of mutations in the Cav3.2 T type calcium channel associated with childhood absence epilepsy. J Biol Chem 2004; 279: 9681–4.
Todorovic SM, Jevtovic-Todorovic V, Meyenburg A, Mennerick S, Perez-Reyes E, Romano C, et al. Redox modulation of T-type calcium channels in rat peripheral nociceptors. Neuron 2001; 31: 75–85.
Beedle AM . Agonist-independent modulation of N-type calcium channels by ORL1 receptors. Nat Neurosci 2004; 7: 118–25.
Kim D . Thalamic control of visceral nociception mediated by T-type Ca2+ channels. Science 2003; 302: 117–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wen, Xj., Li, Zj., Chen, Zx. et al. Intrathecal administration of Cav3.2 and Cav3.3 antisense oligonucleotide reverses tactile allodynia and thermal hyperalgesia in rats following chronic compression of dorsal root of ganglion. Acta Pharmacol Sin 27, 1547–1552 (2006). https://doi.org/10.1111/j.1745-7254.2006.00461.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00461.x
Keywords
This article is cited by
-
Central and peripheral contributions of T-type calcium channels in pain
Molecular Brain (2022)
-
Voltage-dependent CaV3.2 and CaV2.2 channels in nociceptive pathways
Pflügers Archiv - European Journal of Physiology (2022)
-
5-Oxo-hexahydroquinoline: an attractive scaffold with diverse biological activities
Molecular Diversity (2019)
-
Surfen is a broad-spectrum calcium channel inhibitor with analgesic properties in mouse models of acute and chronic inflammatory pain
Pflügers Archiv - European Journal of Physiology (2017)
-
Two heterozygous Cav3.2 channel mutations in a pediatric chronic pain patient: recording condition-dependent biophysical effects
Pflügers Archiv - European Journal of Physiology (2016)